Abstract Session
Fibrosing rheumatic diseases (scleroderma, MCTD, IgG4-related disease, scleroderma mimics)
Session: Abstracts: Systemic Sclerosis & Related Disorders II: Clinical Research (1699–1704)
1700: Incidence and Risk Factors for New Onset of Interstitial Lung Disease in Systemic Sclerosis: A EUSTAR Analysis
Monday, November 13, 2023
4:15 PM - 4:25 PM PT
Location: Room 29A-D
- LP
Liubov Petelytska, MD, PhD
Bogomolets National Medical University
Kyiv, UkraineDisclosure information not submitted.
Presenting Author(s)
liubov Petelytska1, Arthiha Velauthapillai2, Lorenzo Tofani3, Eric Hachulla4, Ulf Müller-Ladner5, Elise Siegert6, Yannick ALLANORE7, Gabriela Riemekasten8, Christina Bergmann9, Radim Becvar10, Kamal Solanki11, Branimir Anic12, Simona Rednic13, Bojana Stamenkovic14, Lisa Stamp15, Joerg Distler16, Madelon Vonk17, Jeska de Vries-Bouwstra18, Anna Maria Hoffmann-Vold19, marco Matucci Cerinic20, Oliver Distler21 and Cosimo Bruni21, 1Department of Rheumatology, University Hospital Zurich, University of Zurich, Zürich, Switzerland, 2Department of Rheumatology, Radboud University Medical Center, Nijmegen, Netherlands, 3Department of Statistics, Computer Science, Applications, University of Florence, Florence, Italy, 4University of Lille, Lille, France, 5Justus Liebig University Gießen, Campus Kerckhoff, Bad Nauheim, Germany, 6Department of Rheumatology, Charité University Hospital, Charité Platz 1, D-10117, Berlin, Germany, 7Université Paris Cité, Paris, France, 8University Clinic Schleswit-Holstein (UKSH), Lübeck, Germany, 9Department of Internal Medicine 3-Rheumatology and Immunology, Friedrich-Alexander-University Erlangen-Nürnberg (FAU) and University Hospital Erlangen, Erlangen, Germany, 10Institute of Rheumatology, Prague, Czech Republic, Department of Rheumatology, 1st Faculty of Medicine, Charles University, Praha, Czech Republic, 11Department of Rheumatology, Te Whatu Ora Health New Zealand Waikato, Hamilton, New Zealand, 12Division of Clinical Immunology and Rheumatology, Department of Internal Medicine, University of Zagreb, School of Medicine, University Hospital Center Zagreb, Kišpatićeva 12, 10 000, Zagreb, Croatia, 13Prof Dr Simona Rednic, Cluj, Romania, 14Institut Niska Banja, Niska Banja, Serbia, 15University of Otago, Christchurch, New Zealand, 16Clinic for Rheumatology University Hospital Düsseldorf, Medical Faculty of Heinrich Heine University, Düsseldorf, Germany; Hiller Research Center, University Hospital Düsseldorf, Medical Faculty of Heinrich Heine University, Düsseldorf, Germany, 17Radboud University Nijmegen Medical Centre, Nijmegen, Netherlands, 18Leiden University Medical Center, Leiden, Netherlands, 19Oslo University Hospital, Department of Rheumatology, Oslo, Norway, 20Unit of Immunology, Rheumatology, Allergy and Rare diseases, IRCCS San Raffaele Hospital, Milan, Milan, Italy, 21Department of Rheumatology, University Hospital Zurich, University of Zurich, Zurich, Switzerland
Background/Purpose: Although prevalence of interstitial lung disease (ILD) in systemic sclerosis (SSc) and associated risk factors are established, less is known about its incidence and the risk factors associated with new onset of ILD following a negative baseline. We aimed to (1) estimate the annual incidence of ILD and (2) identify risk factors for new onset of ILD in baseline negative patients.
