Abstract Session
Systemic lupus erythematosus (SLE)
Session: Abstracts: SLE – Etiology & Pathogenesis (1597–1602)
1598: Single-cell Spatial Proteomics Identifies Intraglomerular Myeloid Cells in Membranous Lupus Nephritis
Monday, November 13, 2023
2:15 PM - 2:25 PM PT
Location: Room 6F/6C
- AF
Andrea Fava, MD
Johns Hopkins University
Baltimore, MD, United StatesDisclosure(s): Annexon Biosciences: Consultant (Ongoing); Sanofi: Advisor or Review Panel Member (Terminated, April 30, 2022)
Presenting Author(s)
Chen-Yu Lee1, Caleb Marlin2, Xiaoping Yang1, Tayte Stephens2, Alessandra Ida Celia3, Jeff Hodgin4, Peter Izmirly5, H Michael Belmont6, Jill Buyon7, Chaim Putterman8, Judith James2, the Accelerating Medicines Partnership in RA/SLE9, Michelle Petri10, Joel Guthridge2, Avi Rosenberg11 and Andrea Fava1, 1Johns Hopkins University, Baltimore, MD, 2Oklahoma Medical Research Foundation, Oklahoma City, OK, 3John Hopkins University of Medicine, Rome, Italy, 4University of Michigan, Ann Arbor, MI, 5New York University School of Medicine, New York, NY, 6NYU School of Medicine, New York, NY, 7NYU Grossman School of Medicine, New York, NY, 8Albert Einstein College of Medicine, Bronx, NY, 9Multiple, Multiple, 10Department of Medicine, Division of Rheumatology, Johns Hopkins University School of Medicine, Timonium, MD, 11Department of Pathology, The Johns Hopkins University School of Medicine, Baltimore, MD
Background/Purpose: Lupus nephritis (LN) leads to end-stage kidney disease (ESKD) in more than 20% of patients despite optimal treatment. Up to 30% of LN patients have membranous LN which is characterized by subepithelial immune deposits without immune infiltration in the glomeruli. Despite its association with ESKD, membranous LN is considered a milder type of LN with no consensus on the optimal use of immunosuppression. To develop mechanistic hypotheses of disease, we analyzed kidney samples from patients with LN using a whole slide spatially resolved proteomic approach as part of the Accelerating Medicines Partnership in RA/SLE. We report here the initial analysis.
Methods: We developed a serial immunohistochemistry (sIHC) staining workflow to stain for 18 antibodies, DNA, and PAS to be visualized on a single section via a cycle of staining, imaging, and destaining. This included incubation of FFPE slides with primary antibody, secondary HRP reagents, AEC-Red Chromogen, and Hematoxylin. Image processing was performed using HALO (Indica Labs) and included deconvolution of single-color channels, registration, fusion, cell segmentation, and automated tissue classification (glomeruli, tubulointerstitium, connective tissue, vasculature, background, and biopsy edges). To minimize batch effect, the analytical pipeline included within sample CLR-normalization and scaling, followed by harmonization (Harmony).
