Poster Session C
Systemic lupus erythematosus (SLE)
Session: (2257–2325) SLE – Diagnosis, Manifestations, & Outcomes Poster III
2297: Interferon-α as a Biomarker to Predict Flares in Lupus Nephritis
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- LW
Laura Whittall Garcia, MD
University Health Network
Toronto, ON, CanadaDisclosure information not submitted.
Abstract Poster Presenter(s)
Laura Patricia Whittall Garcia1, Dafna Gladman2, Murray Urowitz3, Zahi Touma4 and Joan Wither1, 1University Health Network, Toronto, ON, Canada, 2Schroeder Arthritis Institute, Krembil Research Institute, Toronto Western Hospital, Department of Medicine, University of Toronto, Toronto, ON, Canada, 3Schroeder Arthritis Institute, Krembil Research Institute; University of Toronto Lupus Clinic; Division of Rheumatology, Toronto, ON, Canada, 4University of Toronto, Toronto, ON, Canada
Background/Purpose: Type I Interferons (IFN-I) play a role in SLE and Lupus Nephritis (LN) pathogenesis. We have recently shown that IFN-I gene expression predicts the risk of SLE flares and a more severe disease course in SLE patients. Furthermore, recently it has been suggested that patients with higher IFN-I gene expression in their renal tubular cells are less likely to respond to conventional therapy.
The aim of this study was to determine if the amount of IFN-α in serum at the time of an LN flare predicts response to therapy, subsequent LN flares, and decline in kidney function.
Methods: Patients with active LN were included in this study. All patients had 1) active LN, defined as a 24H urine protein >500mg/day with a subsequent modification in therapy by the treating physician, 2) stored serum sample ±3 months from the renal flare and 3) baseline estimated glomerular filtration rate (eGFR) ≥60ml/min (3 months prior to the flare).
The following outcomes were ascertained: 1) Complete Response (CR), defined as proteinuria < 500 mg/day and a serum creatinine within 15% of the baseline at 1 and 2 years after the flare, 2) number of LN flares during follow-up (defined by an increase in proteinuria of at least 1000 mg/d if the baseline was < 500 mg/d or doubling of proteinuria in baseline was ≥500 mg/d and a change in therapy by the treating physician, and 3) decline in eGFR to ≤59ml/min, ≤44ml/min,≤29ml/min and < 15ml/min during follow-up.
Serum IFN-α was measured by Simoa®
Results: A total of 95 patients with active LN were included in the study. The median (IQR) age of the patients was 29 (23-41) years, 79 (83.7%) were women, and the disease duration was 6.0 (0.2-10.0) years. Forty (42.1%) were Caucasian, 21 (22.1%) were Afro-Caribbean, and 22 (23.1.%) were Asian. The baseline eGFR was 112 (98.7-127) ml/min, and the median (IQR) follow-up was 132 (96-156) months.76.8% had a kidney biopsy at the time of the LN flare, of whom 54.7% had a proliferative or mixed class, 17.8% class V, and 4.2% class I or II. The median (IQR) of prednisone was 40mg (30-38mg), 89 (93%) received antimalarial and all patients received immunosuppressive therapy.
The serum baseline levels of IFN-α predicted CR at 2 years from the LN flare. Furthermore, patients with higher baseline levels of IFN-α had a greater risk of having 2 or more subsequent renal flares. Every increase in 1 unit of IFN-α increased the risk of having 2 or more flares by 35%, and the risk of having a decline in kidney function during follow-up (Table 1).
Using ROC analysis, two IFN-α cut-offs were identified, 0.6 and 1.3 for predicting ≥2 LN flares and progression to eGFR ≤15, respectively. Even though the areas under the curve suggested that IFN-α is only a weak predictor (AUC=0.62 (0.51-0.74) and 0.60 (0.47-0.73) for ≥2 LN flares and progression to eGFR ≤15, respectively), these cutoffs continued to predict renal outcomes on Cox regression analysis, as seen in Figure 2 and Table 2.
Conclusion: IFN-α serum levels predicted failure to respond to treatment at year 2 after the renal flare, the development of ≥2 LN flares, and decline in kidney function during follow-up.
