Poster Session C
Spondyloarthritis (SpA) including psoriatic arthritis (PsA)
Session: (2195–2226) Spondyloarthritis Including Psoriatic Arthritis – Diagnosis, Manifestations, & Outcomes Poster III: SpA
2225: Long-term Evolution of Spondyloarthritis Patients Included in REGISPONSER, 17 Years Later (REGISPON-3 Study)
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall

Maria Angeles Puche Larrubia, MD, PhD
Reina Sofia University Hospital
Granada, SpainDisclosure information not submitted.
Abstract Poster Presenter(s)
María Ángeles Puche Larrubia1, Lourdes Ladehesa Pineda2, María del carmen Ábalos Aguilera3, Desiree Ruiz Vilchez4, Laura Berbel Arcobé5, Xavier Juanola Roura6, Marta Arévalo Salaet7, Mireia Moreno8, Raquel Almodóvar González9, Cristina Pijoan Moratalla9, Marta Valero Expósito10, Verónica García García10, Beatriz Joven Ibáñez11, Manuel José Moreno Ramos12, Enrique Tomás Ornilla Laraudogoitia13, Alejandro Escudero Contreras14, Eduardo Collantes Estévez15 and Clementina López Medina16, 1Department of Rheumatology, Reina Sofia University Hospital, Cordoba, Spain, 2Rheumatology Department Reina Sofia Universitary Hospital, Cordoba, Spain, 3IMIBIC/Reina Sofia Hospital/University of Cordoba, Cordoba, Spain, 4Rheumatology Department, Reina Sofia University Hospital/Maimonides Biomedical Research Institute of Cordoba (IMIBIC), Córdoba, Spain, 5H.U. Bellvitge, Barcelona, Spain, 6Hospital Universitari de Bellvitge, Barcelona, Spain, 7Rheumatology Department University Hospital Parc Taulí, Sabadell, Spain, 8Parc Tauli Hospital Universitari, I3PT(UAB), Barcelona, Spain, 9Alcorcón Foundation University Hospital, Madrid, Spain, 10Ramón y Cajal University Hospital, Madrid, Spain, 11Hospital Universitario 12 de Octubre, Madrid, Spain, 12Rheumatology Department Hospital Virgen de la Arrixaca, El Palmar Murcia, Spain, 13University Hospital of Navarra, Navarra, Spain, 14Reina Sofia University Hospital, Córdoba, Spain, 15Maimonides Biomedical Research Institute of Cordoba (IMIBIC)/University of Cordoba, Cordoba, Spain, 16Rheumatology Department, Cochin Hospital; INSERM (U1153): Clinical Epidemiology and Biostatistics, University of Paris; Rheumatology Department, Reina Sofia Hospital, Cordoba / IMIBIC / University of Cordoba, Cordoba, Spain
Background/Purpose: The cross-sectional REGISPONSER study was conducted in 2004 in 31 centers from Spain, including a total of 2367 patients who met the European Spondyloarthropathy Study Group (ESSG) criteria for Spondyloarthritis (SpA). To date, the evolution/progression of SpA is not clearly defined due to the significant heterogeneity in the clinical profiles of patients and the challenges of conducting long-term prospective studies. Therefore, in this REGISPON-3 study, patients who participated in REGISPONSER from 8 out the 31 centers were reevaluated in a single visit 17 years after their initial assessment in REGISPONSER. The objective of this study is to describe, 17 years later, the confirmation or not of the initial diagnosis, clinical and radiographic evolution/progression, treatments used, and disease burden of these patients.
Methods: This is a longitudinal and multicenter study in which the REGISPONSER dataset has been combined with the REGISPON-3 data. In a single face-to-face visit, the following is carried out: a) interview with the patient; b) physical examination; c) plain X-ray of the spine, sacroiliac and hip; d) blood sample. Firstly, a descriptive analysis has been carried out to evaluate the change in the diagnosis of the patients 17 years later. The different treatments used in REGISPONSER and subsequently in REGISPON-3 have been described. Among the deceased patients, the different causes of death and diagnoses have been analysed. Finally, the average age of death has been calculated. The study was carried out in accordance with the Good Clinical Practice guidelines and after approval of the Ethics Committee in each center.
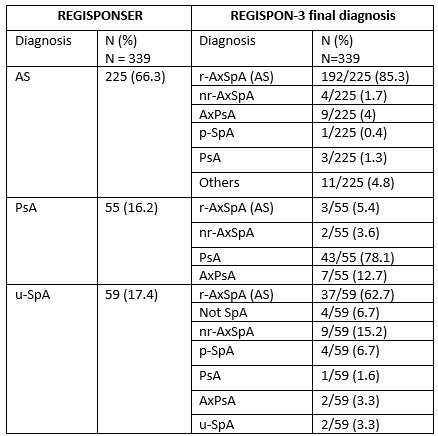
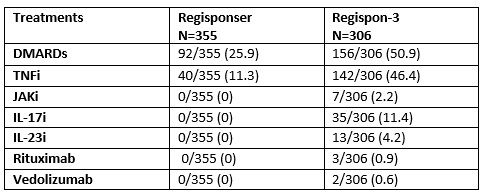
Results: From the total patients evaluated in REGISPONSER in the 8 participating centers (n=1151), we were able to recruit 437 patients (37.9%), of whom 339 were alive and 83 deceased. Table 1 shows the description of the REGISPONSER diagnoses and how they have changed 17 years later. Interestingly, among the 59 patients with undifferentiated SpA, 37 developed r-axSpA, 9 nr-axSpA and 4 finally did not have SpA. Table 2 shows the treatments used in REGISPONSER and later in REGISPON-3. In general, the use of biologics increased after 17 years in the REGISPON-3 study. Among the 83 patients who died, the most frequent causes were infection (29.3%), cardiovascular (CV) events (24%), and cancer (22.7%). The mean age of death was 72.6 (10.1) years. The most frequent diagnoses of deceased patients were axial SpA (AxSpA) (69.1%), psoriatic arthritis (PsA) (19.8%) and axial PsA (AxPsA) (4.9%).