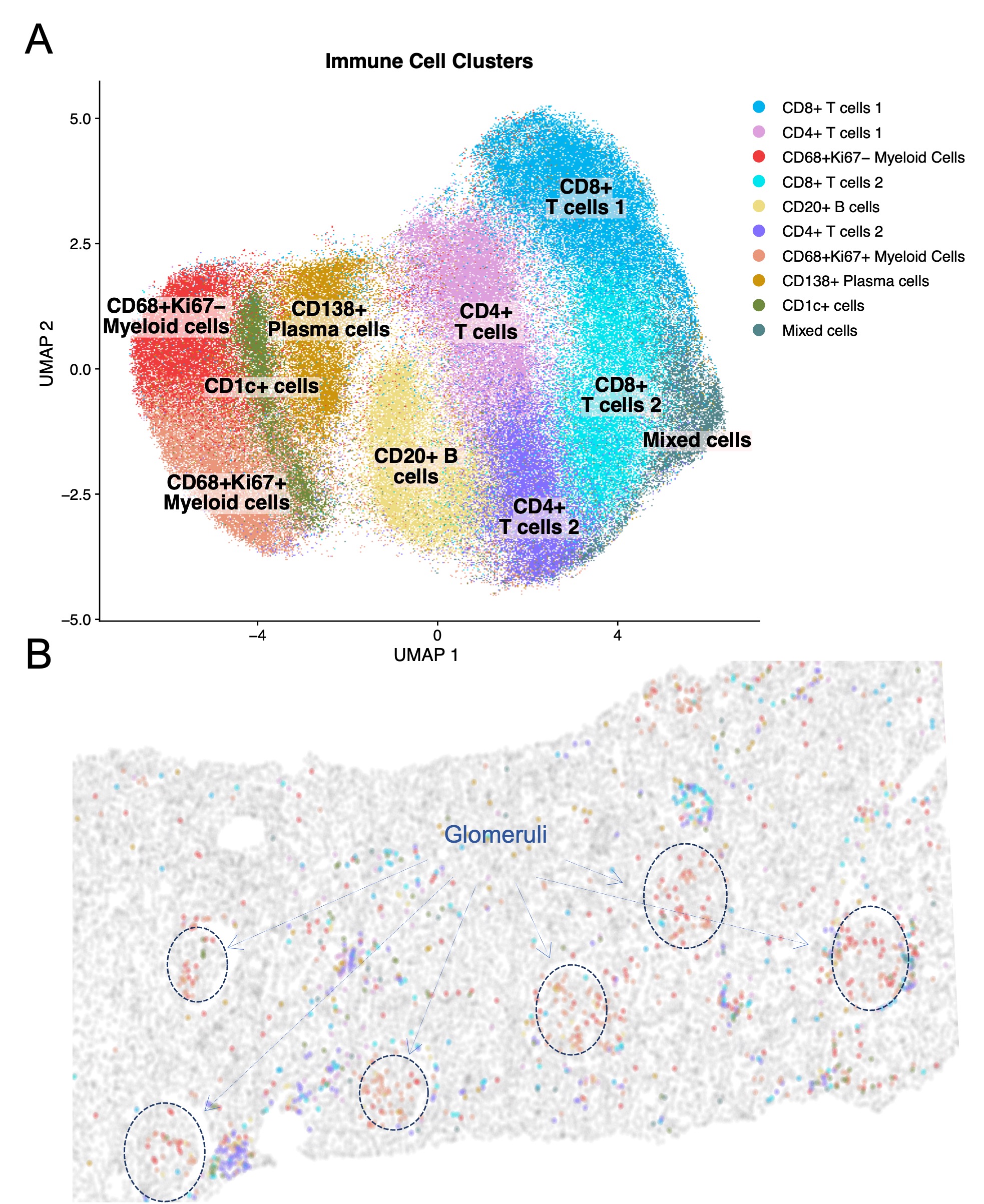
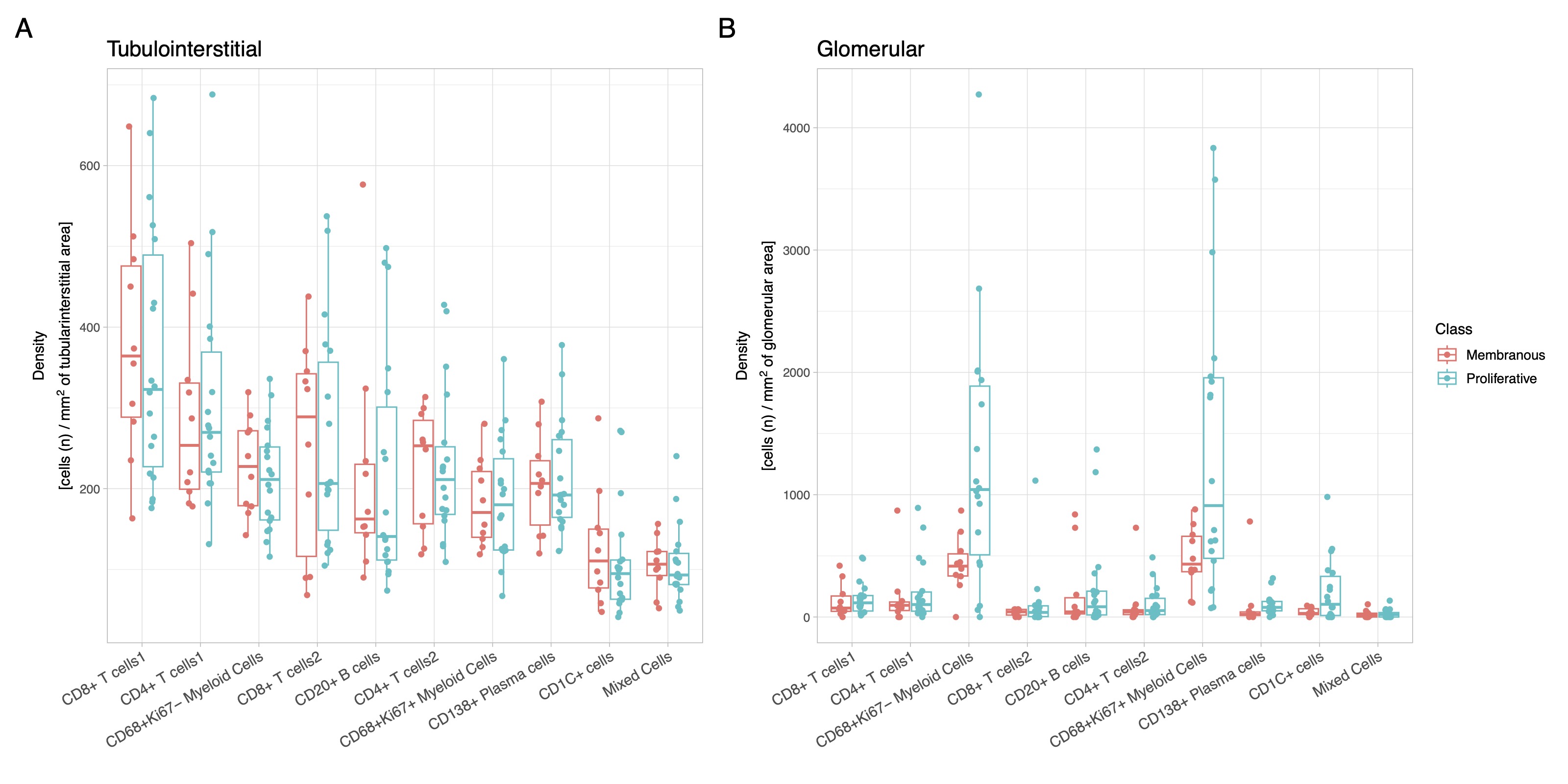
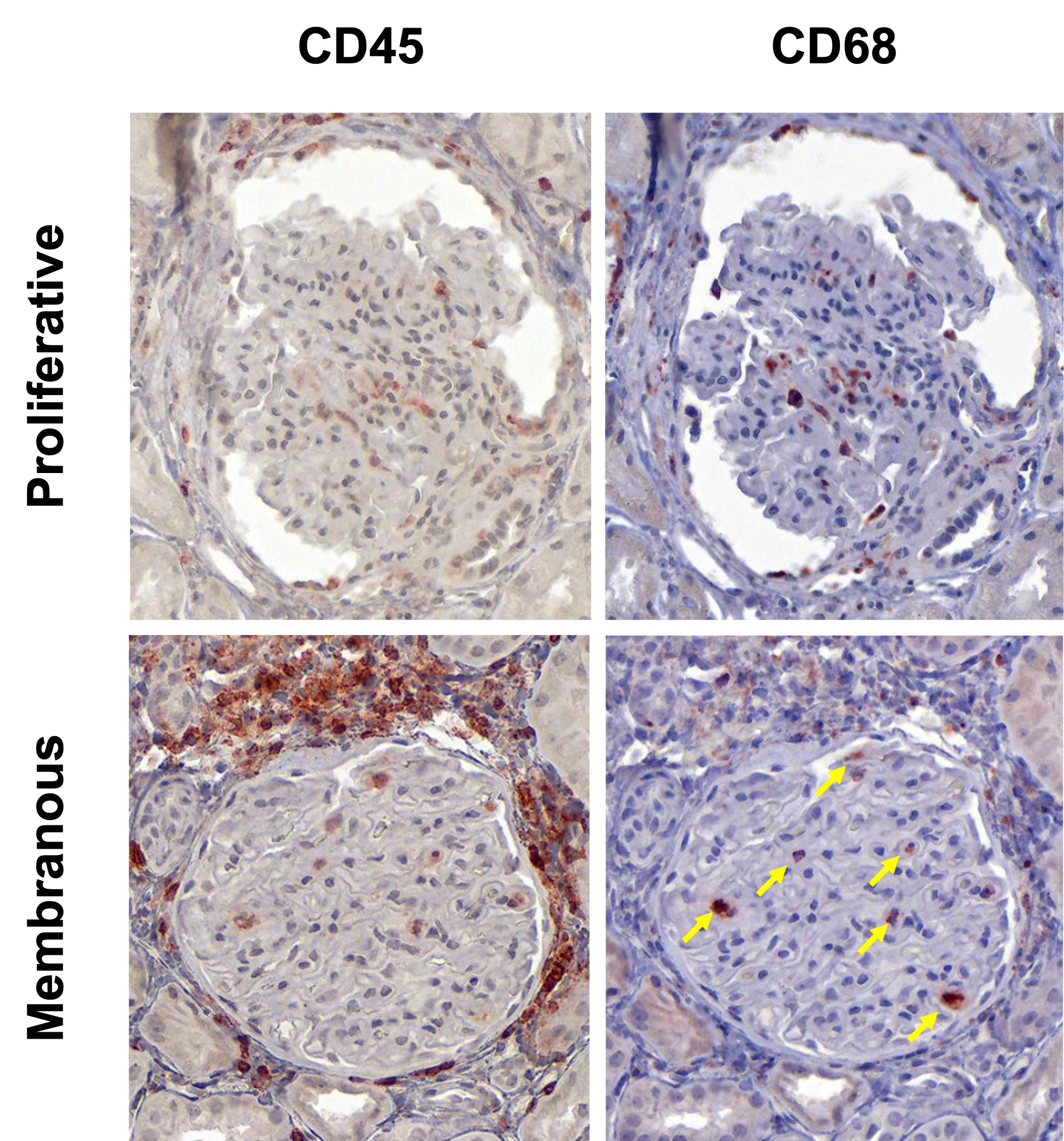
Results: In this initial analysis, we included 29 clinically indicated kidney biopsies classified as LN (13 pure proliferative, 10 pure membranous, 5 mixed, and 1 ISN class II). Patients were 79% female, 34% White, 31% Black, 10% Asian, and 24% identified with Other race/ethnicity. We detected 182,783 CD45+ cells out of 1,913,845 cell objects. Our analysis identified 10 immune cell clusters at low resolution (Figure 1A). Figure 1B displays the tissue distribution of each cell subset. B and T lymphocytes dominated the tubulointerstitium. The CD68+ myeloid subsets were the predominant cell type in the glomeruli (Figure 2). More than half of CD68+ cells expressed Ki67 indicating active proliferation. Surprisingly, we identified intraglomerular CD68+ cells (including endocapillary) also in patients with pure membranous LN, but at a lower tissue density than proliferative LN (Figure 2). Figure 3 demonstrates intraglomerular CD68+ cells in a biopsy classified as pure membranous LN by two experienced renal pathologists.
Conclusion: sIHC can be successfully employed to perform multiplexed whole slide analysis harnessing both the subcellular resolution (brightfield) and the reliability of IHC. Our analysis revealed intraglomerular CD68+ myeloid cells in pure membranous LN. By traditional clinical pathology, intraglomerular / endocapillary immune cells characterize proliferative LN and are not consistent with pure membranous LN. These findings implicate macrophages/monocytes in the glomerular disease in membranous LN with therapeutic implications. The analysis of 90 additional biopsies and a myeloid-focused panel is underway to validate and extend these findings.