Methods: SSc patients classified according to the 2013 ACR/EULAR criteria from the EUSTAR database, with absence of ILD on high resolution computed tomography (HRCT) at baseline and having at least 1 follow-up visit were included. Patients with pulmonary arterial hypertension diagnosed by right heart catheterization were excluded. Based on follow-up HRCT, our population was divided into patients with new onset of ILD (incident group) and patients who remained ILD negative (negative group). Incidence of SSc-ILD was calculated as rate per 1000 person-years and presented starting from the first visit at the center. Prediction models were built to identify risk factors for ILD onset, with logistic regression for the 1-year and Cox regression for the long-term observation. Known predictors of new onset of ILD were chosen as covariates from previous literature and based on expert opinion.
Results: We identified 5336 ILD negative patients at baseline. New onset of ILD occurred in 1080 (20.2%) cases with median of 3.8 (IQR 1.6-7.3) years follow-up.
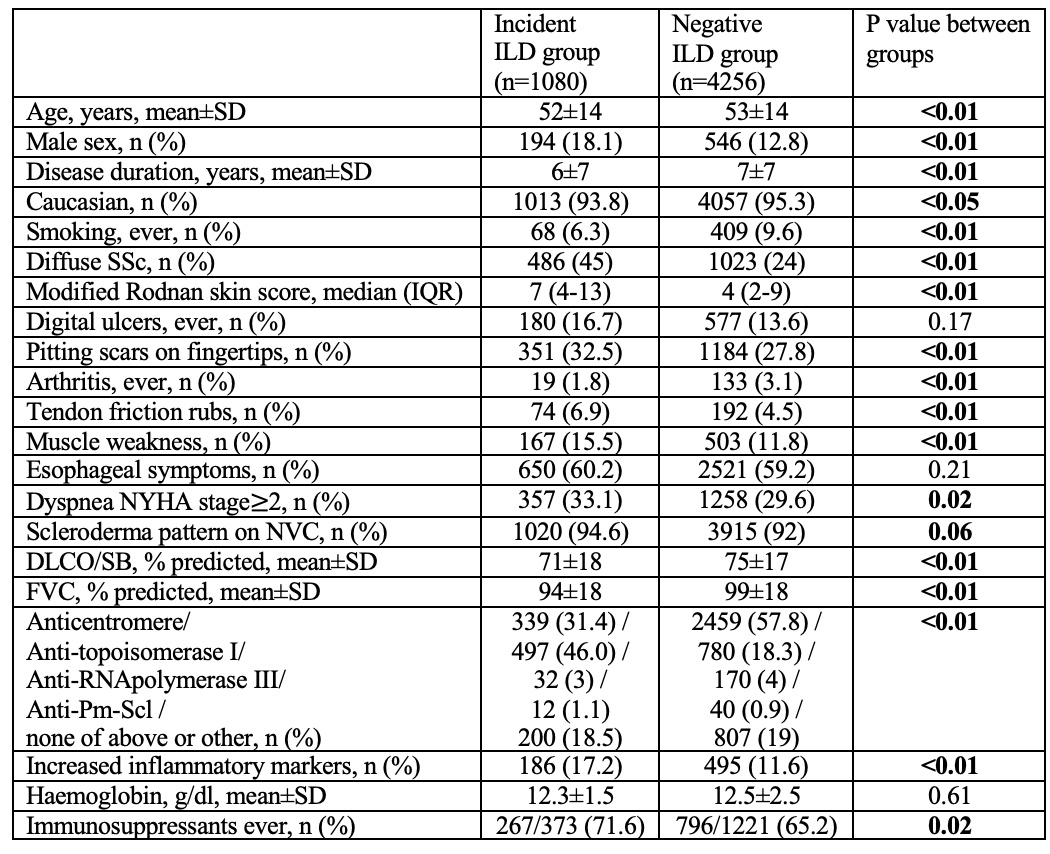
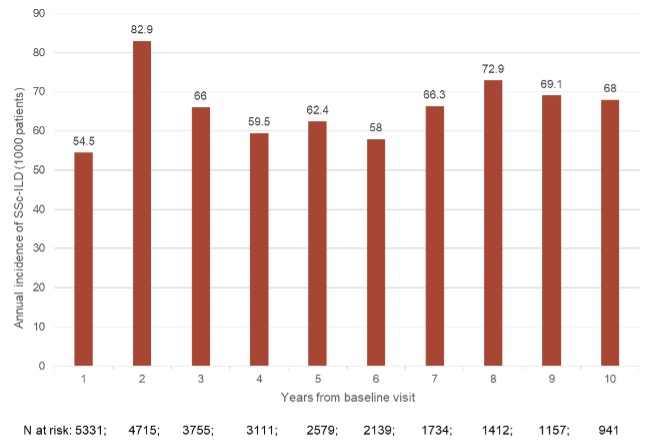
The overall ILD incidence rate was 74.0 (95% CI: 68.5-79.5) per 1000 person-years. There was a continuous detection of new onset of ILD during the longitudinal observation, up to 10 years from baseline (incidence rate 54.5-82.9 per 1000 person-years; Figure 1). The baseline characteristics of incident and negative groups are shown in Table 1.
