Abstract Session
Rheumatoid arthritis (RA)
Session: Abstracts: RA – Diagnosis, Manifestations, & Outcomes I: RA-ILD (0769–0774)
0774: Deciphering Rheumatoid Arthritis-associated Interstitial Lung Disease Phenotypes Using Unsupervised Hierarchical Clustering Analysis: Results from a Large Collaborative International Study
Sunday, November 12, 2023
3:15 PM - 3:25 PM PT
Location: Room 6E/6D
- PJ
Pierre-Antoine Juge, MD, PhD (he/him/his)
Division of Rheumatology, Inflammation, and Immunity Brigham & Women’s Hospital
Boston, MA, United StatesDisclosure information not submitted.
Presenting Author(s)
Pierre-Antoine Juge1, Benjamin Granger2, Lina El Houari3, Gregory McDermott4, Tracy J. Doyle4, Clive Kelly5, Gouri M Koduri6, Robert Vassallo7, Amarilys Alarcon Calderon7, Umut Kalyoncu8, Antonio Gonzalez9, Natalia Mena Vazquez10, Marta Rojas11, Raquel Dos-Santos12, Miriam Retuerto-Guerrero13, Cristina Vadillo-Font14, PALOMA VELA15, Antonio Fernandez-Nebro16, Alejandro Escudero-Contreras17, Eva Perez-Pampin18, Charlotte Hyldgaard19, Antoine Froidure20, Patrick Durez21, Jorge Rojas Serrano22, Coline van Moorsel23, Jan C Grutters23, Leticia Kawano-Dourado24, Charlotte Lucas25, Stephane Jouneau26, Raimon Sanmarti27, Raul Castellanos Moreira28, Katarina Antoniou29, Maria Molina molina30, joshua Solomon31, Sophia raia31, Miguel A Gonzalez-Gay32, Belén Atienza-Mateo33, Sofia Flouda34, Effrosyni Manali34, Boumpas Dimitrios35, Spyros Papiris34, THEOFANIS KARAGEORGAS36, Marco Sebastiani37, Andreina Manfredi38, Ana Catarina Duarte39, Santos Castañeda40, Lorenzo Cavagna41, Bruno Crestani42, Jeffrey Sparks43 and Philippe Dieudé44, 1Division of Rheumatology, Inflammation, and Immunity Brigham & Women's Hospital, Boston, MA, 2Sorbonne Université, INSERM, and Pitié Salpêtrière Hospital, Paris, France, 3Institut Pierre Louis d’Epidémiologie et de Santé Publique, Paris, France, 4Brigham and Women's Hospital, Boston, MA, 5James Cook University Hospital, Middlesbrough, United Kingdom, 6Southend University Hospitals NHS Trust, Essex, United Kingdom, 7Mayo Clinic, Rochester, MN, 8Hacettepe University, Faculty of Medicine, Department of Internal Medicine, Division of Rheumatology, Ankara, Turkey, 9Hospital Clinico Universitario de Santiago, Santiago de Compostela, Spain, 10IBIMA, Málaga, Spain, 11Hospital Universitario Reina