Poster Session B
Spondyloarthritis (SpA) including psoriatic arthritis (PsA)
Session: (1412–1441) Spondyloarthritis Including Psoriatic Arthritis – Treatment Poster II: SpA
1417: Do Long-term Patient-reported Outcomes Improve Similarly in Psoriatic Arthritis and Axial Spondyloarthritis Patients Treated with Secukinumab? Results from the EuroSpA Collaboration
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- MP
Marion Pons, MD
Rigshospitalet, Valdemar Hansens Vej 17, 2600 Glostrup, Denmark.
Glostrup, DenmarkDisclosure information not submitted.
Abstract Poster Presenter(s)
Marion Pons1, Simon Horskjær Rasmussen1, Sara Nysom Christiansen1, Brigitte Michelsen2, Bente Glintborg1, Bjorn Gudbjornsson3, Gerdur Maria Grondal4, Jiri Vencovsky5, Anne Gitte Loft6, Ziga Rotar7, Katja Perdan Pirkmajer8, Michael Nissen9, Burkhard Moeller10, Gary Macfarlane11, Gareth T. Jones11, Florenzo Iannone12, Roberto F. Caporali13, Karin Laas14, Sigrid Vorobjov15, Daniela Di Giuseppe16, Belkis Nihan Coskun17, Burcu Yagız17, Sella Provan18, Karen Fagerli18, Isabel Castrejon19, Lucia Otero-Valera20, Marleen van de Sande21, Irene van der Horst-Bruinsma22, Dan Nordstrom23, Laura Kuusalo24, Elsa Vieira-Sousa25, Miguel Bernardes26, Tor Olofsson27, Jana Baranová28, Merete Hetland1, Mikkel Østergaard29 and Lykke Ørnbjerg1, 1Copenhagen Center for Arthritis Research, Rigshospitalet, Copenhagen, Denmark, 2Center for Treatment of Rheumatic and Musculoskeletal Diseases (REMEDY), Diakonhjemmet Hospital, Oslo, Norway, 3Centre for Rheumatology Research, University Hospital, Reykjavik, Iceland, 4Department for Rheumatology, Landspitali University Hospital, Reykjavik, Iceland, 5Institute of Rheumatology, Prague, Czech Republic, Department of Rheumatology, 1st Faculty of Medicine, Charles University, Prague, Czech Republic, 6Aarhus University, Horsens, Denmark, 7University Medical Centre Ljubljana, Ljubljana, Slovenia, 8University Medical Centre Ljubljana; University of Ljubljana, Ljubljana, Slovenia, 9Geneva University Hospitals, Geneva, Switzerland, 10Inselspital - University Hospital Bern, Bern, Switzerland, 11Aberdeen Centre for Arthritis and Musculoskeletal Health (Epidemiology Group), University of Aberdeen, Aberdeen, United Kingdom, 12Rheumatology Unit, Department of Precision and Regenerative Medicine and Ionian Area, University of Bari "Aldo Moro", Bari, Italy, 13Department of Clinical Sciences and Community Health, University of Milan, and Department of Rheumatology and Medical Sciences, ASST Gaetano Pini-CTO, Milano, Italy, 14Department of Rheumatology, East-Tallinn Central Hospital, Tallinn, Estonia, 15National Institute for Health Development, Tallinn, Estonia, 16Karolinska Institutet, Stockholm, Sweden, 17Department of Rheumatology, Bursa Uludag University, Bursa, Turkey, 18Diakonhjemmet Hospital, Oslo, Norway, 19Hospital Universitario Gregorio Marañón, Madrid, Spain, 20Spanish Society of Rheumatology, Madrid, Spain, 21Amsterdam UMC, University of Amsterdam, Department of Rheumatology & Clinical Immunology and Department of Experimental Immunology, Amsterdam Infection & Immunity Institute; Amsterdam Rheumatology & Immunology Center (ARC), Academic Medical Center, Amsterdam, Netherlands, 22Rheumatology, Radboud University Medical Center, Nijmegen, Netherlands, 23Helsinki University Hospital, Helsinki, Finland, 24Centre for Rheumatology and Clinical Immunology, University of Turku and Turku University Hospital, Turku, Finland, 25Faculdade de Medicina da Universidade de Lisboa, Centro Académico de Medicina de Lisboa, Lisboa, Portugal, 26Rheumatology Department, Centro Hospitalar e Universitário de São João, Porto, Portugal, 27Lund University and Skåne University Hospital, Lund, Sweden, 28Institute of Biostatistics and Analyses, Brno, Czech Republic, 29Copenhagen Center for Arthritis Research, Center for Rheumatology and Spine Diseases, Centre for Head and Orthopaedics, Rigshospitalet; University of Copenhagen, Copenhagen, Denmark
Background/Purpose: Patient reported outcomes (PROs) are important in the treatment evaluation of patients with spondyloarthritis, including axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA). For secukinumab, limited real world data on PROs is available – and no comparison between axSpA and PsA patients has been performed. In European patients with axSpA and PsA who initiated secukinumab, we aimed to determine: 1) the proportion of patients achieving 6-, 12- and 24-month pain, fatigue, patient global assessment (PGA) and health assessment questionnaire (HAQ) remission, and 2) the 24-month secukinumab retention rate.
