Abstract Session
Pediatric autoimmune diseases: Kawasaki disease, juvenile dermatomyositis and juvenile localized scleroderma
Session: Abstracts: Pediatric Rheumatology – Clinical II: Connective Tissue Disease (1621–1626)
1622: Myositis-associated Autoantibodies in Juvenile Myositis Are Associated with Severe Disease Features and Mortality
Monday, November 13, 2023
2:15 PM - 2:25 PM PT
Location: Room 7A-B
- MS
Matthew Sherman, MD, MHS
DAIT/NIAID/NIH
Washington, DC, United StatesDisclosure information not submitted.
Presenting Author(s)
Matthew Sherman1, Payam Noroozi Farhad2, Katherine Pak3, Iago Pinal-Fernandez3, Kakali Sarkar2, Megan Neely4, Ira Targoff5, Frederick Miller6, Andrew Mammen7 and Lisa Rider8, 1NIAMS/NIH, Washington, DC, 2Environmental Autoimmunity Group, National Institute of Environmental Health Sciences, National Institutes of Health, Bethesda, MD, 3National Institutes of Health, Bethesda, MD, 4Department of Biostatistics and Bioinformatics, Duke University, Durham, NC, 5Veterans Affairs Medical Center, University of Oklahoma Health Sciences Center, and Oklahoma Medical Research Foundation, Oklahoma City, OK, 6NIH, NIEHS, Chapel Hill, NC, 7NIH, Bethesda, MD, 8NIEHS, NIH, Bethesda, MD