Sofía, Cordoba, Spain, 12Clinical University Hospital in Santiago de Compostela, Santiago de Compostela, Spain, 13Rheumatology, Hospital Universitario de León, León, Spain, 14Hospital Clínico San Carlos – Instituto Investigación Sanitaria San Carlos (IdISSC), Madrid, Spain, 15Rheumatology, Hospital General Universitario Alicante, Alicante, Spain, 16Hospital Regional Universitario de Málaga, Malaga, Spain, 17Rheumatology Department, Reina Sofia University Hospital, Cordoba/Maimonides Biomedical Research Institute of Cordoba (IMIBIC)/University of Cordoba, Córdoba, Spain, 18Rheumatology Department Complejo Hospitalario Universitario Santiago, Santiago de compostela, Spain, 19Silkeborg Regional Hospital, Silkeborg, Denmark, 20Université Catholique de Louvain, Louvain, Belgium, 21UCLouvain, Brussels, Belgium, 22Hospital Angeles Villahermosa, Ciudad de Mexico, Mexico, 23St. Antonius Hospital, Nieuwegein, Netherlands, 24Hospital do Coração (HCor), São Paulo, Brazil, 25CHU, Rennes, France, 26Amicus, Boca Raton, FL, 27Hospital Clinic, Rheumatology, Barcelona, Spain, 28Hospital Clínic de Barcelona, Madrid, Spain, 29University of Crete, Heraklion, Greece, 30IDIBELL Bellvitge Biomedical Research Institute, Barcelona, Spain, 31National Jewish Health, Denver, CO, 32IDIVAL and School of Medicine, UC, Santander; Department of Rheumatology, IIS-Fundación Jiménez Díaz, Madrid, Santander, Spain, 33Rheumatology, Hospital Universitario Marqués de Valdecilla, IDIVAL, Santander, Spain, 34Unit of Attikon University Hospital, Athens, Greece, 35National and Kapodistrian University of Athens, Athens, Greece, 36n/a, Athina, Greece, 37Azienda Policlinico di Modena, Modena, Italy, 38University of Modena, Modena, Italy, 39Hospital Garcia de Orta, Almada, Portugal, 40Hospital Universitario de la Princesa, Madrid, Spain, 41Fondazione IRCCS Policlinico San Matteo, Pavia, Italy, 42Hopital Bichat, Paris University, Paris, France, 43Division of Rheumatology, Inflammation, and Immunity, Brigham and Women’s Hospital and Harvard Medical School, Boston, MA, 44Assistance Publique-Hôpitaux de Paris, Bichat-Claude Bernard University Hospital, INSERM UMR1152, University de Paris Cité, Department of Rheumatology, Paris, France
