Poster Session A
Epidemiology, health policy and outcomes
Session: (0325–0344) Patient Outcomes, Preferences, & Attitudes Poster I
0325: Understanding the Burden of Cutaneous Lupus: A Subset Analysis from the 2022 World Lupus Federation Global Impact (WLFGI) Patient Survey
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- NE
Nnenna Ezeh, MD (she/her/hers)
Brigham and Women's Hospital
Boston, MA, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Nnenna Ezeh1, Joy Buie2, Mike Donnelly3, Daria McClamb3, Lydia Oberholtzer4, Jana Sharp4 and Joseph Merola5, 1Department of Dermatology; Department of Medicine, Harvard Medical School, Brigham and Women’s Hospital, Boston, MA, 2Lupus Foundation of America, York, SC, 3Lupus Foundation of America, Washington, DC, 4Sharp Insight, LLC, Montgomery County, MD, 5Harvard Medical School, Brigham and Women's Hospital, Newton, MA
Background/Purpose: There is a paucity of data among a global population exploring the burden of CLE on patients, especially as compared to patients with SLE. This has negatively impacted research efforts including the development of new therapeutics specific to CLE. The 2022 World Lupus Federation Global Impact (WLFGI) patient survey assessed multiple aspects of patients’ lupus including subsets of disease, therapeutic beliefs, quality of life burden. We utilized the WLFGI survey to characterize the burden of cutaneous disease and the comparative burden on quality of life among patients with self-reported cutaneous lupus compared to systemic lupus erythematosus.
Methods: The 2022 WLFGI survey was a multilingual, anonymous, online survey designed by the World Lupus Federation and disseminated worldwide via email during 3/1/2022-3/22/2023 with 6,704 respondents. 199 respondents had cutaneous lupus (CLE only) compared to 2,929 with systemic lupus with skin involvement (CLE+SLE) and 2,094 with systemic lupus without skin involvement (SLE only). Respondents with incomplete diagnosis data were removed from analysis, however, incomplete responses to questions were included as negative responses. Comparative statistics (odds ratio analysis) was utilized to assess comparative burden of quality of life measures between subgroups.
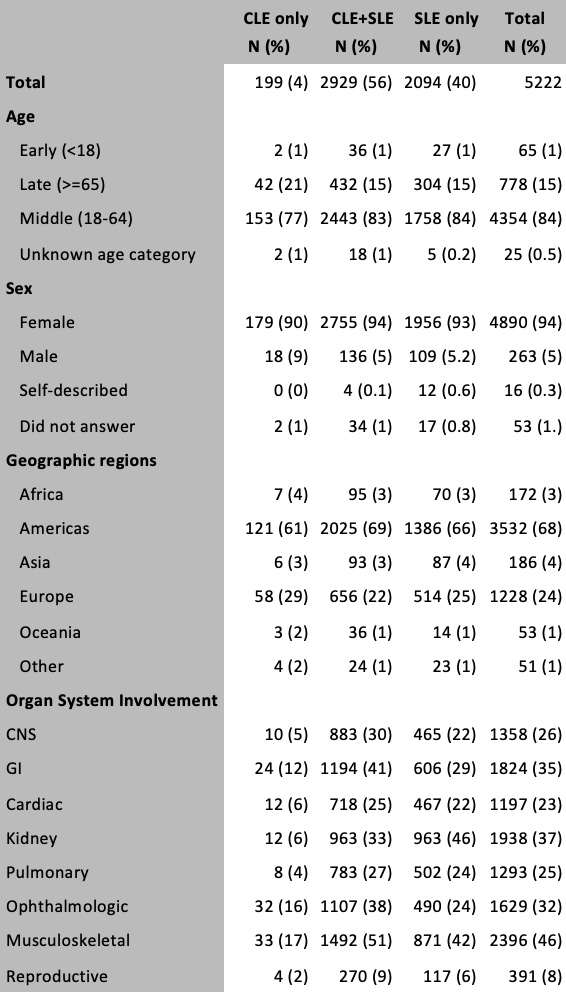
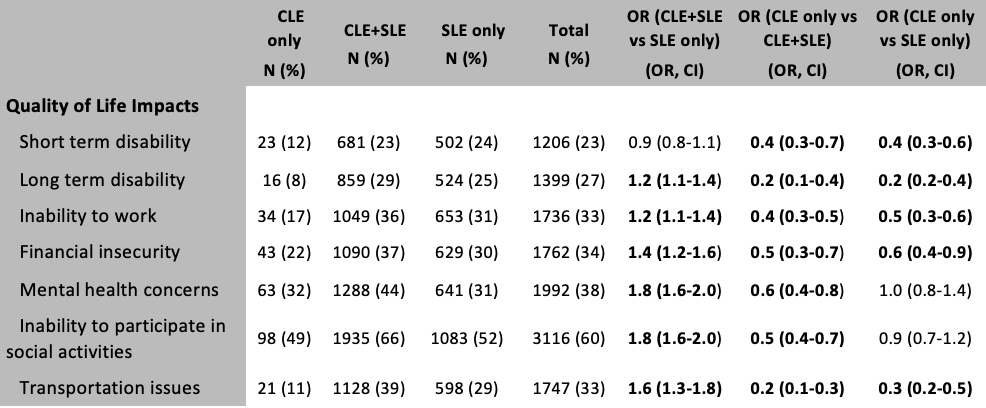
Results: Patient demographics and clinical manifestations are noted in Table 1. Respondents were more likely to be middle age, female, from the Americas and Europe. Impact on quality of life was observed in all groups, particularly financial insecurity (34%), mental health concerns (38%) and decreased social engagement (60%) (Table 2). There was an additive quality of life burden of 20-80% from cutaneous disease for all quality of life measures except short term disability observed in the CLE+SLE group, compared to the SLE only group. While respondents with cutaneous-limited disease generally had less quality of life burden compared to the SLE-only subgroup, mental health concerns and inabilities to socially engage were notable outliers. About one-third of CLE-only patients reported mental health concerns (32%), similar to the SLE-only population (31%), with CLE+SLE patients having nearly half (44%) reporting mental health concerns, highlighting the significant stigma associated with a potentially disfiguring disease.
