Poster Session C
Spondyloarthritis (SpA) including psoriatic arthritis (PsA)
Session: (2227–2256) Spondyloarthritis Including Psoriatic Arthritis – Treatment Poster III: SpA
2241.5: Long-term Retention of Secukinumab and Effect of Treatment Interruption in Patients with Active Psoriatic Arthritis or Ankylosing Spondylitis: Results from the Observational SERENA Study
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- UK
Abstract Poster Presenter(s)
Uta Kiltz1, Petros Sfikakis2, Nicola Gullick3, Pelagia Katsimbri4, Anastasios Kotrotsios5, Jan Brandt-Juergens6, Eric Lespessailles7, Nicola Maiden8, Karl Gaffney9, Daniel Peterlik10, Barbara Schulz11 and Piotr Jagiello12, 1Rheumazentrum Ruhrgebiet, Herne, Germany, 2National Kapodistrian University of Athens Medical School, Athens, Greece, 3University Hospitals Coventry & Warwickshire, Coventry, UK and Warwick Medical School, University of Warwick, Coventry, United Kingdom, 4Rheumatology and Clinical Immunology Unit, 4th Dept of Internal Medicine, Medical School, National and Kapodistrian University of Athens, Attikon University Hospital of Athens, Athens, Greece, 5Adjunct Professor of Anatomy, Faculty of Public and Integrated Health, University of Thessaly and Research Associate of IASO Hospital, Thessaly, Greece, 6rheumatologische Schwerpunktpraxis, Berlin, Germany, 7Regional University Hospital of Orleans, Orleans, France, 8Craigavon Area Hospital, Southern Health & Social Care Trust, Portadown, Craigavon, United Kingdom, 9Norfolk and Norwich University Hospitals NHS Foundation Trust, Norwich, United Kingdom, 10Novartis Pharma GmbH, Nürnberg, Germany, 11GKM Gesellschaft fuer Therapieforschung mbH, Lessingstrasse, München, Germany, 12Novartis Pharma AG, Basel, Switzerland
Background/Purpose: SERENA is an ongoing, longitudinal, observational study with more than 2900 patients (pts) with moderate to severe psoriasis, active psoriatic arthritis (PsA), and ankylosing spondylitis (AS) conducted at 438 sites across Europe and an expected duration of up to 5 years.1 Herein, we report long-term data on retention and effectiveness of secukinumab (SEC) and impact of intermediate treatment interruption in pts with active PsA or AS from the SERENA study.
Methods: This analysis includes data of 524 and 473 pts with PsA and AS enrolled in the study and followed up for ≥3 years. Pts (aged ≥18 years) had received ≥16 weeks of SEC treatment prior to enrollment in the study. Retention rate was defined as percentage of pts who have not discontinued SEC treatment. Effectiveness assessments included swollen and tender joint counts (SJC and TJC) in pts with PsA, and Pt Global Assessment (PtGA) and Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) score in pts with AS. Treatment interruption was defined as interruption of SEC therapy for ≥3 months between the last injection and re-initiation.