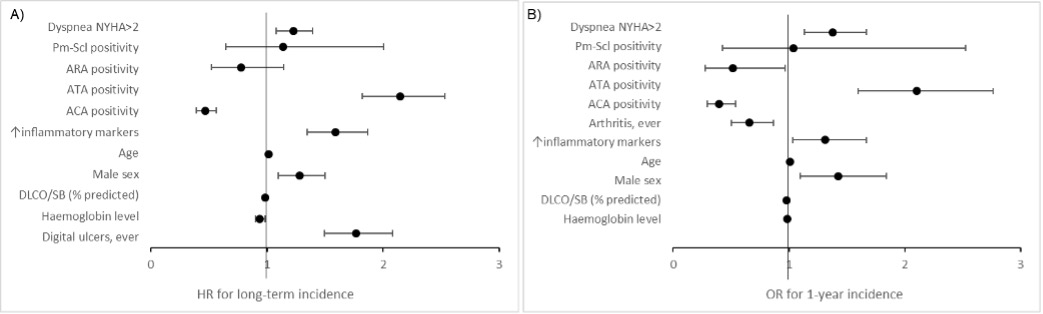
In the analysis of long-term incidence, new onset of ILD was independently predicted by dyspnea NYHA stage≥2 (HR 1.23, 95% CI1.08-1.40), male sex (HR 1.28, 95% CI1.10-1.50), age (HR 1.02, 95% CI 1.01-1.02), DLCO/SB% (HR 0.99, 95% CI 0.98-0.99), elevated inflammatory markers (HR 1.59, 95% CI 1.35-1.87), haemoglobin level (HR 0.94, 95% CI: 0.90-0.98), anti-topoisomerase I antibody (HR 2.15, 95% CI 1.82-2.53), anti-centromere antibody (HR 0.47, 95% CI0.39-0.56,) and digital ulcers ever (HR1.77, 95% CI: 1.49-2.08) (Figure 2 A). Surprisingly, the incidence of new onset of ILD was independent from disease duration.
The analysis for 1-year incidence included 13339 yearly follow-ups from 4067 baseline negative patients. All risk factors of new onset of ILD identified in the analysis of long-term incidence analysis, except for digital ulcers ever, were confirmed (Figure 2B).
When exposure to immunosuppressants was forced into the two models, this was not retained as a protective factor.
Conclusion: ILD can appear at any time after SSc diagnosis, with similar incidence during the disease course. We identified risk factors for new onset of ILD both at 1-year and long-term follow-up. Therefore, SSc patients should be screened following a negative baseline HRCT.