Background/Purpose: RA-associated interstitial lung disease (ILD) is not a single entity as illustrated by different HRCT patterns, different risk factors and prognoses suggesting heterogenous phenotypes. Our objective was to identify definite sub-phenotypes among a large RA-ILD dataset performing unsupervised clustering analysis.
Methods: In this international collaborative study (22 centers from 13 countries), we included patients with RA (ACR/EULAR 2010 classification criteria) having ILD defined by high-resolution computed tomography (HRCT) chest scan. Clinical, biological, imaging, and functional data were retrospectively collected from extensive chart review. Missing data were handled using multiple imputation with chained equations. A multiple correspondence analysis was performed on each sample to reduce dimensions and a non-parametric Bayesian algorithm was used to attribute a cluster to each patient. The clusters were aggregated over the multiple imputations using weighted Non-negative Matrix Factorization.MUC5B rs35705950 T risk allele distribution in the identified clusters was investigate for patients with available genotype. A surrogate decision tree was conducted using the non-imputed dataset to predict a cluster for each patient. The prediction error rate of the tree was estimated using cross-validation.
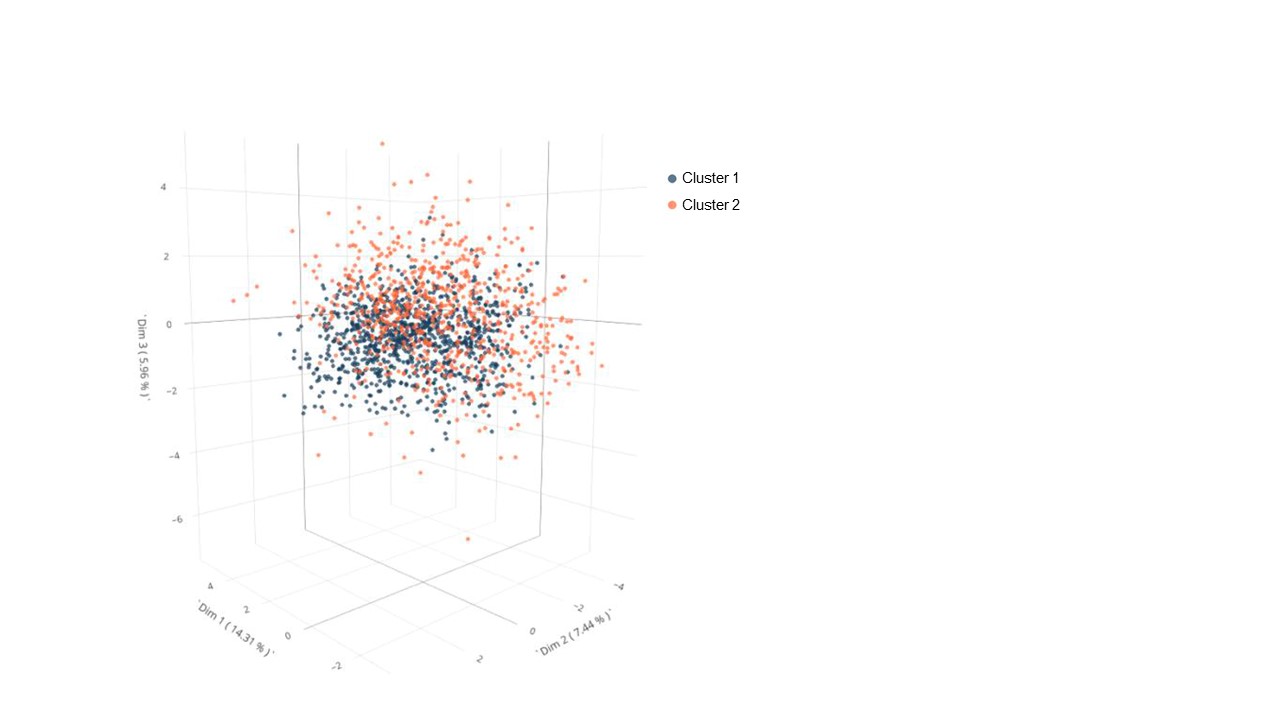
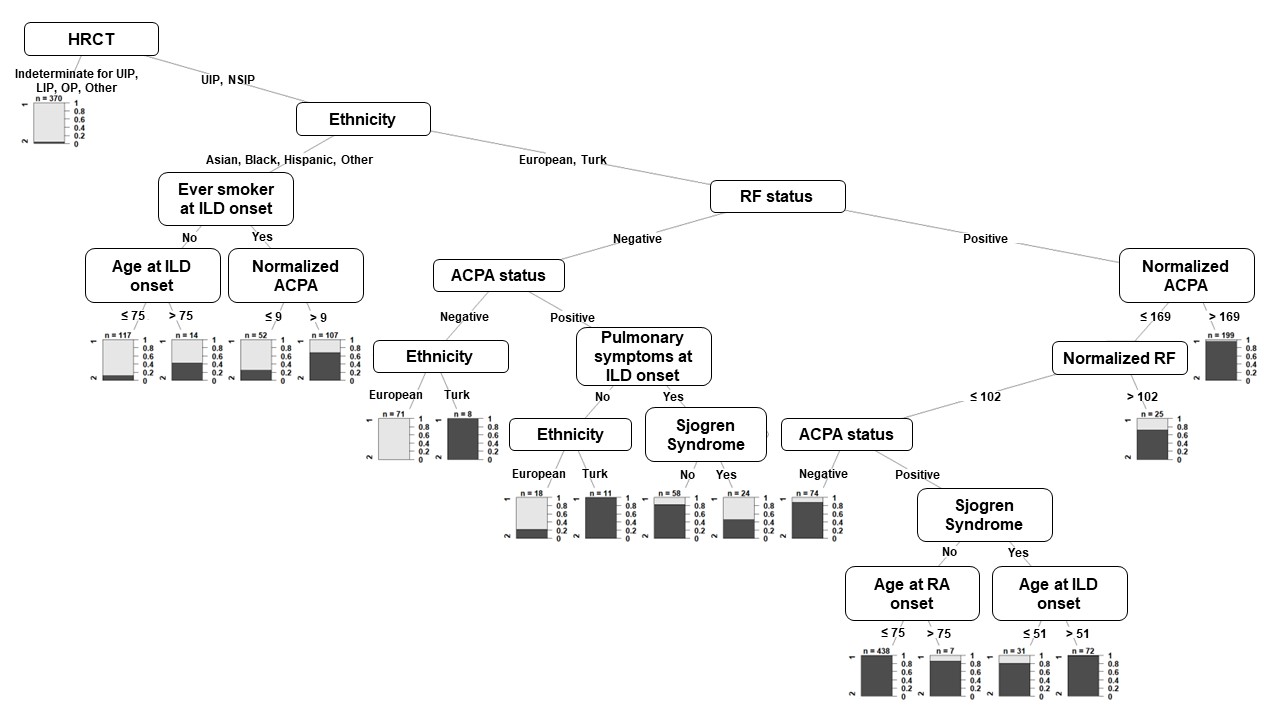
Results: A total of 1696 patients with RA-ILD were included in the cluster analysis; Table 1. Among the 37 collected variables, 20 active variables were used to construct clusters. The non-parametric Bayesian clustering algorithm detected 2 definite clusters: cluster 1 (n=1015) and cluster 2 (n=681); Table1, Figure 1. Compared to cluster 2, patients from cluster 1 were more frequently male (45.5% vs 38.3%, p=0.003), older at RA onset (56.9 ± 13.3 vs 53.8 ± 14.5, p< 0.001) and at ILD onset (65.5 ± 10.1 vs 61.9 ± 12.3, p< 0.001), more frequently with European ethnicity (85.9% vs 68.3%, p< 0.001), more frequently ever smokers at ILD onset (64.2% vs 55.7%, p< 0.001) and had less Sjogren syndrome (10.8% vs 20.6%, p< 0.001). They were more frequently positive for anti-citrullinated peptides antibodies (90.7% vs 70.0%, p< 0.001) and for rheumatoid factor (95.8% vs 67.4%, p< 0.001). UIP HRCT pattern was predominant in cluster 1 (72.5%) whereas various patterns were observed in cluster 2 (UIP = 30.8%, NSIP = 17.2%), p< 