C. Lee: None; C. Marlin: None; X. Yang: None; T. Stephens: None; A. Celia: None; J. Hodgin: AstraZeneca, 5, 6, Eli Lilly, 5, Gilead, 5, Janssen, 5, Moderna, 5, Novo Nordisk, 5, Regeneron, 5; P. Izmirly: None; H. Belmont: Alexion, 6, Aurinia, 6; J. Buyon: Bristol-Myers Squibb(BMS), 2, GlaxoSmithKlein(GSK), 2, Related Sciences, 1; C. Putterman: Equillium, 2, KidneyCure, 1, Progentec, 2; J. James: Bristol-Myers Squibb(BMS), 5, GlaxoSmithKlein(GSK), 2, Novartis, 2, Progentec Biosciences, 5; t. Accelerating Medicines Partnership in RA/SLE: None; M. Petri: Alexion, 1, Amgen, 1, AnaptysBio, 1, Annexon Bio, 1, Argenx, 1, Arhros-Focus Med/Ed, 6, AstraZeneca, 1, 5, Aurinia, 1, 5, 6, Axdev, 1, Biogen, 1, Boxer Capital, 2, Cabaletto Bio, 2, Caribou Biosciences, 2, CVS Health, 1, Eli Lilly, 1, 5, Emergent Biosolutions, 1, Exagen, 5, Exo Therapeutics, 2, Gilead Biosciences, 2, GlaxoSmithKlein(GSK), 1, 5, 6, Horizon Therapeutics, 2, Idorsia Pharmaceuticals, 2, IQVIA, 1, Janssen, 1, 5, Kira Pharmaceuticals, 2, MedShr, 6, Merck/EMD Serono, 1, Momenta Pharmaceuticals, 2, Nexstone Immunology, 2, Nimbus Lakshmi, 2, Proviant, 2, Sanofi, 2, Sinomab Biosciences, 2, Thermofisher, 5, UCB, 2; J. Guthridge: None; A. Rosenberg: None; A. Fava: Annexon Biosciences, 2, Sanofi, 1.
Background/Purpose: Lupus nephritis (LN) leads to end-stage kidney disease (ESKD) in more than 20% of patients despite optimal treatment. Up to 30% of LN patients have membranous LN which is characterized by subepithelial immune deposits without immune infiltration in the glomeruli. Despite its association with ESKD, membranous LN is considered a milder type of LN with no consensus on the optimal use of immunosuppression. To develop mechanistic hypotheses of disease, we analyzed kidney samples from patients with LN using a whole slide spatially resolved proteomic approach as part of the Accelerating Medicines Partnership in RA/SLE. We report here the initial analysis.
Methods: We developed a serial immunohistochemistry (sIHC) staining workflow to stain for 18 antibodies, DNA, and PAS to be visualized on a single section via a cycle of staining, imaging, and destaining. This included incubation of FFPE slides with primary antibody, secondary HRP reagents, AEC-Red Chromogen, and Hematoxylin. Image processing was performed using HALO (Indica Labs) and included deconvolution of single-color channels, registration, fusion, cell segmentation, and automated tissue classification (glomeruli, tubulointerstitium, connective tissue, vasculature, background, and biopsy edges). To minimize batch effect, the analytical pipeline included within sample CLR-normalization and scaling, followed by harmonization (Harmony).
Results: In this initial analysis, we included 29 clinically indicated kidney biopsies classified as LN (13 pure proliferative, 10 pure membranous, 5 mixed, and 1 ISN class II). Patients were 79% female, 34% White, 31% Black, 10% Asian, and 24% identified with Other race/ethnicity. We detected 182,783 CD45+ cells out of 1,913,845 cell objects. Our analysis identified 10 immune cell clusters at low resolution (Figure 1A). Figure 1B displays the tissue distribution of each cell subset. B and T lymphocytes dominated the tubulointerstitium. The CD68+ myeloid subsets were the predominant cell type in the glomeruli (Figure 2). More than half of CD68+ cells expressed Ki67 indicating active proliferation. Surprisingly, we identified intraglomerular CD68+ cells (including endocapillary) also in patients with pure membranous LN, but at a lower tissue density than proliferative LN (Figure 2). Figure 3 demonstrates intraglomerular CD68+ cells in a biopsy classified as pure membranous LN by two experienced renal pathologists.