l. Petelytska: None; A. Velauthapillai: None; L. Tofani: None; E. Hachulla: Bayer, 2, CSL Behring, 5, GlaxoSmithKlein(GSK), 2, 5, 6, johnson&Johnson, 2, 5, 6, Novartis, 2, 5, Otsuka, 6, Roche-Chugai, 2, 5, 6, sanofi-genzyme, 2, 5, Sobi, 5; U. Müller-Ladner: None; E. Siegert: None; Y. ALLANORE: AbbVie/Abbott, 2, Alpine Immunoscience, 5, AstraZeneca, 2, Bayer, 2, Boehringer-Ingelheim, 2, Janssen, 2, Medsenic, 2, 5, Mylan, 2, OSE Immunotherapeutics, 5, Prometeus, 2, Roche, 2, Sanofi, 2; G. Riemekasten: None; C. Bergmann: Boehringer-Ingelheim, 2, 5, Janssen, 2; R. Becvar: Boehringer-Ingelheim, 2, GlaxoSmithKlein(GSK), 2; K. Solanki: None; B. Anic: None; S. Rednic: None; B. Stamenkovic: None; L. Stamp: None; J. Distler: 4D Science and FibroCure, 8, 11, AbbVie, Active Biotech, Anamar, ARXX, AstraZeneca, Bayer Pharma, Boehringer Ingelheim, Celgene, Galapagos, Genentech, GSK, Inventiva, Janssen, Novarti, 2, Anamar, Argenx, ARXX, BMS, Bayer Pharma, Boehringer Ingelheim, Cantargia, Celgene, CSL Behring, Galapagos, GSK, 5, Inventiva, Kiniksa, Lassen, Sanofi-Aventis, RedX, UCB, 5; M. Vonk: Boehringer Ingelheim, 5, 6, Corbus, 1, EUSTAR, 4, Ferrer, 5, Galapagos, 5, Janssen, 5, 6, MSD, 6, Systemic Sclerosis ERN ReCONNET, 4; J. de Vries-Bouwstra: AbbVie/Abbott, 2, 6, Boehringer-Ingelheim, 2, 6, galapagos, 5, Janssen, 2, 6, Janssen-Cilag, 5, Roche, 5; A. Hoffmann-Vold: Arxx Therapeutics, 2, Boehringer-Ingelheim, 2, 5, 6, 12, Support for travel, Genentech, 2, Janssen, 2, 5, 6, Medscape, 2, 6, 12, Support for travel, Roche, 2, 6, 12, Support for travel; m. Matucci Cerinic: accelerong, 2, 6, actelion, 2, 6, bayer, 2, 6, biogen, 2, 6, Boehringer-Ingelheim, 2, 6, Chemomab, 2, 6, corbus, 2, 6, CSL Behring, 2, 6, Eli Lilly, 2, 6, galapagos, 2, 6, Inventiva, 2, 6, Janssen, 2, 6, Merck/MSD, 2, 6, Mitsubishi, 2, 6, Pfizer, 2, 6, regeneron, 2, 6, Roche, 2, 6, samsung, 2, 6; O. Distler: 4P-Pharma, 2, 5, 6, AbbVie, 2, 5, 6, Acceleron, 2, 5, 6, Alcimed, 2, 5, 6, Altavant Sciences, 2, 5, 6, Amgen, 2, 5, 6, AnaMar, 2, 5, 6, Arxx, 2, 5, 6, AstraZeneca, 2, 5, 6, Bayer, 2, 5, 6, Blade Therapeutics, 2, 5, 6, Boehringer Ingelheim, 2, 5, 6, Citus AG, 12, Co-Founder, Corbus Pharmaceuticals, 2, 5, 6, CSL Behring, 2, 5, 6, Galapagos, 2, 5, 6, Galderma, 2, 5, 6, Glenmark, 2, 5, 6, Gossamer, 2, 5, 6, Horizon Therapeutics, 2, 5, 6, Janssen, 2, 5, 6, Kymera, 2, 5, 6, Lupin, 2, 5, 6, Medscape, 2, 5, 6, Miltenyi Biotec, 2, 5, 6, Mitsubishi Tanabe, 2, 5, 6, MSD, 2, 5, 6, Novartis, 2, 5, 6, Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143), 10, Prometheus Biosciences, 2, 5, 6, Redx Pharma, 2, 5, 6, Roivant, 2, 5, 6, Topadur, 2, 5, 6; C. Bruni: AbbVie/Abbott, 5, Boehringer-Ingelheim, 2, 12, Travel Support, Eli Lilly, 6.