Conclusion: This study offers a unique, global insight into the patient-reported burden of CLE +/- SLE. Skin-limited disease appears to carry as high a burden as SLE in several domains of disease impact. The presence of CLE among SLE patients is associated with higher quality of life burden, with particular impact on mental health and social interaction. These data support the need for further research dedicated to CLE and CLE disease-specific therapeutics.
N. Ezeh: None; J. Buie: None; M. Donnelly: None; D. McClamb: None; L. Oberholtzer: None; J. Sharp: None; J. Merola: Abbvie, 2, 12, Investigator, Amgen, 2, 12, Investigator, Biogen, 2, 12, Investigator, Bristol-Myers Squibb, 2, 12, Investigator, Dermavant, 2, 12, Investigator, Eli Lilly, 2, 6, 12, Investigator, Janssen, 2, 12, Investigator, Leo Pharma, 2, 12, Investigator, Novartis, 2, 12, Investigator, Pfizer, 2, 12, Investigator, Regeneron, 2, 12, Investigator, Sanofi, 2, 12, Investigator, Sun Pharma, 2, 12, Investigator, UCB Pharma, 2, 12, Investigator.
Background/Purpose: There is a paucity of data among a global population exploring the burden of CLE on patients, especially as compared to patients with SLE. This has negatively impacted research efforts including the development of new therapeutics specific to CLE. The 2022 World Lupus Federation Global Impact (WLFGI) patient survey assessed multiple aspects of patients’ lupus including subsets of disease, therapeutic beliefs, quality of life burden. We utilized the WLFGI survey to characterize the burden of cutaneous disease and the comparative burden on quality of life among patients with self-reported cutaneous lupus compared to systemic lupus erythematosus.
Methods: The 2022 WLFGI survey was a multilingual, anonymous, online survey designed by the World Lupus Federation and disseminated worldwide via email during 3/1/2022-3/22/2023 with 6,704 respondents. 199 respondents had cutaneous lupus (CLE only) compared to 2,929 with systemic lupus with skin involvement (CLE+SLE) and 2,094 with systemic lupus without skin involvement (SLE only). Respondents with incomplete diagnosis data were removed from analysis, however, incomplete responses to questions were included as negative responses. Comparative statistics (odds ratio analysis) was utilized to assess comparative burden of quality of life measures between subgroups.
Results: Patient demographics and clinical manifestations are noted in Table 1. Respondents were more likely to be middle age, female, from the Americas and Europe. Impact on quality of life was observed in all groups, particularly financial insecurity (34%), mental health concerns (38%) and decreased social engagement (60%) (Table 2). There was an additive quality of life burden of 20-80% from cutaneous disease for all quality of life measures except short term disability observed in the CLE+SLE group, compared to the SLE only group. While respondents with cutaneous-limited disease generally had less quality of life burden compared to the SLE-only subgroup, mental health concerns and inabilities to socially engage were notable outliers. About one-third of CLE-only patients reported mental health concerns (32%), similar to the SLE-only population (31%), with CLE+SLE patients having nearly half (44%) reporting mental health concerns, highlighting the significant stigma associated with a potentially disfiguring disease.
Conclusion: This study offers a unique, global insight into the patient-reported burden of CLE +/- SLE. Skin-limited disease appears to carry as high a burden as SLE in several domains of disease impact. The presence of CLE among SLE patients is associated with higher quality of life burden, with particular impact on mental health and social interaction. These data support the need for further research dedicated to CLE and CLE disease-specific therapeutics.

Table 1. Cohort demographics and clinical data by disease subphenotype

Table 2. Odds ratio and 95% confidence interval of quality of life impacts by disease subphenotype
N. Ezeh: None; J. Buie: None; M. Donnelly: None; D. McClamb: None; L. Oberholtzer: None; J. Sharp: None; J. Merola: Abbvie, 2, 12, Investigator, Amgen, 2, 12, Investigator, Biogen, 2, 12, Investigator, Bristol-Myers Squibb, 2, 12, Investigator, Dermavant, 2, 12, Investigator, Eli Lilly, 2, 6, 12, Investigator, Janssen, 2, 12, Investigator, Leo Pharma, 2, 12, Investigator, Novartis, 2, 12, Investigator, Pfizer, 2, 12, Investigator, Regeneron, 2, 12, Investigator, Sanofi, 2, 12, Investigator, Sun Pharma, 2, 12, Investigator, UCB Pharma, 2, 12, Investigator.