Background/Purpose: Myositis-associated autoantibodies (MAAs), such as anti-Ro52 autoantibodies (Abs), have been found to be associated with interstitial lung disease (ILD) and worse prognosis in the idiopathic inflammatory myopathies. MAAs remain largely uncharacterized in juvenile-onset myositis. Moreover, MAAs often co-exist and it is unknown whether the number of MAA specificities may be associated with increased disease severity.
Methods: Patients with juvenile myositis enrolled in cross-sectional NIH myositis natural history studies who underwent testing for myositis Abs were included. Demographics, clinical manifestations, treatments, and outcomes of those with and without MAAs were compared using Chi-squared, Fisher's exact test, or Wilcoxon rank-sum test. Multivariable logistic regression with adjustment for year of diagnosis and myositis autoantibodies was performed for statistically significant variables from the univariable analyses. Multivariable logistic regression was also used to determine whether the number of MAA specificities is predictive of severe disease features. A two-sided p< 0.05 was considered significant.
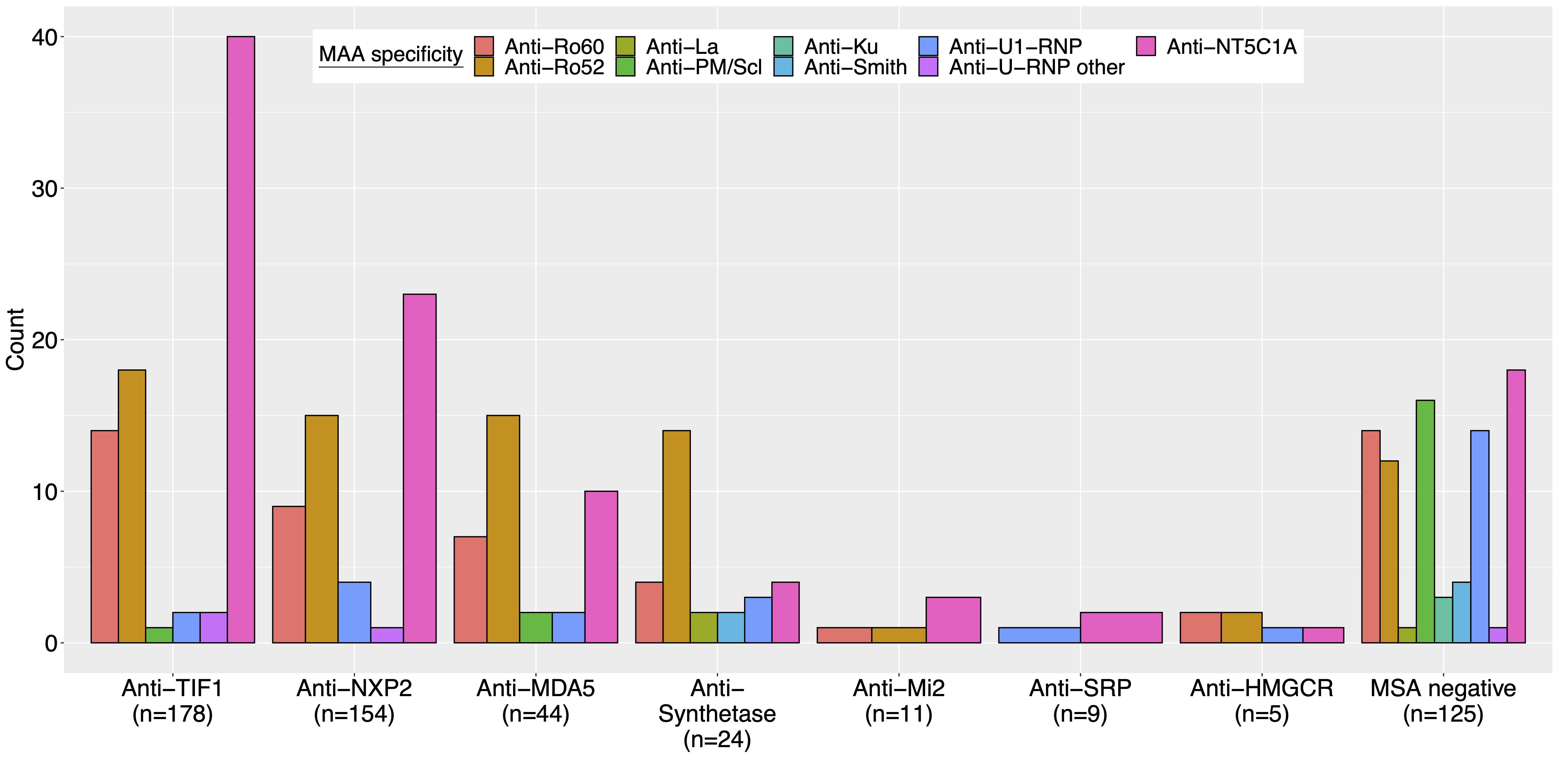
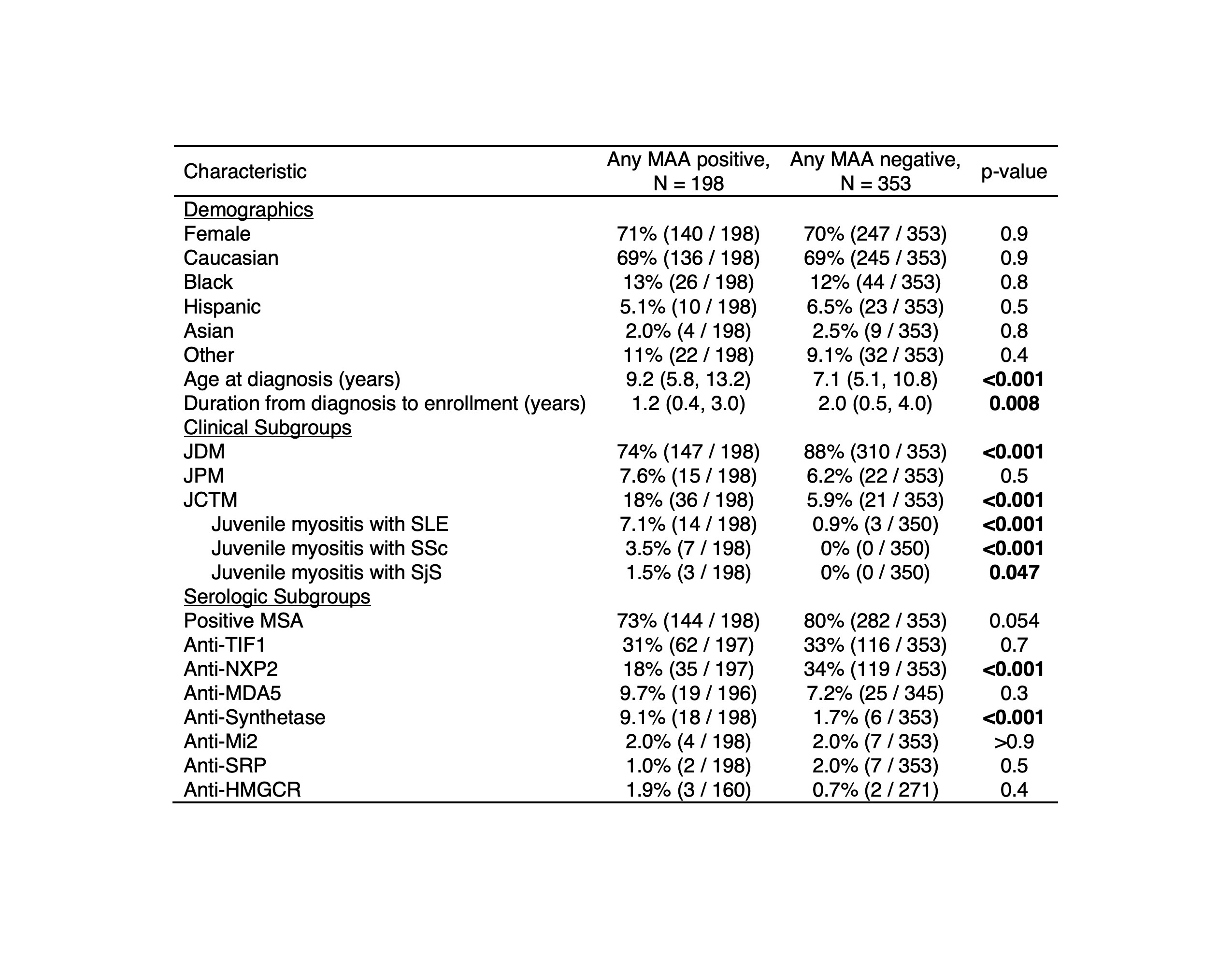
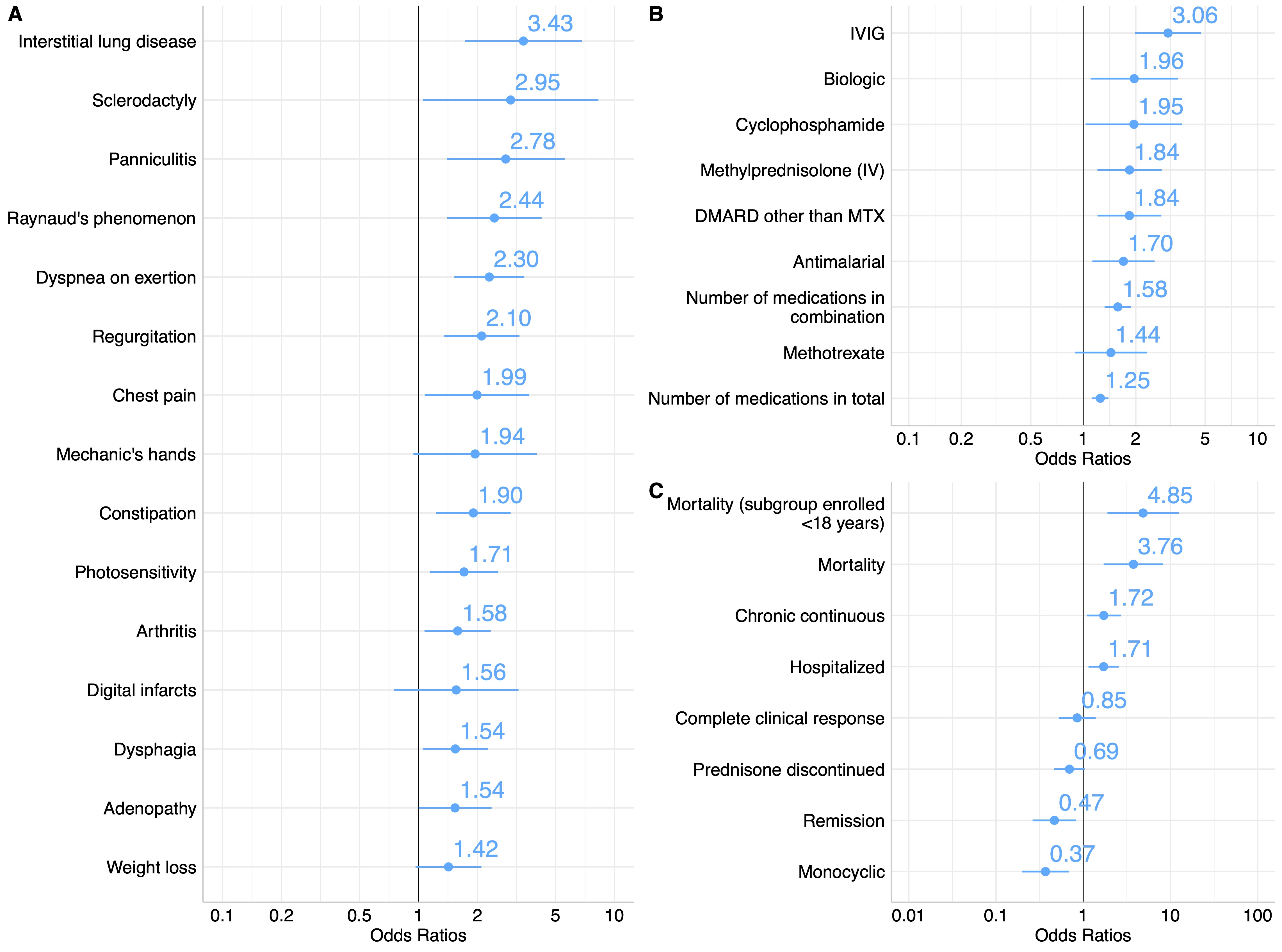