0.001. There was no difference in lung function at ILD onset between the 2 clusters. MUC5B rs35705950 genotype was available for 444 patient (26.2%) with a higher minor allele frequency (MAF) in cluster 1 (33% compared to 18%, p< 0.001). Figure 2 shows the decision tree that can be used to predict the cluster for a new patient with a prediction error rate of 9.8%.
Conclusion: Our results provide evidence that RA-ILD is a heterogeneous disease and allow the identification of at least two distinct subsets. This heterogeneity is illustrated by a contribution of the MUC5B promoter variant restricted to cluster 1, promoting the identification of specific risk scores for each RA-ILD subset. This heterogeneity underlies distinct physio pathological mechanisms which could influence not only the prognosis of patients with RA-ILD but also their therapeutic management.
.jpg)
P. Juge: None; B. Granger: Bristol-Myers Squibb(BMS), 2; L. El Houari: None; G. McDermott: None; T. Doyle: None; C. Kelly: None; G. Koduri: None; R. Vassallo: None; A. Alarcon Calderon: None; U. Kalyoncu: None; A. Gonzalez: None; N. Mena Vazquez: None; M. Rojas: None; R. Dos-Santos: None; M. Retuerto-Guerrero: None; C. Vadillo-Font: None; P. VELA: AbbVie/Abbott, 5, AstraZeneca, 5, Eli Lilly, 5, 6, GlaxoSmithKlein(GSK), 6, Novartis, 5, Pfizer, 5; A. Fernandez-Nebro: None; A. Escudero-Contreras: None; E. Perez-Pampin: None; C. Hyldgaard: None; A. Froidure: None; P. Durez: None; J. Rojas Serrano: None; C. van Moorsel: None; J. Grutters: None; L. Kawano-Dourado: None; C. Lucas: None; S. Jouneau: None; R. Sanmarti: None; R. Castellanos Moreira: None; K. Antoniou: None; M. Molina molina: None; j. Solomon: None; S. raia: None; M. Gonzalez-Gay: AbbVie/Abbott, 5, 6, Amgen, 5, 6, Pfizer, 5, 6; B. Atienza-Mateo: None; S. Flouda: None; E. Manali: None; B. Dimitrios: None; S. Papiris: None; T. KARAGEORGAS: None; M. Sebastiani: None; A. Manfredi: None; A. Duarte: None; S. Castañeda: None; L. Cavagna: None; B. Crestani: None; J. Sparks: AbbVie, 2, Amgen, 2, Boehringer Ingelheim, 2, Bristol-Myers Squibb, 2, 5, Gilead, 2, Inova Diagnostics, 2, Janssen, 2, Optum, 2, Pfizer, 2, ReCor, 2; P. Dieudé: AbbVie, 2, 6, Boehringer Ingelheim, 1, 2, 6, Bristol-Myers Squibb, 1, 2, 5, 6, Chugai, 5, Galapagos, 5, 6, Janssen, 2, 6, Novartis, 2, Pfizer, 1, 2, 5, 6.