Conclusion: This study shows the preliminary results of the REGISPON-3 project. It has been observed that over the years some diagnoses changed and there are even patients whose diagnosis is not confirmed. The treatments have also evolved with a greater use and a greater variety of biological treatments, which has allowed to improve the prognosis of the disease. Finally, the causes of death in SpA have been studied, being infection, CV events and cancer the most frequent causes.
Funding: This work was supported by grants from the Spanish Institute of Health Carlos III (PMP21/00119 and RICOR-RD21/0002/0033).
M. Puche Larrubia: None; L. Ladehesa Pineda: None; M. Ábalos Aguilera: None; D. Ruiz Vilchez: None; L. Berbel Arcobé: None; X. Juanola Roura: None; M. Arévalo Salaet: None; M. Moreno: None; R. Almodóvar González: None; C. Pijoan Moratalla: None; M. Valero Expósito: None; V. García García: None; B. Joven Ibáñez: None; M. Moreno Ramos: None; E. Ornilla Laraudogoitia: None; A. Escudero Contreras: None; E. Collantes Estévez: None; C. López Medina: AbbVie, 5, 6, Eli Lilly, 2, 5, 6, Janssen, 6, MSD, 6, Novartis, 2, 5, 6, UCB Pharma, 2, 5, 6.
Background/Purpose: The cross-sectional REGISPONSER study was conducted in 2004 in 31 centers from Spain, including a total of 2367 patients who met the European Spondyloarthropathy Study Group (ESSG) criteria for Spondyloarthritis (SpA). To date, the evolution/progression of SpA is not clearly defined due to the significant heterogeneity in the clinical profiles of patients and the challenges of conducting long-term prospective studies. Therefore, in this REGISPON-3 study, patients who participated in REGISPONSER from 8 out the 31 centers were reevaluated in a single visit 17 years after their initial assessment in REGISPONSER. The objective of this study is to describe, 17 years later, the confirmation or not of the initial diagnosis, clinical and radiographic evolution/progression, treatments used, and disease burden of these patients.
Methods: This is a longitudinal and multicenter study in which the REGISPONSER dataset has been combined with the REGISPON-3 data. In a single face-to-face visit, the following is carried out: a) interview with the patient; b) physical examination; c) plain X-ray of the spine, sacroiliac and hip; d) blood sample. Firstly, a descriptive analysis has been carried out to evaluate the change in the diagnosis of the patients 17 years later. The different treatments used in REGISPONSER and subsequently in REGISPON-3 have been described. Among the deceased patients, the different causes of death and diagnoses have been analysed. Finally, the average age of death has been calculated. The study was carried out in accordance with the Good Clinical Practice guidelines and after approval of the Ethics Committee in each center.
Results: From the total patients evaluated in REGISPONSER in the 8 participating centers (n=1151), we were able to recruit 437 patients (37.9%), of whom 339 were alive and 83 deceased. Table 1 shows the description of the REGISPONSER diagnoses and how they have changed 17 years later. Interestingly, among the 59 patients with undifferentiated SpA, 37 developed r-axSpA, 9 nr-axSpA and 4 finally did not have SpA. Table 2 shows the treatments used in REGISPONSER and later in REGISPON-3. In general, the use of biologics increased after 17 years in the REGISPON-3 study. Among the 83 patients who died, the most frequent causes were infection (29.3%), cardiovascular (CV) events (24%), and cancer (22.7%). The mean age of death was 72.6 (10.1) years. The most frequent diagnoses of deceased patients were axial SpA (AxSpA) (69.1%), psoriatic arthritis (PsA) (19.8%) and axial PsA (AxPsA) (4.9%).
Conclusion: This study shows the preliminary results of the REGISPON-3 project. It has been observed that over the years some diagnoses changed and there are even patients whose diagnosis is not confirmed. The treatments have also evolved with a greater use and a greater variety of biological treatments, which has allowed to improve the prognosis of the disease. Finally, the causes of death in SpA have been studied, being infection, CV events and cancer the most frequent causes.
Funding: This work was supported by grants from the Spanish Institute of Health Carlos III (PMP21/00119 and RICOR-RD21/0002/0033).

Table 1: Description of the evolution of the diagnosis of spondyloarthritis over 17 years from REGISPONSER I-II to REGISPON-3

Table 2: Description of the different treatments used in REGISPONSER y REGISPON-3
M. Puche Larrubia: None; L. Ladehesa Pineda: None; M. Ábalos Aguilera: None; D. Ruiz Vilchez: None; L. Berbel Arcobé: None; X. Juanola Roura: None; M. Arévalo Salaet: None; M. Moreno: None; R. Almodóvar González: None; C. Pijoan Moratalla: None; M. Valero Expósito: None; V. García García: None; B. Joven Ibáñez: None; M. Moreno Ramos: None; E. Ornilla Laraudogoitia: None; A. Escudero Contreras: None; E. Collantes Estévez: None; C. López Medina: AbbVie, 5, 6, Eli Lilly, 2, 5, 6, Janssen, 6, MSD, 6, Novartis, 2, 5, 6, UCB Pharma, 2, 5, 6.