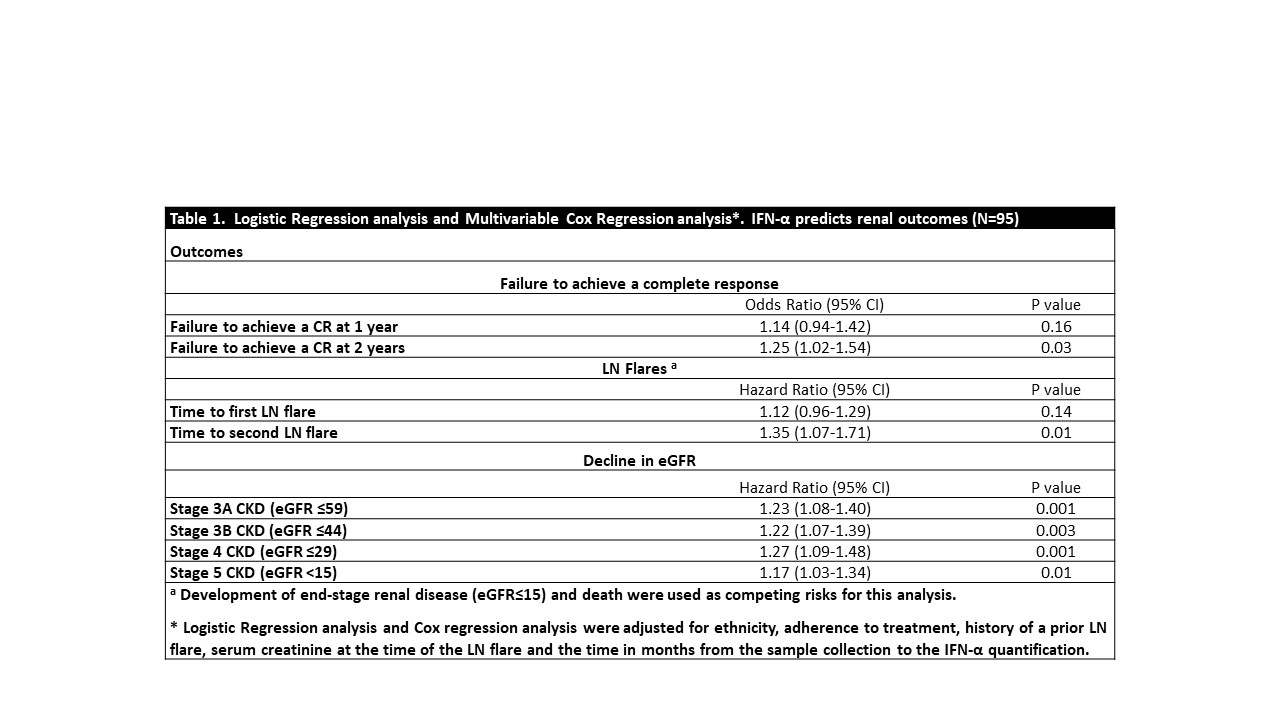
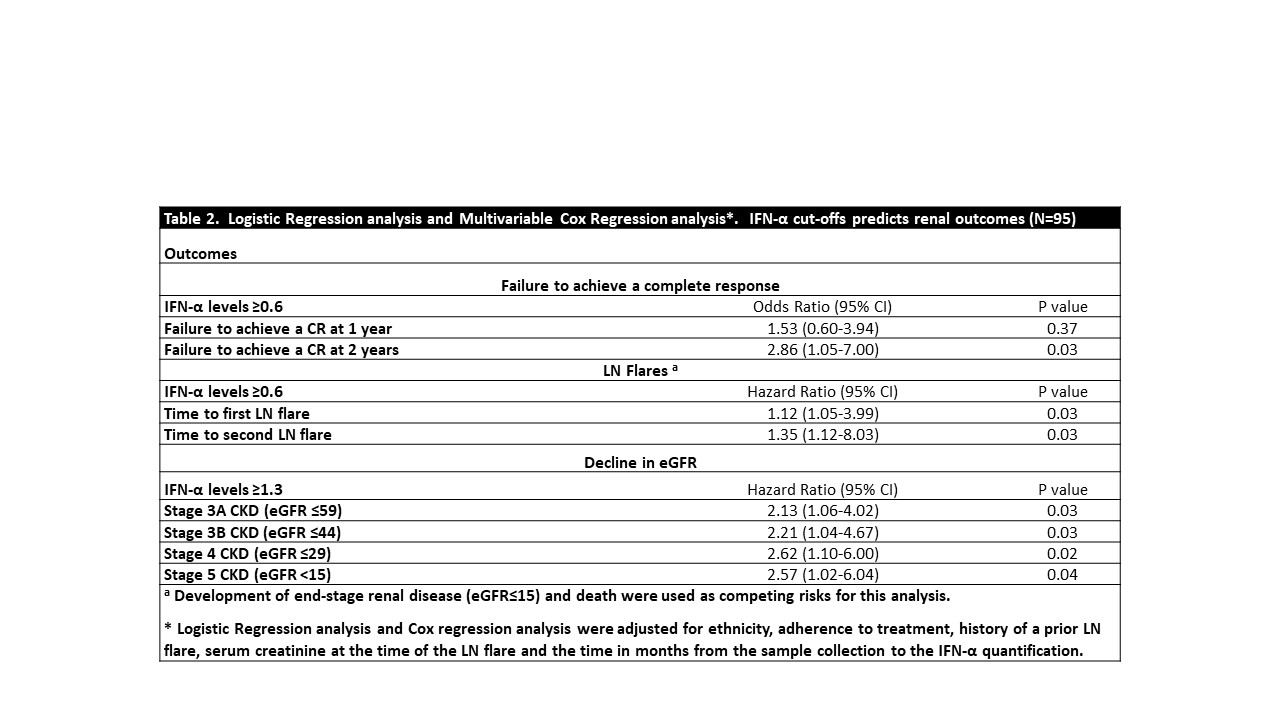
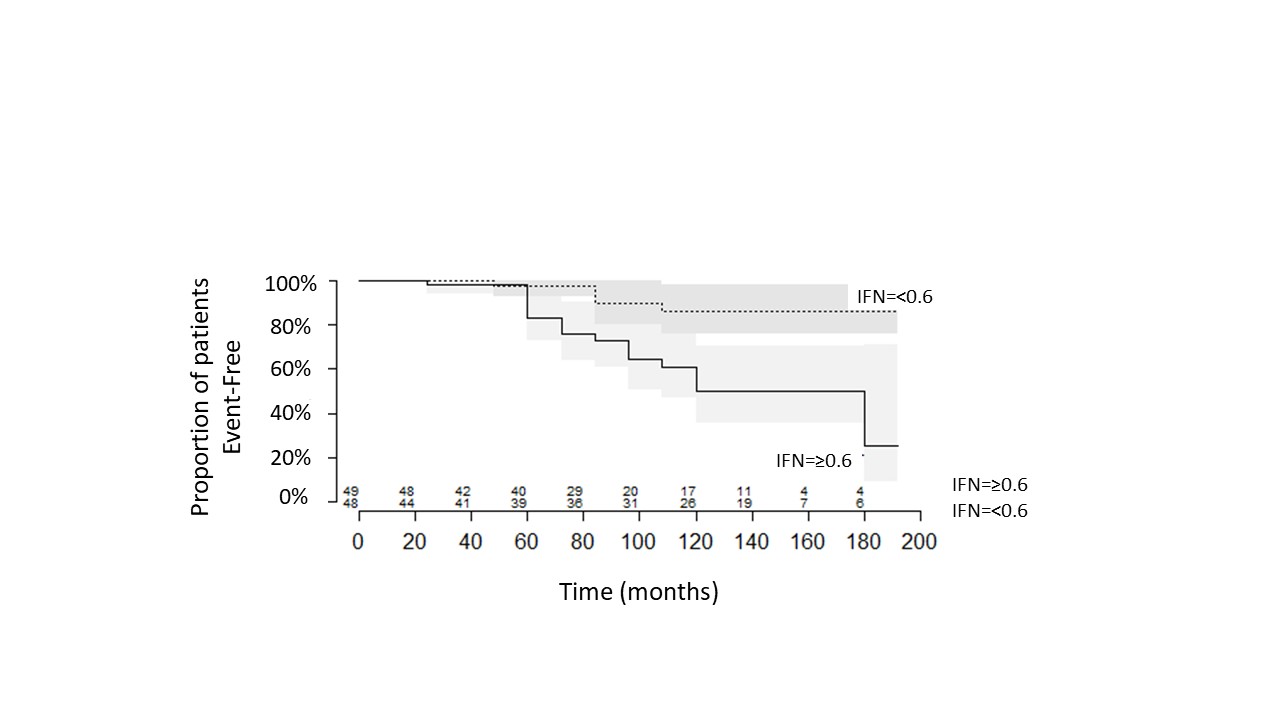
Figure 2. Time to ≥2 LN flares or end-stage renal disease (eGFR ≤15). Kaplan-Meier survival curves stratified by IFN-α levels ≥ 0.6 and <0.6, P=0.04.
L. Whittall Garcia: None; D. Gladman: AbbVie, 2, 5, Amgen, 2, 5, Bristol Myers Squibb, 2, Celgene, 2, 5, Eli Lilly, 2, 5, Galapagos, 2, Gilead Sciences, 2, Janssen, 2, 5, Novartis, 2, 5, Pfizer Inc, 2, 5, UCB, 2, 5; M. Urowitz: None; Z. Touma: AstraZeneca, 2, GSK, 2; J. Wither: AstraZeneca, 1, 6, Pfizer, 12, Indirect salary support through a Chair award to the Division of Rheumatology at the University of Toronto.
