Abstract Session
Sjögren’s syndrome
Session: Abstracts: Sjögren’s Syndrome – Basic & Clinical Science (1633–1638)
1634: Iscalimab (CFZ533) in Patients with Sjögren’s Disease: Week 24 Efficacy and Safety Results of a Randomized, Placebo-controlled, Phase 2b Dose-ranging Study
Monday, November 13, 2023
2:15 PM - 2:25 PM PT
Location: Room 5A-B
- BF
Benjamin A. Fisher, MD, MBBS
University of Birmingham
Birmingham, United KingdomDisclosure information not submitted.
Presenting Author(s)
Benjamin A Fisher1, Xavier Mariette2, Athena S Papas3, Thomas Grader-Beck4, Hendrika Bootsma5, Wan-Fai Ng6, Paul Van Daele7, Stephanie Finzel8, Sergio Elgueta9, Josef Hermann10, Sara McCoy11, Arthur Bookman12, Monika Sopala13, Wen-Lin Luo14, Cornelia Scheurer13 and Wolfgang Hueber13, 1University of Birmingham, Birmingham, United Kingdom, 2Université Paris-Saclay, Le Kremlin-Bicêtre, France, 3Division of Oral Medicine, Tufts School of Dental Medicine, Boston, MA, 4Division of Rheumatology, Johns Hopkins School of Medicine, Baltimore, MD, 5Department of Rheumatology, University Medical Centre Groningen, Groningen, Netherlands, 6NIHR Newcastle Clinical Research Facility, Newcastle upon Tyne Hospitals NHS Foundation Trust, Newcastle upon Tyne, United Kingdom, 7Department of internal medicine, Erasmus MC, Rotterdam, Netherlands, 8Department of Rheumatology and Clinical Immunology, University Medical Center Freiburg, University of Freiburg, Freiburg, Germany, 9Clinical Research Chile SpA, Biomedical Research Centre, Valdivia, Chile, 10Division of Rheumatology and Immunology, Department of Internal Medicine, Medical University Graz, Graz, Austria, 11University of Wisconsin School of Medicine and Public Health, Middleton, WI, 12Toronto Western Hospital, Toronto, ON, Canada, 13Novartis Pharma AG, Basel, Switzerland, 14Novartis Pharmaceuticals Corporation, East Hanover, NJ
Background/Purpose: Sjögren's disease (SjD) is an autoimmune disease affecting secretory glands and other organs with no approved systemic treatments. Iscalimab (CFZ533) is a mAb directed against CD40, a novel way to target underlying disease pathophysiology. TWINSS is an ongoing phase 2b dose-ranging trial assessing safety and efficacy of multiple CFZ533 doses in two distinct patient (pt) populations with SjD of moderate/severe systemic disease (Cohort 1/C1) and of low systemic disease and high symptom burden (Cohort 2/C2). We report Week 24 primary results from both cohorts (N=273).
Methods: In C1, 173 pts were randomized (1:1:1:1) to CFZ533 150 mg or 300 mg or 600 mg s.c. q2w or placebo (PBO). In C2, 100 pts were assigned (1:1) to CFZ533 600 mg or PBO. Key inclusion criteria were: fulfilled ACR/EULAR 2016 classification criteria; stimulated salivary flow rates of ≥0.1 mL/min; anti-Ro/SSA positivity; C1: EULAR Sjögren's Syndrome (SS) Disease Activity Index (ESSDAI) ≥5 (on 8 selected domains), EULAR SS Patient Reported Index (ESSPRI) ≥5; and C2: ESSDAI < 5 with presence of activity in biologic domain ( >0), ESSPRI (fatigue or dryness ≥5, impact of dry eye on everyday life [IDEEL] ≥30). Statistical methods included Multiple Comparison Procedure – Modelling methodology to assess the dose response of the ESSDAI change from baseline (C1) and mixed model for repeated measures to estimate the adjusted mean change from baseline and difference vs PBO (C1 and C2). Similar methods were applied to all other endpoints including ESSPRI.