Results: MAAs were present in 36% of this North American cohort of 551 juvenile myositis patients and were found across serologic subgroups (Figure 1). Among patients with any MAA, there was a higher frequency of juvenile connective tissue myositis (JCTM) (18% vs 5.9%, p< 0.001) and lower frequency of juvenile dermatomyositis (JDM) (74% vs 88%, p< 0.001) (Table 1). Anti-synthetase Abs (9.1% vs 1.7%, p< 0.001) were more frequent and anti-NXP2 Abs (18% vs 34%, p< 0.001) less frequent among those with an MAA. The presence of an MAA was associated with certain clinical manifestations, such as ILD (OR 3.43; 95% CI 1.75-6.96), Raynaud's phenomenon (OR 2.44; 95% CI 1.41-4.28), and arthritis (OR 1.58; 95% CI 1.07-2.34) (Figure 2). The presence of any MAA was associated with increased treatment burden, including a greater odds of receiving IVIG (OR 3.06; 95% CI 1.98-4.76), IV methylprednisolone (OR 1.84; 95% CI 1.21-2.83), and a greater number of major medications in combination (OR 1.58; 95% CI 1.33-1.88). Positivity for any MAA was also associated with worse outcomes, including higher odds of mortality (OR 3.76; 95% CI 1.72-8.43) and a chronic continuous disease course (OR 1.72; 95% CI 1.10-2.72) and lower odds of a monocyclic disease course (OR 0.37; 95% CI 0.19-0.67) and achievement of remission (OR 0.47; 95% CI 0.26-0.82). Moreover, the number of MAA specificities was predictive of mortality (OR 1.83; 95% CI 1.16-2.86).