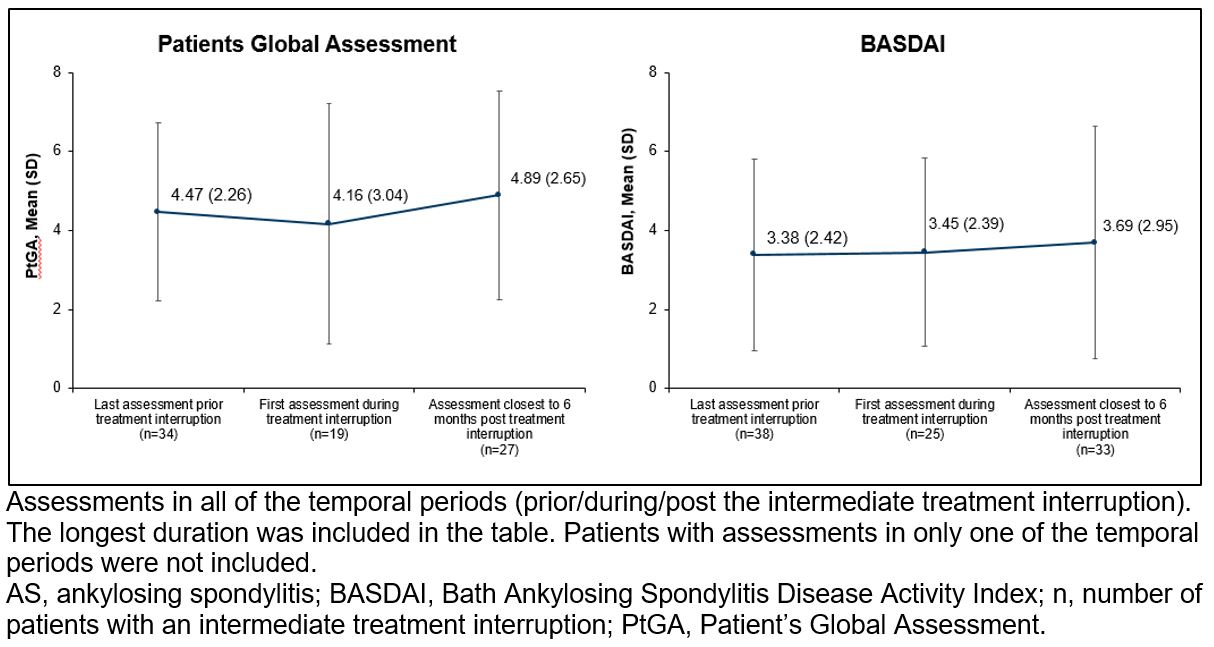
Results: In total, 101 (19.3%) and 422 (80.7%) pts with PsA, and 460 (97.3%) and 13 (2.7%) pts with AS received SEC 150 and 300 mg, respectively. Within 3 years of follow up period, 9.7% pts with PsA and 14.2% of pts with AS had ≥1 increase in dosage. After ≥3 years since enrollment, retention rates were 67.3% and 72.1% for pts with PsA and AS, respectively. After ≥3 years since SEC initiation (prior enrollment to the study), retention rates were 74.1% and 77.4% for pts with PsA and AS, respectively. The mean (SD) SJC (BL: 3.2 [5.6] and year 3: 1.7 [2.7]) and TJC (BL: 6.4 [9.4] and year 3: 4.9 [6.4]) in pts with PsA and mean (SD) BASDAI scores (BL: 3.2 [2.3] and year 3: 2.7 [2.2]) in pts with AS were sustained through 3 years after enrollment. Overall, 38 (7.3%) pts with PsA and 55 (11.6%) pts with AS experienced ≥1 intermediate treatment interruption. Most common reason for treatment interruption was adverse events (Figure 1). Median duration of SEC treatment was 675 and 557 days prior and after treatment interruption for pts with PsA, respectively, and 668 and 535 days prior and after interruption for pts with AS, respectively. Median treatment interruption duration was greater for pts with PsA and AS who restarted treatment with re-loading dose (183 days each) compared with those who restarted treatment with maintenance dose (PsA, 106; AS, 92 days). Mean (SD) SJC and TJC scores increased in pts with PsA from the last assessment prior to the treatment interruption to the first assessment during the treatment interruption, and gradually decreased post SEC re-initiation (Figure 2). PtGA and BASDAI remained stable in pts with AS from the last assessment prior to the treatment interruption to the first assessment during the treatment interruption and after SEC re-initiation (Figure 3).
Conclusion: SEC demonstrated high retention rates and sustained effectiveness up to 3 years of follow-up in the real-world population with PsA and AS. Treatment interruption occurred due to a variety of reasons but had no notable impact on long-term effectiveness of SEC.
Reference:
1. Augustin M, et al. JEur Acad Dermatol Venereol. 2022;36(10):1796–1804.
.jpg)
.jpg)
U. Kiltz: AbbVie, 2, 5, 6, Amgen, 5, Biocad, 2, 6, Biogen, 5, Bristol-Myers Squibb(BMS), 2, 5, Chugai, 2, 6, Eli Lilly, 2, 6, Fresenius, 5, Gilead, 2, 5, GlaxoSmithKline (GSK), 5, Grünenthal, 2, 6, Hexal, 5, Janssen, 2, 6, MSD, 2, 6, Novartis, 2, 5, 6, onkowiessen.de, 2, 5, Pfizer, 2, 5, 6, Roche, 2, 6, UCB, 2, 6, Viatris, 2, 5; P. Sfikakis: AbbVie/Abbott, 2, 5, Amgen, 2, 5, Boehringer-Ingelheim, 2, 5, Celgene, 2, 5, Eli Lilly, 2, 5, Janssen, 2, 5, Novartis, 2, 5, Pfizer, 2, 5; N. Gullick: AbbVie/Abbott, 2, 5, 6, AstraZeneca, 5, Eli Lilly, 5, 6, Janssen, 2, 5, 6, Novartis, 2, 5, 6, UCB, 2, 6; P. Katsimbri: AbbVie/Abbott, 2, 5, 6, Genesis Pharma, 2, 5, 6, Janssen, 2, 5, 6, Novartis, 2, 5, 6, Pfizer, 2, 5, 6, UCB, 2, 5, 6; A. Kotrotsios: None; J. Brandt-Juergens: AbbVie/Abbott, 2, 6, Affibody, 2, Bristol-Myers Squibb(BMS), 2, 6, Eli Lilly, 2, 6, Gilead, 2, Janssen, 2, 6, Medac, 2, 6, Merck/MSD, 2, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 2, 6, Sanofi-Aventis, 2, 6, UCB, 2, 6; E. Lespessailles: AbbVie/Abbott, 5, 6, Amgen, 2, 5, 6, Eli Lilly, 2, 5, 6, Expanscience, 2, 6, Galapagos, 6, Merck/MSD, 2, 5, 6, Novartis, 5, UCB, 5; N. Maiden: Eli Lilly, 2, UCB, 2; K. Gaffney: AbbVie/Abbott, 2, 5, 6, Celgene, 2, 5, 6, Eli Lilly, 2, 5, 6, Gilead, 2, 5, 6, Merck/MSD, 2, 5, 6, Novartis, 2, 5, 6, Pfizer, 2, 5, 6, UCB, 2, 5, 6; D. Peterlik: Novartis, 3; B. Schulz: GKM Gesellschaft fuer Therapieforschung mbH, 3, Novartis, 12, Provides services; P. Jagiello: Novartis, 3.
Background/Purpose: SERENA is an ongoing, longitudinal, observational study with more than 2900 patients (pts) with moderate to severe psoriasis, active psoriatic arthritis (PsA), and ankylosing spondylitis (AS) conducted at 438 sites across Europe and an expected duration of up to 5 years.1 Herein, we report long-term data on retention and effectiveness of secukinumab (SEC) and impact of intermediate treatment interruption in pts with active PsA or AS from the SERENA study.
Methods: This analysis includes data of 524 and 473 pts with PsA and AS enrolled in the study and followed up for ≥3 years. Pts (aged ≥18 years) had received ≥16 weeks of SEC treatment prior to enrollment in the study. Retention rate was defined as percentage of pts who have not discontinued SEC treatment. Effectiveness assessments included swollen and tender joint counts (SJC and TJC) in pts with PsA, and Pt Global Assessment (PtGA) and Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) score in pts with AS. Treatment interruption was defined as interruption of SEC therapy for ≥3 months between the last injection and re-initiation.
Results: In total, 101 (19.3%) and 422 (80.7%) pts with PsA, and 460 (97.3%) and 13 (2.7%) pts with AS received SEC 150 and 300 mg, respectively. Within 3 years of follow up period, 9.7% pts with PsA and 14.2% of pts with AS had ≥1 increase in dosage. After ≥3 years since enrollment, retention rates were 67.3% and 72.1% for pts with PsA and AS, respectively. After ≥3 years since SEC initiation (prior enrollment to the study), retention rates were 74.1% and 77.4% for pts with PsA and AS, respectively. The mean (SD) SJC (BL: 3.2 [5.6] and year 3: 1.7 [2.7]) and TJC (BL: 6.4 [9.4] and year 3: 4.9 [6.4]) in pts with PsA and mean (SD) BASDAI scores (BL: 3.2 [2.3] and year 3: 2.7 [2.2]) in pts with AS were sustained through 3 years after enrollment. Overall, 38 (7.3%) pts with PsA and 55 (11.6%) pts with AS experienced ≥1 intermediate treatment interruption. Most common reason for treatment interruption was adverse events (Figure 1). Median duration of SEC treatment was 675 and 557 days prior and after treatment interruption for pts with PsA, respectively, and 668 and 535 days prior and after interruption for pts with AS, respectively. Median treatment interruption duration was greater for pts with PsA and AS who restarted treatment with re-loading dose (183 days each) compared with those who restarted treatment with maintenance dose (PsA, 106; AS, 92 days). Mean (SD) SJC and TJC scores increased in pts with PsA from the last assessment prior to the treatment interruption to the first assessment during the treatment interruption, and gradually decreased post SEC re-initiation (Figure 2). PtGA and BASDAI remained stable in pts with AS from the last assessment prior to the treatment interruption to the first assessment during the treatment interruption and after SEC re-initiation (Figure 3).