Background/Purpose: Type I Interferons (IFN-I) play a role in SLE and Lupus Nephritis (LN) pathogenesis. We have recently shown that IFN-I gene expression predicts the risk of SLE flares and a more severe disease course in SLE patients. Furthermore, recently it has been suggested that patients with higher IFN-I gene expression in their renal tubular cells are less likely to respond to conventional therapy.
The aim of this study was to determine if the amount of IFN-α in serum at the time of an LN flare predicts response to therapy, subsequent LN flares, and decline in kidney function.
Methods: Patients with active LN were included in this study. All patients had 1) active LN, defined as a 24H urine protein >500mg/day with a subsequent modification in therapy by the treating physician, 2) stored serum sample ±3 months from the renal flare and 3) baseline estimated glomerular filtration rate (eGFR) ≥60ml/min (3 months prior to the flare).
The following outcomes were ascertained: 1) Complete Response (CR), defined as proteinuria < 500 mg/day and a serum creatinine within 15% of the baseline at 1 and 2 years after the flare, 2) number of LN flares during follow-up (defined by an increase in proteinuria of at least 1000 mg/d if the baseline was < 500 mg/d or doubling of proteinuria in baseline was ≥500 mg/d and a change in therapy by the treating physician, and 3) decline in eGFR to ≤59ml/min, ≤44ml/min,≤29ml/min and < 15ml/min during follow-up.
Serum IFN-α was measured by Simoa®
Results: A total of 95 patients with active LN were included in the study. The median (IQR) age of the patients was 29 (23-41) years, 79 (83.7%) were women, and the disease duration was 6.0 (0.2-10.0) years. Forty (42.1%) were Caucasian, 21 (22.1%) were Afro-Caribbean, and 22 (23.1.%) were Asian. The baseline eGFR was 112 (98.7-127) ml/min, and the median (IQR) follow-up was 132 (96-156) months.76.8% had a kidney biopsy at the time of the LN flare, of whom 54.7% had a proliferative or mixed class, 17.8% class V, and 4.2% class I or II. The median (IQR) of prednisone was 40mg (30-38mg), 89 (93%) received antimalarial and all patients received immunosuppressive therapy.
The serum baseline levels of IFN-α predicted CR at 2 years from the LN flare. Furthermore, patients with higher baseline levels of IFN-α had a greater risk of having 2 or more subsequent renal flares. Every increase in 1 unit of IFN-α increased the risk of having 2 or more flares by 35%, and the risk of having a decline in kidney function during follow-up (Table 1).
Using ROC analysis, two IFN-α cut-offs were identified, 0.6 and 1.3 for predicting ≥2 LN flares and progression to eGFR ≤15, respectively. Even though the areas under the curve suggested that IFN-α is only a weak predictor (AUC=0.62 (0.51-0.74) and 0.60 (0.47-0.73) for ≥2 LN flares and progression to eGFR ≤15, respectively), these cutoffs continued to predict renal outcomes on Cox regression analysis, as seen in Figure 2 and Table 2.
Conclusion: IFN-α serum levels predicted failure to respond to treatment at year 2 after the renal flare, the development of ≥2 LN flares, and decline in kidney function during follow-up.

Table 1. Logistic Regression analysis and Multivariable Cox Regression analysis*. IFN-α predicts renal outcomes (N=95)

Table 2. Logistic Regression analysis and Multivariable Cox Regression analysis*. IFN-α cut-offs predicts renal outcomes (N=95)

Figure 2. Time to ≥2 LN flares or end-stage renal disease (eGFR ≤15). Kaplan-Meier survival curves stratified by IFN-α levels ≥ 0.6 and <0.6, P=0.04.
L. Whittall Garcia: None; D. Gladman: AbbVie, 2, 5, Amgen, 2, 5, Bristol Myers Squibb, 2, Celgene, 2, 5, Eli Lilly, 2, 5, Galapagos, 2, Gilead Sciences, 2, Janssen, 2, 5, Novartis, 2, 5, Pfizer Inc, 2, 5, UCB, 2, 5; M. Urowitz: None; Z. Touma: AstraZeneca, 2, GSK, 2; J. Wither: AstraZeneca, 1, 6, Pfizer, 12, Indirect salary support through a Chair award to the Division of Rheumatology at the University of Toronto.