Conclusion: MAAs were prevalent in this large cohort of patients with juvenile myositis and were associated with severe disease features, more refractory disease, and mortality. Future prospective studies are needed to determine whether early detection of MAAs may improve outcomes for patients with juvenile myositis.
Acknowledgements: Intramural Research Program of NIAMS and NIEHS of the NIH.
M. Sherman: None; P. Noroozi Farhad: None; K. Pak: None; I. Pinal-Fernandez: None; K. Sarkar: None; M. Neely: None; I. Targoff: Oklahoma Medical Research Foundation Clinical Immunology Laboratory, 2, UpToDate (Wolters Kluwer), 9; F. Miller: Argenx, 1, Cabaletta, 1, Pfizer, 2, 2; A. Mammen: None; L. Rider: AstraZeneca, 5, Bristol-Myers Squibb(BMS), 5, Hope Pharmaceuticals, 5.
Background/Purpose: Myositis-associated autoantibodies (MAAs), such as anti-Ro52 autoantibodies (Abs), have been found to be associated with interstitial lung disease (ILD) and worse prognosis in the idiopathic inflammatory myopathies. MAAs remain largely uncharacterized in juvenile-onset myositis. Moreover, MAAs often co-exist and it is unknown whether the number of MAA specificities may be associated with increased disease severity.