Results: Baseline characteristics were balanced across treatments except in C1 where more pts on iscalimab 300 mg had severe disease activity (ESSDAI >13; 44%) and used HCQ (70%), compared to the other treatment arms (ESSDAI >13, 19-28% and HCQ, 47-64%, respectively).
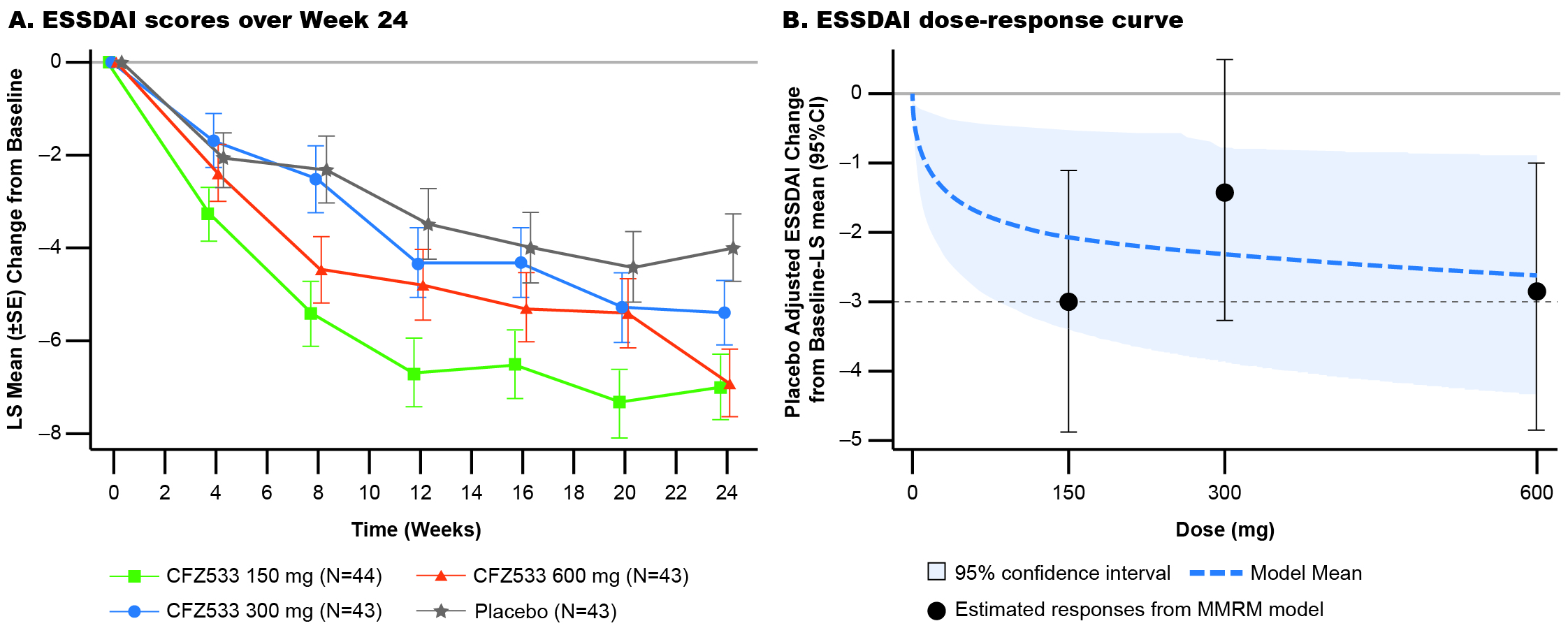
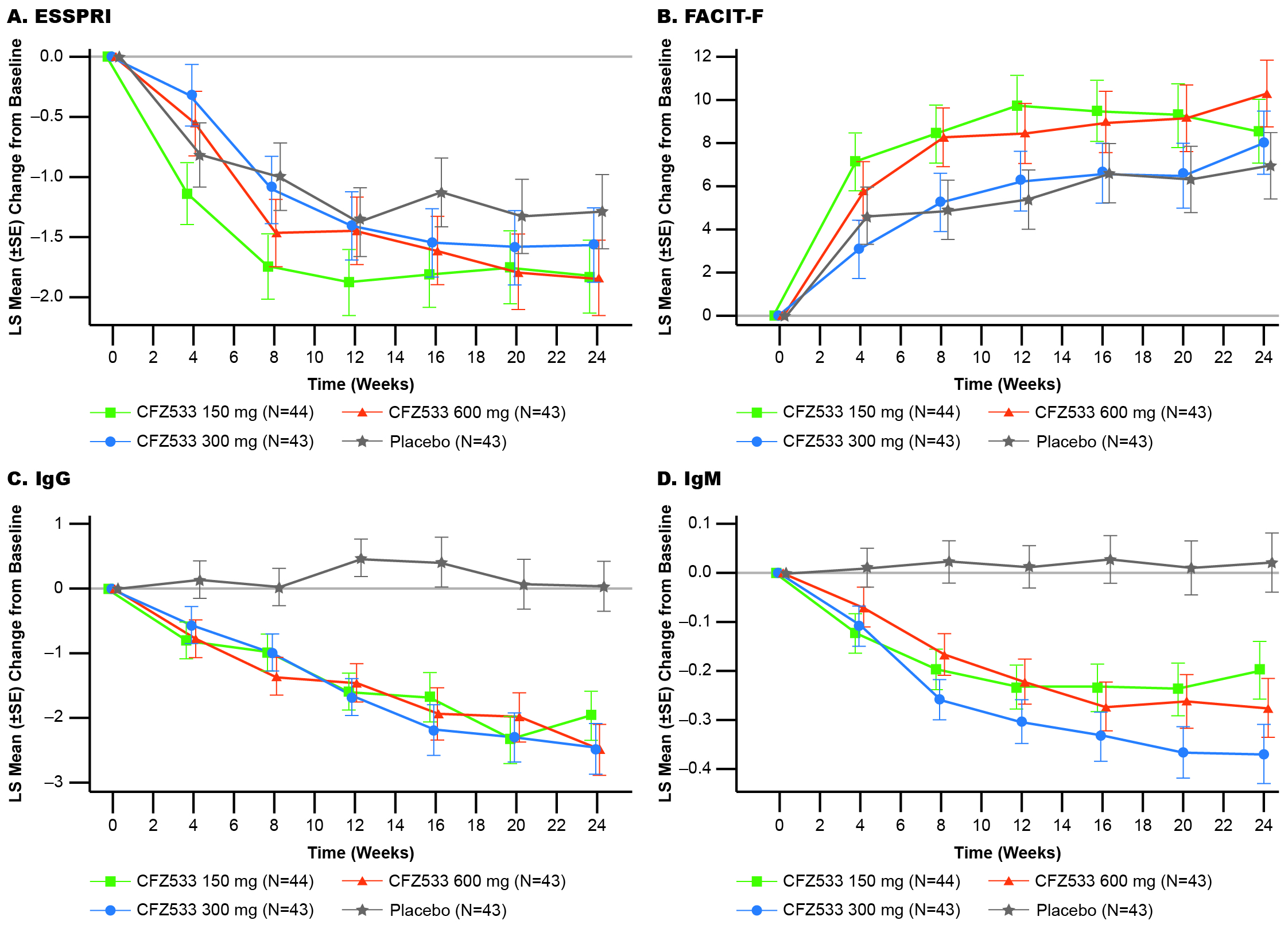
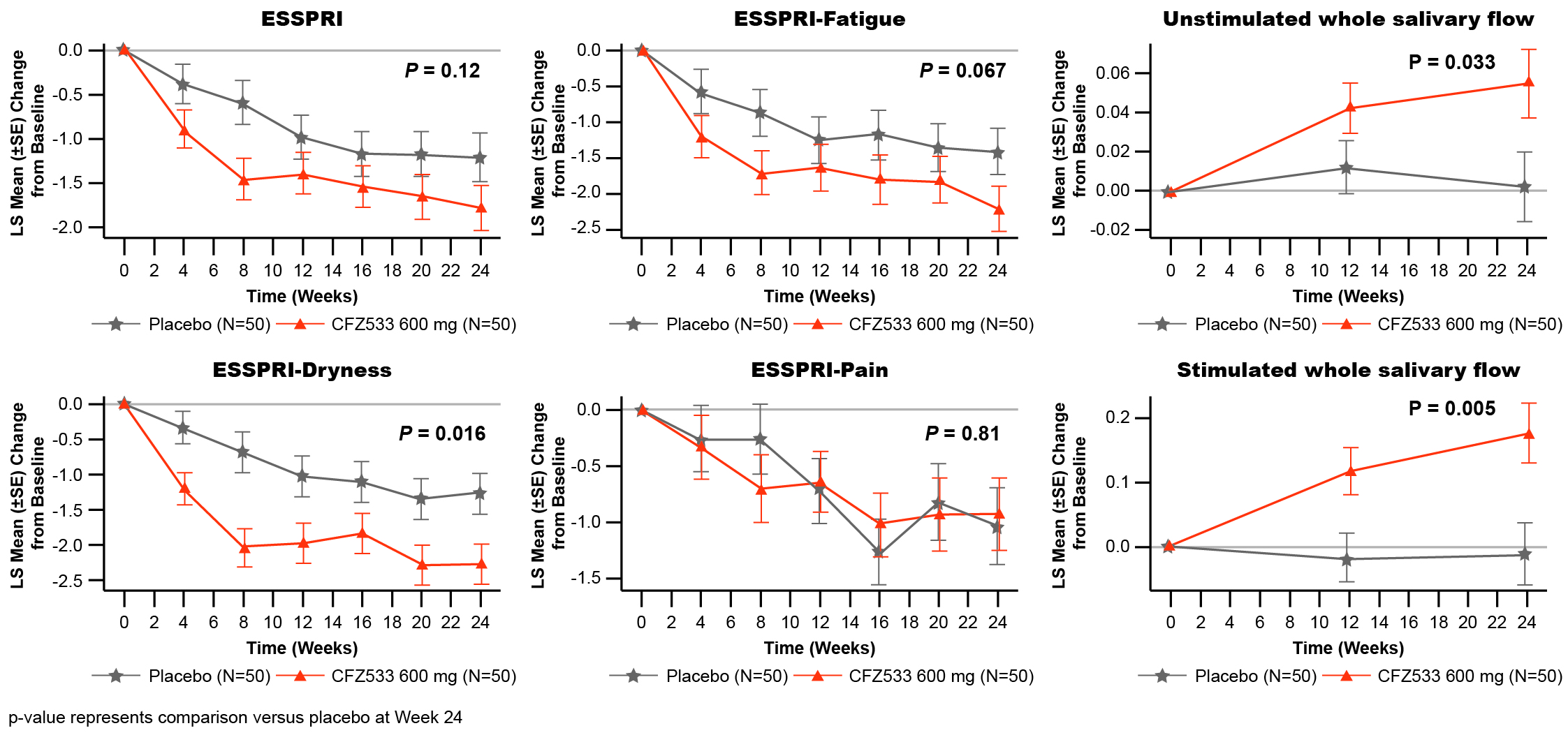
In C1, ESSDAI change from baseline at Week 24 showed statistically significant improvements compared to PBO for CFZ533 150 and 600 mg (least square mean difference (Δ) = –3.0 and –2.9; p < 0.005), and 300-mg dose showed a similar trend (Δ= –1.4; p = 0.16) (Fig 1). Primary objective of significant dose response of ESSDAI change from baseline at Week 24 (p = 0.004) with a log-linear dose-response was met. Predicted dose-response curve suggested that the drug effect plateaued at ≥150 mg. ESSPRI, FACIT-F (Fig 2), tear flow, and salivary flow (SF) rates showed numerical trends for improvement with active treatment. By Week 24, IgG/IgM as objective pharmacodynamic markers showed significant and similar plateaus of reduction for all doses of CFZ533 compared to PBO. In C2, the primary objective of ESSPRI change from baseline showed a strong trend (Δ = –0.57; p = 0.12) toward improvement with dryness (Δ = –1.0; p = 0.016) and fatigue (Δ = –0.8; p = 0.067) as the response drivers (Fig 3). For exploratory outcomes, statistically significant increases in SF were seen.