Background/Purpose: Although prevalence of interstitial lung disease (ILD) in systemic sclerosis (SSc) and associated risk factors are established, less is known about its incidence and the risk factors associated with new onset of ILD following a negative baseline. We aimed to (1) estimate the annual incidence of ILD and (2) identify risk factors for new onset of ILD in baseline negative patients.
Methods: SSc patients classified according to the 2013 ACR/EULAR criteria from the EUSTAR database, with absence of ILD on high resolution computed tomography (HRCT) at baseline and having at least 1 follow-up visit were included. Patients with pulmonary arterial hypertension diagnosed by right heart catheterization were excluded. Based on follow-up HRCT, our population was divided into patients with new onset of ILD (incident group) and patients who remained ILD negative (negative group). Incidence of SSc-ILD was calculated as rate per 1000 person-years and presented starting from the first visit at the center. Prediction models were built to identify risk factors for ILD onset, with logistic regression for the 1-year and Cox regression for the long-term observation. Known predictors of new onset of ILD were chosen as covariates from previous literature and based on expert opinion.
Results: We identified 5336 ILD negative patients at baseline. New onset of ILD occurred in 1080 (20.2%) cases with median of 3.8 (IQR 1.6-7.3) years follow-up.
The overall ILD incidence rate was 74.0 (95% CI: 68.5-79.5) per 1000 person-years. There was a continuous detection of new onset of ILD during the longitudinal observation, up to 10 years from baseline (incidence rate 54.5-82.9 per 1000 person-years; Figure 1). The baseline characteristics of incident and negative groups are shown in Table 1.
In the analysis of long-term incidence, new onset of ILD was independently predicted by dyspnea NYHA stage≥2 (HR 1.23, 95% CI1.08-1.40), male sex (HR 1.28, 95% CI1.10-1.50), age (HR 1.02, 95% CI 1.01-1.02), DLCO/SB% (HR 0.99, 95% CI 0.98-0.99), elevated inflammatory markers (HR 1.59, 95% CI 1.35-1.87), haemoglobin level (HR 0.94, 95% CI: 0.90-0.98), anti-topoisomerase I antibody (HR 2.15, 95% CI 1.82-2.53), anti-centromere antibody (HR 0.47, 95% CI0.39-0.56,) and digital ulcers ever (HR1.77, 95% CI: 1.49-2.08) (Figure 2 A). Surprisingly, the incidence of new onset of ILD was independent from disease duration.
The analysis for 1-year incidence included 13339 yearly follow-ups from 4067 baseline negative patients. All risk factors of new onset of ILD identified in the analysis of long-term incidence analysis, except for digital ulcers ever, were confirmed (Figure 2B).
When exposure to immunosuppressants was forced into the two models, this was not retained as a protective factor.
Conclusion: ILD can appear at any time after SSc diagnosis, with similar incidence during the disease course. We identified risk factors for new onset of ILD both at 1-year and long-term follow-up. Therefore, SSc patients should be screened following a negative baseline HRCT.

Table 1. Baseline characteristics of SSc patients according to the ILD onset at follow up.

Figure 1. Annual incidence rate of new onset of ILD per 1000 patients from the baseline visit.

Figure 2. Prediction models of new onset of ILD for long-term (A) and 1-year observation (B).