Methods: The study was conducted within the European Spondyloarthritis Research Collaboration Network (EuroSpA)(1). From 16 European registries, patients with axSpA or PsA who initiated secukinumab between 2015 and 2021 were included. Based on the ASAS working group definition of partial remission in axSpA(2), we applied the following definitions of PRO remission: pain≤20, PGA≤20, fatigue≤20 (all on visual analogue scales 0-100 mm) and HAQ≤0.5 for both axSpA and PsA patients, to make comparisons feasible.PRO remission rates were calculated as crude and adjusted for secukinumab adherence (LUNDEX). Comparisons of remission rates were performed with univariable and multivariable (baseline covariates: age, gender, registry, and number of previous b/tsDMARDs)logistic regression analyses. Kaplan-Meier with log-rank test and Cox regression analyses were performed to assess and compare the 24-month secukinumab retention rate between axSpA and PsA patients.
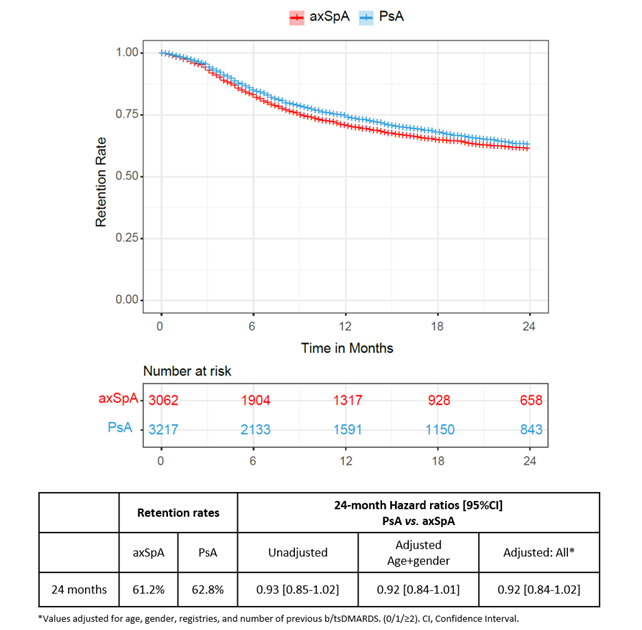
Results: We included 3062 axSpA patients and 3217 PsA patients initiating secukinumab in routine care. At secukinumab treatment start (baseline), axSpA patients had a mean (SD) age of 46.8 (12.0) years, with 52.7% being male. PsA patients had a mean age (SD) of 51.9 (11.9) years and were predominantly female (56.9%). No clinically relevant differences in disease duration, physician global assessment (PhGA), number of previous b/tsDMARDs or PROs values were found at baseline (Table 1). The reductions in pain, fatigue, and PGA values from baseline to 24-month follow-up were greater in axSpA patients compared to PsA patients. While crude PRO remission rates (pain, fatigue, PGA and HAQ) were higher for axSpA than for PsA patients at all timepoints, LUNDEX-adjusted remission rates were similar (Table 2). After correction for multiple confounders no difference was found between the groups, except for a higher remission rate for fatigue in PsA at 24 months (OR= 1.8 [95% CI 1.1-3.0]) (Table 2). The 24-month retention rates were similar in axSpA and PsA (61.2% vs. 62.8%, HR= 0.92 [95% CI 0.84-1.02], p=0.14 in fully adjusted analyses) (Figure 1).