At Week 24, SAE rates in C1 (PBO, CFZ533 150 mg, 300 mg, 600 mg) were 2.3%, 2.3%, 7.1%, and 9.1%, respectively, in C2 (PBO or CFZ533 600mg) 4% each. Infection rates in C1 were 39.5%, 45.5%, 26.2%, and 43.2%; and in C2 42.0% and 56.0%, respectively.
Conclusion: Iscalimab showed a clinically important improvement over placebo in 2 distinct populations of SjD patients and was well tolerated without obvious safety signals.
B. A Fisher: Bristol-Myers Squibb(BMS), 2, Celgene, 5, Galapagos, 2, 5, Janssen, 2, 5, Novartis, 2, Roche, 2, Sanofi, 2, Servier, 2, 5, UCB, 2; X. Mariette: AstraZeneca, 2, 6, BMS, 2, 6, Galapagos, 2, 6, GSK, 2, 6, Novartis, 2, 6, Pfizer, 2, 6; A. S Papas: Novartis, 1, 5; T. Grader-Beck: IQVIA, 2, Novartis, 2; H. Bootsma: None; W. Ng: AbbVie, 12, Research Collaboration, Argenx, 2, GlaxoSmithKlein(GSK), 12, Research Collaboration, Janssen, 2, Novartis, 2, Resolve Therapeutics, 2, Sanofi, 2; P. Van Daele: None; S. Finzel: AbbVie, 6, AstraZeneca, 2, 6, Chugai, 6, Galapagos, 2, 6, Novartis, 2, Novo Nordisk, 2, UCB, 6; S. Elgueta: None; J. Hermann: None; S. McCoy: Bristol-Myers Squibb(BMS), 2, Horizon, 2, Kiniksa, 2, Novartis, 2, Otsuka/Visterra, 2, Target RWE, 2; A. Bookman: GlaxoSmithKline(GSK), 12, Regional Principal Investigator, Novartis, 12, Regional Principal Investigator, VIELA BIO, 12, Regional Principal Investigator; M. Sopala: Novartis, 3; W. Luo: Novartis, 3; C. Scheurer: Novartis, 3; W. Hueber: Novartis, 3, 11.
Background/Purpose: Sjögren's disease (SjD) is an autoimmune disease affecting secretory glands and other organs with no approved systemic treatments. Iscalimab (CFZ533) is a mAb directed against CD40, a novel way to target underlying disease pathophysiology. TWINSS is an ongoing phase 2b dose-ranging trial assessing safety and efficacy of multiple CFZ533 doses in two distinct patient (pt) populations with SjD of moderate/severe systemic disease (Cohort 1/C1) and of low systemic disease and high symptom burden (Cohort 2/C2). We report Week 24 primary results from both cohorts (N=273).
Methods: In C1, 173 pts were randomized (1:1:1:1) to CFZ533 150 mg or 300 mg or 600 mg s.c. q2w or placebo (PBO). In C2, 100 pts were assigned (1:1) to CFZ533 600 mg or PBO. Key inclusion criteria were: fulfilled ACR/EULAR 2016 classification criteria; stimulated salivary flow rates of ≥0.1 mL/min; anti-Ro/SSA positivity; C1: EULAR Sjögren's Syndrome (SS) Disease Activity Index (ESSDAI) ≥5 (on 8 selected domains), EULAR SS Patient Reported Index (ESSPRI) ≥5; and C2: ESSDAI < 5 with presence of activity in biologic domain ( >0), ESSPRI (fatigue or dryness ≥5, impact of dry eye on everyday life [IDEEL] ≥30). Statistical methods included Multiple Comparison Procedure – Modelling methodology to assess the dose response of the ESSDAI change from baseline (C1) and mixed model for repeated measures to estimate the adjusted mean change from baseline and difference vs PBO (C1 and C2). Similar methods were applied to all other endpoints including ESSPRI.