l. Petelytska: None; A. Velauthapillai: None; L. Tofani: None; E. Hachulla: Bayer, 2, CSL Behring, 5, GlaxoSmithKlein(GSK), 2, 5, 6, johnson&Johnson, 2, 5, 6, Novartis, 2, 5, Otsuka, 6, Roche-Chugai, 2, 5, 6, sanofi-genzyme, 2, 5, Sobi, 5; U. Müller-Ladner: None; E. Siegert: None; Y. ALLANORE: AbbVie/Abbott, 2, Alpine Immunoscience, 5, AstraZeneca, 2, Bayer, 2, Boehringer-Ingelheim, 2, Janssen, 2, Medsenic, 2, 5, Mylan, 2, OSE Immunotherapeutics, 5, Prometeus, 2, Roche, 2, Sanofi, 2; G. Riemekasten: None; C. Bergmann: Boehringer-Ingelheim, 2, 5, Janssen, 2; R. Becvar: Boehringer-Ingelheim, 2, GlaxoSmithKlein(GSK), 2; K. Solanki: None; B. Anic: None; S. Rednic: None; B. Stamenkovic: None; L. Stamp: None; J. Distler: 4D Science and FibroCure, 8, 11, AbbVie, Active Biotech, Anamar, ARXX, AstraZeneca, Bayer Pharma, Boehringer Ingelheim, Celgene, Galapagos, Genentech, GSK, Inventiva, Janssen, Novarti, 2, Anamar, Argenx, ARXX, BMS, Bayer Pharma, Boehringer Ingelheim, Cantargia, Celgene, CSL Behring, Galapagos, GSK, 5, Inventiva, Kiniksa, Lassen, Sanofi-Aventis, RedX, UCB, 5; M. Vonk: Boehringer Ingelheim, 5, 6, Corbus, 1, EUSTAR, 4, Ferrer, 5, Galapagos, 5, Janssen, 5, 6, MSD, 6, Systemic Sclerosis ERN ReCONNET, 4; J. de Vries-Bouwstra: AbbVie/Abbott, 2, 6, Boehringer-Ingelheim, 2, 6, galapagos, 5, Janssen, 2, 6, Janssen-Cilag, 5, Roche, 5; A. Hoffmann-Vold: Arxx Therapeutics, 2, Boehringer-Ingelheim, 2, 5, 6, 12, Support for travel, Genentech, 2, Janssen, 2, 5, 6, Medscape, 2, 6, 12, Support for travel, Roche, 2, 6, 12, Support for travel; m. Matucci Cerinic: accelerong, 2, 6, actelion, 2, 6, bayer, 2, 6, biogen, 2, 6, Boehringer-Ingelheim, 2, 6, Chemomab, 2, 6, corbus, 2, 6, CSL Behring, 2, 6, Eli Lilly, 2, 6, galapagos, 2, 6, Inventiva, 2, 6, Janssen, 2, 6, Merck/MSD, 2, 6, Mitsubishi, 2, 6, Pfizer, 2, 6, regeneron, 2, 6, Roche, 2, 6, samsung, 2, 6; O. Distler: 4P-Pharma, 2, 5, 6, AbbVie, 2, 5, 6, Acceleron, 2, 5, 6, Alcimed, 2, 5, 6, Altavant Sciences, 2, 5, 6, Amgen, 2, 5, 6, AnaMar, 2, 5, 6, Arxx, 2, 5, 6, AstraZeneca, 2, 5, 6, Bayer, 2, 5, 6, Blade Therapeutics, 2, 5, 6, Boehringer Ingelheim, 2, 5, 6, Citus AG, 12, Co-Founder, Corbus Pharmaceuticals, 2, 5, 6, CSL Behring, 2, 5, 6, Galapagos, 2, 5, 6, Galderma, 2, 5, 6, Glenmark, 2, 5, 6, Gossamer, 2, 5, 6, Horizon Therapeutics, 2, 5, 6, Janssen, 2, 5, 6, Kymera, 2, 5, 6, Lupin, 2, 5, 6, Medscape, 2, 5, 6, Miltenyi Biotec, 2, 5, 6, Mitsubishi Tanabe, 2, 5, 6, MSD, 2, 5, 6, Novartis, 2, 5, 6, Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143), 10, Prometheus Biosciences, 2, 5, 6, Redx Pharma, 2, 5, 6, Roivant, 2, 5, 6, Topadur, 2, 5, 6; C. Bruni: AbbVie/Abbott, 5, Boehringer-Ingelheim, 2, 12, Travel Support, Eli Lilly, 6.