Conclusion: sIHC can be successfully employed to perform multiplexed whole slide analysis harnessing both the subcellular resolution (brightfield) and the reliability of IHC. Our analysis revealed intraglomerular CD68+ myeloid cells in pure membranous LN. By traditional clinical pathology, intraglomerular / endocapillary immune cells characterize proliferative LN and are not consistent with pure membranous LN. These findings implicate macrophages/monocytes in the glomerular disease in membranous LN with therapeutic implications. The analysis of 90 additional biopsies and a myeloid-focused panel is underway to validate and extend these findings.

Figure 1. Phenotype and spatial distribution of intrarenal immune cells. (A) UMAP of CD45+ cells (n=182,783, 29 patients) indicating the low-resolution cluster annotation. (B) Digital reproduction of a representative biopsy displaying the distribution of the cells clusters (colors matching panel A). The biopsy in panel B was classified as ISN class III, NIH Activity Index 6, NIH Chronicity Index 3.

Figure 2. Myeloid cells dominate intraglomerular inflammation, including membranous LN. Box plots displaying the density (cells / area) of the immune cell clusters in the tubulointerstitium (A) and glomeruli (B) according to ISN class group. Both CD68+ myeloid cell clusters showed a statistically significant higher intraglomerular density compared to all other clusters in both proliferative and membranous LN (p<0.05, Wilcoxon test). Proliferative LN includes ISN class III and IV +/- V (n=18); membranous LN indicate pure ISN class V (n=10).

Figure 3. Intraglomerular CD68+ monocyte/macrophages in proliferative and membranous LN. Immunohistochemistry images displaying the expression of CD45 and CD68 in a representative glomerulus from a patient with proliferative LN (upper panels) and a patient with pure membranous LN (lower panels). The yellow arrows indicate intraglomerular CD68+ cells in membranous LN.
C. Lee: None; C. Marlin: None; X. Yang: None; T. Stephens: None; A. Celia: None; J. Hodgin: AstraZeneca, 5, 6, Eli Lilly, 5, Gilead, 5, Janssen, 5, Moderna, 5, Novo Nordisk, 5, Regeneron, 5; P. Izmirly: None; H. Belmont: Alexion, 6, Aurinia, 6; J. Buyon: Bristol-Myers Squibb(BMS), 2, GlaxoSmithKlein(GSK), 2, Related Sciences, 1; C. Putterman: Equillium, 2, KidneyCure, 1, Progentec, 2; J. James: Bristol-Myers Squibb(BMS), 5, GlaxoSmithKlein(GSK), 2, Novartis, 2, Progentec Biosciences, 5; t. Accelerating Medicines Partnership in RA/SLE: None; M. Petri: Alexion, 1, Amgen, 1, AnaptysBio, 1, Annexon Bio, 1, Argenx, 1, Arhros-Focus Med/Ed, 6, AstraZeneca, 1, 5, Aurinia, 1, 5, 6, Axdev, 1, Biogen, 1, Boxer Capital, 2, Cabaletto Bio, 2, Caribou Biosciences, 2, CVS Health, 1, Eli Lilly, 1, 5, Emergent Biosolutions, 1, Exagen, 5, Exo Therapeutics, 2, Gilead Biosciences, 2, GlaxoSmithKlein(GSK), 1, 5, 6, Horizon Therapeutics, 2, Idorsia Pharmaceuticals, 2, IQVIA, 1, Janssen, 1, 5, Kira Pharmaceuticals, 2, MedShr, 6, Merck/EMD Serono, 1, Momenta Pharmaceuticals, 2, Nexstone Immunology, 2, Nimbus Lakshmi, 2, Proviant, 2, Sanofi, 2, Sinomab Biosciences, 2, Thermofisher, 5, UCB, 2; J. Guthridge: None; A. Rosenberg: None; A. Fava: Annexon Biosciences, 2, Sanofi, 1.