Background/Purpose: RA-associated interstitial lung disease (ILD) is not a single entity as illustrated by different HRCT patterns, different risk factors and prognoses suggesting heterogenous phenotypes. Our objective was to identify definite sub-phenotypes among a large RA-ILD dataset performing unsupervised clustering analysis.
Methods: In this international collaborative study (22 centers from 13 countries), we included patients with RA (ACR/EULAR 2010 classification criteria) having ILD defined by high-resolution computed tomography (HRCT) chest scan. Clinical, biological, imaging, and functional data were retrospectively collected from extensive chart review. Missing data were handled using multiple imputation with chained equations. A multiple correspondence analysis was performed on each sample to reduce dimensions and a non-parametric Bayesian algorithm was used to attribute a cluster to each patient. The clusters were aggregated over the multiple imputations using weighted Non-negative Matrix Factorization.MUC5B rs35705950 T risk allele distribution in the identified clusters was investigate for patients with available genotype. A surrogate decision tree was conducted using the non-imputed dataset to predict a cluster for each patient. The prediction error rate of the tree was estimated using cross-validation.
Results: A total of 1696 patients with RA-ILD were included in the cluster analysis; Table 1. Among the 37 collected variables, 20 active variables were used to construct clusters. The non-parametric Bayesian clustering algorithm detected 2 definite clusters: cluster 1 (n=1015) and cluster 2 (n=681); Table1, Figure 1. Compared to cluster 2, patients from cluster 1 were more frequently male (45.5% vs 38.3%, p=0.003), older at RA onset (56.9 ± 13.3 vs 53.8 ± 14.5, p< 0.001) and at ILD onset (65.5 ± 10.1 vs 61.9 ± 12.3, p< 0.001), more frequently with European ethnicity (85.9% vs 68.3%, p< 0.001), more frequently ever smokers at ILD onset (64.2% vs 55.7%, p< 0.001) and had less Sjogren syndrome (10.8% vs 20.6%, p< 0.001). They were more frequently positive for anti-citrullinated peptides antibodies (90.7% vs 70.0%, p< 0.001) and for rheumatoid factor (95.8% vs 67.4%, p< 0.001). UIP HRCT pattern was predominant in cluster 1 (72.5%) whereas various patterns were observed in cluster 2 (UIP = 30.8%, NSIP = 17.2%), p< 0.001. There was no difference in lung function at ILD onset between the 2 clusters. MUC5B rs35705950 genotype was available for 444 patient (26.2%) with a higher minor allele frequency (MAF) in cluster 1 (33% compared to 18%, p< 0.001). Figure 2 shows the decision tree that can be used to predict the cluster for a new patient with a prediction error rate of 9.8%.
Conclusion: Our results provide evidence that RA-ILD is a heterogeneous disease and allow the identification of at least two distinct subsets. This heterogeneity is illustrated by a contribution of the MUC5B promoter variant restricted to cluster 1, promoting the identification of specific risk scores for each RA-ILD subset. This heterogeneity underlies distinct physio pathological mechanisms which could influence not only the prognosis of patients with RA-ILD but also their therapeutic management.
.jpg)
Table 1. Distribution of active variables within the clusters among patients with RA-ILD (n=1696). Data are numbers (percentages) for categorical variables or means (Standard Deviation) for continuous variables. 95% confidence interval for clustering analysis are indicated in the brackets for each variable. Normalized ACPA and RF represents fold above upper limit normal.
ACPA: anti-citrullinated peptides antibodies; DLCO: diffusion capacity of the lung for carbon monoxide; HRCT: high-resolution computed tomography; ILD: interstitial lung disease; LIP: lymphocytic interstitial pneumonia; NSIP: non-specific interstitial pneumonia; OP: organizing pneumonia; RF: rheumatoid factor; UIP: usual interstitial pneumonia
ACPA: anti-citrullinated peptides antibodies; DLCO: diffusion capacity of the lung for carbon monoxide; HRCT: high-resolution computed tomography; ILD: interstitial lung disease; LIP: lymphocytic interstitial pneumonia; NSIP: non-specific interstitial pneumonia; OP: organizing pneumonia; RF: rheumatoid factor; UIP: usual interstitial pneumonia

Figure 1. Clusters representation on the first three dimensions of Multiple Correspondence Analysis (MCA)
The map is a 3D representation of patients and their clusters (n = 1696). A point represents a patient, and its color represents the cluster assigned to this patient (cluster 1 in blue and cluster 2 in orange). The first three dimensions of MCA together explain 27.7% of the total variance.
