Poster Session C
Spondyloarthritis (SpA) including psoriatic arthritis (PsA)
Session: (2195–2226) Spondyloarthritis Including Psoriatic Arthritis – Diagnosis, Manifestations, & Outcomes Poster III: SpA
2197: Diagnostic Delay in Patients Included in the International Map of Axial Spondyloarthrtis: Associations with Geographic, Socio-demographic and Disease-related Factors
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
.png)
Denis Poddubnyy, MD, PhD, MSc
Charité-Universitätsmedizin Berlin
Berlin, GermanyDisclosure information not submitted.
Abstract Poster Presenter(s)
Denis Poddubnyy1, Marco Garrido-Cumbrera2, Fernando Sommerfleck3, Victoria Navarro-Compán4, Christine Bundy5, Souzi Makri6, Jose Correa Fernandez7, Shashank Murlidhar Akerkar8, Jo Davies9, Laura Christen10 and Elie Karam11, 1Department of Gastroenterology, Infectious Diseases and Rheumatology, Charité – Universitätsmedizin Berlin, Berlin, Germany, 2University of Seville, Sevilla, Spain, 3Sanatorio Julio Mendez, Buenos Aires, Argentina, 4Department of Rheumatology, La Paz University Hospital, IdiPaz, Madrid, Spain, 5Cardiff University, Cardiff, United Kingdom, 6Cyprus League for People with Rheumatism (CYLPER), Nicosia, Cyprus, 7Universidad de Sevilla, Health & Territory Research (HTR), Sevilla, Spain, 8Mumbai Arthritis Clinic, Mumbai, India, 9Axial Spondyloarthritis International Federation (ASIF), London, United Kingdom, 10Novartis Pharma AG, Basel, Switzerland, 11Canadian Spondylitis Association (CSA), Toronto, ON, Canada
Background/Purpose: Despite efforts for early detection, delayed diagnosis in axial spondyloarthritis (axSpA) remains an unresolved challenge. This analysis aimed to assess diagnostic delay and its associated factors around the world in a large sample of patients included in the International Map of Axial Spondyloarthritis (IMAS).
Methods: IMAS is a cross-sectional online survey (2017-2022) including 5,557 unselected axSpA patients from 27 countries. Diagnostic delay was calculated as the difference between age at diagnosis and age at symptom onset reported by patients. The independent factors evaluated were: age at symptom onset, disease duration, gender, education level, diagnosed by rheumatologist, number of HCPs seen before diagnosis, HLA-B27, uveitis, and inflammatory bowel disease. The factor world region was introduced as a dummy variable taking Europe as the reference region due to its larger sample size and diagnostic delay close to the overall mean. The Mann-Whitney, Kruskal-Wallis test and Pearson correlation were used to evaluate the differences in diagnostic delay and independent variables. Associations between diagnostic delay and regions, sociodemographic characteristics, as well as disease-related factors were explored through univariable and multivariable linear regression analysis.
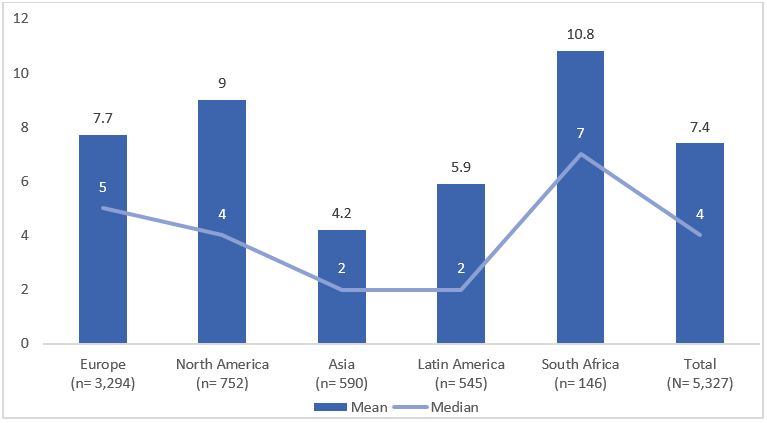
Results: Data from 5,327 patients who reported data to calculate diagnostic delay in IMAS survey were analyzed: 3,231 were from Europe, 770 from North America, 600 from Asia, 548 from Latin America, and 146 from Africa. Overall, patients reported a diagnostic delay of 7.4 years (median: 4.0) since symptom onset, with substantial variation across regions, being the highest in South Africa and the lowest in Asia (Figure 1). Furthermore, mean disease duration was 17.1 ± 13.3. Patients with longer diagnostic delay were more frequently female, younger at symptom onset, with more years with the condition, more commonly diagnosed by the rheumatologist, with a higher number of HCPs seen before diagnosis, had experienced uveitis, and inflammatory bowel disease. The variables independently associated with longer diagnostic delay in the final multivariable regression model were: younger age at symptom onset (b=-0.100), more disease duration (b=0.363), female gender (b=2.274), being diagnosed by rheumatologist (b=1.163), higher number of healthcare professionals (HCPs) seen before diagnosis (b=1.033), and presence of uveitis (b=1.286; Table 1).