Methods: Patients with juvenile myositis enrolled in cross-sectional NIH myositis natural history studies who underwent testing for myositis Abs were included. Demographics, clinical manifestations, treatments, and outcomes of those with and without MAAs were compared using Chi-squared, Fisher's exact test, or Wilcoxon rank-sum test. Multivariable logistic regression with adjustment for year of diagnosis and myositis autoantibodies was performed for statistically significant variables from the univariable analyses. Multivariable logistic regression was also used to determine whether the number of MAA specificities is predictive of severe disease features. A two-sided p< 0.05 was considered significant.
Results: MAAs were present in 36% of this North American cohort of 551 juvenile myositis patients and were found across serologic subgroups (Figure 1). Among patients with any MAA, there was a higher frequency of juvenile connective tissue myositis (JCTM) (18% vs 5.9%, p< 0.001) and lower frequency of juvenile dermatomyositis (JDM) (74% vs 88%, p< 0.001) (Table 1). Anti-synthetase Abs (9.1% vs 1.7%, p< 0.001) were more frequent and anti-NXP2 Abs (18% vs 34%, p< 0.001) less frequent among those with an MAA. The presence of an MAA was associated with certain clinical manifestations, such as ILD (OR 3.43; 95% CI 1.75-6.96), Raynaud's phenomenon (OR 2.44; 95% CI 1.41-4.28), and arthritis (OR 1.58; 95% CI 1.07-2.34) (Figure 2). The presence of any MAA was associated with increased treatment burden, including a greater odds of receiving IVIG (OR 3.06; 95% CI 1.98-4.76), IV methylprednisolone (OR 1.84; 95% CI 1.21-2.83), and a greater number of major medications in combination (OR 1.58; 95% CI 1.33-1.88). Positivity for any MAA was also associated with worse outcomes, including higher odds of mortality (OR 3.76; 95% CI 1.72-8.43) and a chronic continuous disease course (OR 1.72; 95% CI 1.10-2.72) and lower odds of a monocyclic disease course (OR 0.37; 95% CI 0.19-0.67) and achievement of remission (OR 0.47; 95% CI 0.26-0.82). Moreover, the number of MAA specificities was predictive of mortality (OR 1.83; 95% CI 1.16-2.86).
Conclusion: MAAs were prevalent in this large cohort of patients with juvenile myositis and were associated with severe disease features, more refractory disease, and mortality. Future prospective studies are needed to determine whether early detection of MAAs may improve outcomes for patients with juvenile myositis.
Acknowledgements: Intramural Research Program of NIAMS and NIEHS of the NIH.

FIGURE 1: Myositis-associated autoantibodies among juvenile myositis serologic subgroups

TABLE 1: Demographics and clinicoserologic subgroups among juvenile myositis patients with and without myositis-associated autoantibodies.
Abbreviations: JCTM, juvenile connective tissue myositis; JDM, juvenile dermatomyositis; JPM, juvenile polymyositis; MAA, myositis-associated autoantibody; MSA, myositis-specific autoantibody; SjS, Sjogren’s syndrome; SLE, systemic lupus erythematosus; SSc, systemic sclerosis.
Abbreviations: JCTM, juvenile connective tissue myositis; JDM, juvenile dermatomyositis; JPM, juvenile polymyositis; MAA, myositis-associated autoantibody; MSA, myositis-specific autoantibody; SjS, Sjogren’s syndrome; SLE, systemic lupus erythematosus; SSc, systemic sclerosis.

FIGURE 2: Disease features (A), medications received (B), and outcomes (C) among juvenile myositis patients with and without myositis-associated autoantibodies.
Abbreviations: DMARD, disease-modifying antirheumatic drug; IV, intravenous; IVIG, intravenous immunoglobulin; MTX, methotrexate.
Abbreviations: DMARD, disease-modifying antirheumatic drug; IV, intravenous; IVIG, intravenous immunoglobulin; MTX, methotrexate.
M. Sherman: None; P. Noroozi Farhad: None; K. Pak: None; I. Pinal-Fernandez: None; K. Sarkar: None; M. Neely: None; I. Targoff: Oklahoma Medical Research Foundation Clinical Immunology Laboratory, 2, UpToDate (Wolters Kluwer), 9; F. Miller: Argenx, 1, Cabaletta, 1, Pfizer, 2, 2; A. Mammen: None; L. Rider: AstraZeneca, 5, Bristol-Myers Squibb(BMS), 5, Hope Pharmaceuticals, 5.