The map is a 3D representation of patients and their clusters (n = 1696). A point represents a patient, and its color represents the cluster assigned to this patient (cluster 1 in blue and cluster 2 in orange). The first three dimensions of MCA together explain 27.7% of the total variance.

Figure 2. Decision tree to predict a cluster for a new patient with RA-ILD
The tree is a set of paths. Each path is a sequence of conditions that lead to a decision (cluster 1 or cluster 2). The nodes present active variables that are important in the decision-making and the edges present conditions to be checked by a patient in order to classify him in one of the two clusters. The barplot (final node of each path) presents the two clusters (cluster 1 and cluster 2) on the left and the probability to be in cluster 1 on the right. ACPA: anti-citrullinated peptides antibodies; HRCT: high-resolution computed tomography; ILD: interstitial lung disease; LIP: lymphocytic interstitial pneumonia; NSIP: non-specific interstitial pneumonia; OP: organizing pneumonia; RF: rheumatoid factor; UIP: usual interstitial pneumonia
The tree is a set of paths. Each path is a sequence of conditions that lead to a decision (cluster 1 or cluster 2). The nodes present active variables that are important in the decision-making and the edges present conditions to be checked by a patient in order to classify him in one of the two clusters. The barplot (final node of each path) presents the two clusters (cluster 1 and cluster 2) on the left and the probability to be in cluster 1 on the right. ACPA: anti-citrullinated peptides antibodies; HRCT: high-resolution computed tomography; ILD: interstitial lung disease; LIP: lymphocytic interstitial pneumonia; NSIP: non-specific interstitial pneumonia; OP: organizing pneumonia; RF: rheumatoid factor; UIP: usual interstitial pneumonia
P. Juge: None; B. Granger: Bristol-Myers Squibb(BMS), 2; L. El Houari: None; G. McDermott: None; T. Doyle: None; C. Kelly: None; G. Koduri: None; R. Vassallo: None; A. Alarcon Calderon: None; U. Kalyoncu: None; A. Gonzalez: None; N. Mena Vazquez: None; M. Rojas: None; R. Dos-Santos: None; M. Retuerto-Guerrero: None; C. Vadillo-Font: None; P. VELA: AbbVie/Abbott, 5, AstraZeneca, 5, Eli Lilly, 5, 6, GlaxoSmithKlein(GSK), 6, Novartis, 5, Pfizer, 5; A. Fernandez-Nebro: None; A. Escudero-Contreras: None; E. Perez-Pampin: None; C. Hyldgaard: None; A. Froidure: None; P. Durez: None; J. Rojas Serrano: None; C. van Moorsel: None; J. Grutters: None; L. Kawano-Dourado: None; C. Lucas: None; S. Jouneau: None; R. Sanmarti: None; R. Castellanos Moreira: None; K. Antoniou: None; M. Molina molina: None; j. Solomon: None; S. raia: None; M. Gonzalez-Gay: AbbVie/Abbott, 5, 6, Amgen, 5, 6, Pfizer, 5, 6; B. Atienza-Mateo: None; S. Flouda: None; E. Manali: None; B. Dimitrios: None; S. Papiris: None; T. KARAGEORGAS: None; M. Sebastiani: None; A. Manfredi: None; A. Duarte: None; S. Castañeda: None; L. Cavagna: None; B. Crestani: None; J. Sparks: AbbVie, 2, Amgen, 2, Boehringer Ingelheim, 2, Bristol-Myers Squibb, 2, 5, Gilead, 2, Inova Diagnostics, 2, Janssen, 2, Optum, 2, Pfizer, 2, ReCor, 2; P. Dieudé: AbbVie, 2, 6, Boehringer Ingelheim, 1, 2, 6, Bristol-Myers Squibb, 1, 2, 5, 6, Chugai, 5, Galapagos, 5, 6, Janssen, 2, 6, Novartis, 2, Pfizer, 1, 2, 5, 6.