Conclusion: In this global sample of axSpA patients, the mean diagnostic delay was 7.4 years, and had significant differences across regions. Younger age at symptom onset, longer disease duration, female gender, diagnosed by rheumatologist, higher number of HCPs seen before diagnosis, and the presence of uveitis were the parameters associated with a longer diagnostic delay in axSpA patients.
.jpg)
D. Poddubnyy: AbbVie, 2, 5, 6, Biocad, 2, BMS, 6, Eli Lilly, 2, 5, 6, Gilead, 2, GSK, 2, MoonLake, 2, MSD, 2, 5, 6, Novartis, 2, 5, 6, Pfizer, 2, 5, 6, Samsung Bioepis, 2, UCB Pharma, 2, 6; M. Garrido-Cumbrera: Novartis, 5; F. Sommerfleck: AbbVie/Abbott, 2, 6, Eli Lilly, 6, Janssen, 2, 6, Novartis, 2, 6; V. Navarro-Compán: AbbVie, 2, 5, 6, Eli Lilly, 2, 6, Galapagos, 2, Janssen, 6, MoonLake, 2, MSD, 2, 6, Novartis, 2, 5, 6, Pfizer, 2, 6, UCB, 2, 6; C. Bundy: AbbVie/Abbott, 6, Celgene, 6, Eli Lilly, 6, Janssen, 6, Novartis, 6, Pfizer, 6; S. Makri: GlaxoSmithKlein(GSK), 2, Novartis, 2; J. Correa Fernandez: None; S. Murlidhar Akerkar: Eli Lilly, 6, Janssen, 6, Novartis, 6, Pfizer, 6; J. Davies: AbbVie/Abbott, 12, No personal funding, but ASIF has received funding from Abbvie, Boehringer-Ingelheim, 12, No personal funding, but ASIF has received funding from Boehringer Ingleheim, Eli Lilly, 12, No personal funding, but ASIF has received funding from Lilly, Janssen, 12, No personal funding, but ASIF has received funding from Janssen, Novartis, 12, No personal funding, but ASIF has received funding from Novartis, Pfizer, 12, No personal funding, but ASIF has received funding from Pfizer, UCB, 12, No personal funding, but ASIF has received funding from UCB; L. Christen: Novartis, 3; E. Karam: None.
Background/Purpose: Despite efforts for early detection, delayed diagnosis in axial spondyloarthritis (axSpA) remains an unresolved challenge. This analysis aimed to assess diagnostic delay and its associated factors around the world in a large sample of patients included in the International Map of Axial Spondyloarthritis (IMAS).