Results: Baseline characteristics were balanced across treatments except in C1 where more pts on iscalimab 300 mg had severe disease activity (ESSDAI >13; 44%) and used HCQ (70%), compared to the other treatment arms (ESSDAI >13, 19-28% and HCQ, 47-64%, respectively).
In C1, ESSDAI change from baseline at Week 24 showed statistically significant improvements compared to PBO for CFZ533 150 and 600 mg (least square mean difference (Δ) = –3.0 and –2.9; p < 0.005), and 300-mg dose showed a similar trend (Δ= –1.4; p = 0.16) (Fig 1). Primary objective of significant dose response of ESSDAI change from baseline at Week 24 (p = 0.004) with a log-linear dose-response was met. Predicted dose-response curve suggested that the drug effect plateaued at ≥150 mg. ESSPRI, FACIT-F (Fig 2), tear flow, and salivary flow (SF) rates showed numerical trends for improvement with active treatment. By Week 24, IgG/IgM as objective pharmacodynamic markers showed significant and similar plateaus of reduction for all doses of CFZ533 compared to PBO. In C2, the primary objective of ESSPRI change from baseline showed a strong trend (Δ = –0.57; p = 0.12) toward improvement with dryness (Δ = –1.0; p = 0.016) and fatigue (Δ = –0.8; p = 0.067) as the response drivers (Fig 3). For exploratory outcomes, statistically significant increases in SF were seen.
At Week 24, SAE rates in C1 (PBO, CFZ533 150 mg, 300 mg, 600 mg) were 2.3%, 2.3%, 7.1%, and 9.1%, respectively, in C2 (PBO or CFZ533 600mg) 4% each. Infection rates in C1 were 39.5%, 45.5%, 26.2%, and 43.2%; and in C2 42.0% and 56.0%, respectively.
Conclusion: Iscalimab showed a clinically important improvement over placebo in 2 distinct populations of SjD patients and was well tolerated without obvious safety signals.

Figure 1: Least-square mean of change from baseline in ESSDAI scores over time up to Week 24 and estimated dose-response curve in Cohort 1

Figure 2: Least-square mean of change from baseline in patient-reported outcomes (secondary outcome measures) and pharmacodynamic markers up to Week 24 in Cohort 1

Figure 3. Least-square mean of change from baseline in ESSPRI and its subdomains and whole salivary flow up to Week 24 in Cohort 2
B. A Fisher: Bristol-Myers Squibb(BMS), 2, Celgene, 5, Galapagos, 2, 5, Janssen, 2, 5, Novartis, 2, Roche, 2, Sanofi, 2, Servier, 2, 5, UCB, 2; X. Mariette: AstraZeneca, 2, 6, BMS, 2, 6, Galapagos, 2, 6, GSK, 2, 6, Novartis, 2, 6, Pfizer, 2, 6; A. S Papas: Novartis, 1, 5; T. Grader-Beck: IQVIA, 2, Novartis, 2; H. Bootsma: None; W. Ng: AbbVie, 12, Research Collaboration, Argenx, 2, GlaxoSmithKlein(GSK), 12, Research Collaboration, Janssen, 2, Novartis, 2, Resolve Therapeutics, 2, Sanofi, 2; P. Van Daele: None; S. Finzel: AbbVie, 6, AstraZeneca, 2, 6, Chugai, 6, Galapagos, 2, 6, Novartis, 2, Novo Nordisk, 2, UCB, 6; S. Elgueta: None; J. Hermann: None; S. McCoy: Bristol-Myers Squibb(BMS), 2, Horizon, 2, Kiniksa, 2, Novartis, 2, Otsuka/Visterra, 2, Target RWE, 2; A. Bookman: GlaxoSmithKline(GSK), 12, Regional Principal Investigator, Novartis, 12, Regional Principal Investigator, VIELA BIO, 12, Regional Principal Investigator; M. Sopala: Novartis, 3; W. Luo: Novartis, 3; C. Scheurer: Novartis, 3; W. Hueber: Novartis, 3, 11.