Conclusion: SEC demonstrated high retention rates and sustained effectiveness up to 3 years of follow-up in the real-world population with PsA and AS. Treatment interruption occurred due to a variety of reasons but had no notable impact on long-term effectiveness of SEC.
Reference:
1. Augustin M, et al. JEur Acad Dermatol Venereol. 2022;36(10):1796–1804.
.jpg)
Figure 1. Reasons for a secukinumab treatment interruption in patients with PsA and AS
.jpg)
Figure 2. SJC and TJC score prior treatment interruption, during the treatment interruption and post re-initiation in patients with PsA

Figure 3. PtGA and BASDAI score prior treatment interruption, during the treatment interruption, and post re-initiation in patients with AS
U. Kiltz: AbbVie, 2, 5, 6, Amgen, 5, Biocad, 2, 6, Biogen, 5, Bristol-Myers Squibb(BMS), 2, 5, Chugai, 2, 6, Eli Lilly, 2, 6, Fresenius, 5, Gilead, 2, 5, GlaxoSmithKline (GSK), 5, Grünenthal, 2, 6, Hexal, 5, Janssen, 2, 6, MSD, 2, 6, Novartis, 2, 5, 6, onkowiessen.de, 2, 5, Pfizer, 2, 5, 6, Roche, 2, 6, UCB, 2, 6, Viatris, 2, 5; P. Sfikakis: AbbVie/Abbott, 2, 5, Amgen, 2, 5, Boehringer-Ingelheim, 2, 5, Celgene, 2, 5, Eli Lilly, 2, 5, Janssen, 2, 5, Novartis, 2, 5, Pfizer, 2, 5; N. Gullick: AbbVie/Abbott, 2, 5, 6, AstraZeneca, 5, Eli Lilly, 5, 6, Janssen, 2, 5, 6, Novartis, 2, 5, 6, UCB, 2, 6; P. Katsimbri: AbbVie/Abbott, 2, 5, 6, Genesis Pharma, 2, 5, 6, Janssen, 2, 5, 6, Novartis, 2, 5, 6, Pfizer, 2, 5, 6, UCB, 2, 5, 6; A. Kotrotsios: None; J. Brandt-Juergens: AbbVie/Abbott, 2, 6, Affibody, 2, Bristol-Myers Squibb(BMS), 2, 6, Eli Lilly, 2, 6, Gilead, 2, Janssen, 2, 6, Medac, 2, 6, Merck/MSD, 2, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 2, 6, Sanofi-Aventis, 2, 6, UCB, 2, 6; E. Lespessailles: AbbVie/Abbott, 5, 6, Amgen, 2, 5, 6, Eli Lilly, 2, 5, 6, Expanscience, 2, 6, Galapagos, 6, Merck/MSD, 2, 5, 6, Novartis, 5, UCB, 5; N. Maiden: Eli Lilly, 2, UCB, 2; K. Gaffney: AbbVie/Abbott, 2, 5, 6, Celgene, 2, 5, 6, Eli Lilly, 2, 5, 6, Gilead, 2, 5, 6, Merck/MSD, 2, 5, 6, Novartis, 2, 5, 6, Pfizer, 2, 5, 6, UCB, 2, 5, 6; D. Peterlik: Novartis, 3; B. Schulz: GKM Gesellschaft fuer Therapieforschung mbH, 3, Novartis, 12, Provides services; P. Jagiello: Novartis, 3.