Methods: IMAS is a cross-sectional online survey (2017-2022) including 5,557 unselected axSpA patients from 27 countries. Diagnostic delay was calculated as the difference between age at diagnosis and age at symptom onset reported by patients. The independent factors evaluated were: age at symptom onset, disease duration, gender, education level, diagnosed by rheumatologist, number of HCPs seen before diagnosis, HLA-B27, uveitis, and inflammatory bowel disease. The factor world region was introduced as a dummy variable taking Europe as the reference region due to its larger sample size and diagnostic delay close to the overall mean. The Mann-Whitney, Kruskal-Wallis test and Pearson correlation were used to evaluate the differences in diagnostic delay and independent variables. Associations between diagnostic delay and regions, sociodemographic characteristics, as well as disease-related factors were explored through univariable and multivariable linear regression analysis.
Results: Data from 5,327 patients who reported data to calculate diagnostic delay in IMAS survey were analyzed: 3,231 were from Europe, 770 from North America, 600 from Asia, 548 from Latin America, and 146 from Africa. Overall, patients reported a diagnostic delay of 7.4 years (median: 4.0) since symptom onset, with substantial variation across regions, being the highest in South Africa and the lowest in Asia (Figure 1). Furthermore, mean disease duration was 17.1 ± 13.3. Patients with longer diagnostic delay were more frequently female, younger at symptom onset, with more years with the condition, more commonly diagnosed by the rheumatologist, with a higher number of HCPs seen before diagnosis, had experienced uveitis, and inflammatory bowel disease. The variables independently associated with longer diagnostic delay in the final multivariable regression model were: younger age at symptom onset (b=-0.100), more disease duration (b=0.363), female gender (b=2.274), being diagnosed by rheumatologist (b=1.163), higher number of healthcare professionals (HCPs) seen before diagnosis (b=1.033), and presence of uveitis (b=1.286; Table 1).
Conclusion: In this global sample of axSpA patients, the mean diagnostic delay was 7.4 years, and had significant differences across regions. Younger age at symptom onset, longer disease duration, female gender, diagnosed by rheumatologist, higher number of HCPs seen before diagnosis, and the presence of uveitis were the parameters associated with a longer diagnostic delay in axSpA patients.

Figure 1. Mean and median diagnostic delays by region (N= 5,327)
.jpg)
Table 1. Univariable and multivariable linear regression analysis of the association between diagnostic delay and independent variables in patients with axial spondyloarthritis (N= 4,595)
D. Poddubnyy: AbbVie, 2, 5, 6, Biocad, 2, BMS, 6, Eli Lilly, 2, 5, 6, Gilead, 2, GSK, 2, MoonLake, 2, MSD, 2, 5, 6, Novartis, 2, 5, 6, Pfizer, 2, 5, 6, Samsung Bioepis, 2, UCB Pharma, 2, 6; M. Garrido-Cumbrera: Novartis, 5; F. Sommerfleck: AbbVie/Abbott, 2, 6, Eli Lilly, 6, Janssen, 2, 6, Novartis, 2, 6; V. Navarro-Compán: AbbVie, 2, 5, 6, Eli Lilly, 2, 6, Galapagos, 2, Janssen, 6, MoonLake, 2, MSD, 2, 6, Novartis, 2, 5, 6, Pfizer, 2, 6, UCB, 2, 6; C. Bundy: AbbVie/Abbott, 6, Celgene, 6, Eli Lilly, 6, Janssen, 6, Novartis, 6, Pfizer, 6; S. Makri: GlaxoSmithKlein(GSK), 2, Novartis, 2; J. Correa Fernandez: None; S. Murlidhar Akerkar: Eli Lilly, 6, Janssen, 6, Novartis, 6, Pfizer, 6; J. Davies: AbbVie/Abbott, 12, No personal funding, but ASIF has received funding from Abbvie, Boehringer-Ingelheim, 12, No personal funding, but ASIF has received funding from Boehringer Ingleheim, Eli Lilly, 12, No personal funding, but ASIF has received funding from Lilly, Janssen, 12, No personal funding, but ASIF has received funding from Janssen, Novartis, 12, No personal funding, but ASIF has received funding from Novartis, Pfizer, 12, No personal funding, but ASIF has received funding from Pfizer, UCB, 12, No personal funding, but ASIF has received funding from UCB; L. Christen: Novartis, 3; E. Karam: None.