Conclusion: Our study supports the real-world effectiveness of secukinumab in both axSpA and PsA, as measured by PROs, and suggests that PROs (including pain, PGA and HAQ) and 24-month retention rate after secukinumab initiation are similar in axSpA and PsA patients. Further analyses, aiming to evaluate long-term effectiveness of axSpA and PsA are needed.
References
1. Anon. https://eurospa.eu/.
2. Anderson JJ et al. Arthritis and Rheumatism 2001;44:1876–1886.
.gif)
.gif)
M. Pons: Novartis, 5; S. Horskjær Rasmussen: Novartis, 5; S. Nysom Christiansen: Novartis, 5, 6; B. Michelsen: Novartis, 5; B. Glintborg: AbbVie/Abbott, 5, Pfizer, 5, Sandoz, 5; B. Gudbjornsson: Nordic-Pharma, 6, Novartis, 2, 6; G. Grondal: None; J. Vencovsky: Argenx, 2, Eli Lilly, 6, Galapagos, 2, Horizon, 2, Merck, 2; A. Loft: Ucb, 1, 6, 12, Congress participation; Z. Rotar: None; K. Perdan Pirkmajer: None; M. Nissen: AbbVie/Abbott, 2, Eli Lilly, 2, 12, Involved in Clinical Trial, Janssen, 2, Novartis, 6, 12, research funding paid to institution, Pfizer, 6, UCB, 2, 12, funding support to attend EULAR 2023, paid to institution; B. Moeller: None; G. Macfarlane: None; G. Jones: Amgen, 5; F. Iannone: Abbvie, 2, 5, BMS, 2, 5, Janssen, 2, 5, Lilly, 2, 5, MSD, 2, 5, Novartis, 2, 5, Pfizer, 2, 5, Roche, 2, 5, UCB, 2, 5; R. Caporali: AbbVie, 2, 6, Amgen, 2, 6, BMS, 2, 6, Celltrion, 2, 6, Fresenius Kabi, 2, Galapagos, 2, 6, Janssen, 2, 6, Lilly, 2, 6, MSD, 2, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 2, Sandoz, 2, 6, UCB, 2, 6; K. Laas: None; S. Vorobjov: None; D. Di Giuseppe: None; B. Nihan Coskun: None; B. Yagız: None; S. Provan: None; K. Fagerli: None; I. Castrejon: Bristol Myers Squibb, 1, 6, Galapagos, 2, GlaxoSmithKline, 1, 6, Lilly, 1, 6, Merck Sharp & Dohme, 6, Pfizer, 1, 2, 6; L. Otero-Valera: None; M. van de Sande: AbbVie, 2, Eli Lilly, 5, Janssen, 6, Novartis, 2, 5, 6, UCB Pharma, 2, 5, 6; I. van der Horst-Bruinsma: Abbvie, 2, 5, 5, Lilly, 2, MSD, 2, 5, Novartis, 2, Pfizer, 5, UCB, 2, 5, 6; D. Nordstrom: AbbVie/Abbott, 2, BMS, 2, Lilly, 2, MSD, 2, Novartis, 2, Pfizer, 2, UCB, 2; L. Kuusalo: None; E. Vieira-Sousa: Abbvie, 6, Celgene, 6, MSD, 5, 6, Novartis, 6, Pfizer, 5, UCB, 5, 6; M. Bernardes: None; T. Olofsson: Merck Sharp & Dohme, 2, UCB Pharma, 2; J. Baranová: None; M. Hetland: AbbVie/Abbott, 1, 5, Bristol-Myers Squibb(BMS), 5, Danbio, 12, Chari of Danbio registry, Eli Lilly, 5, MEDAC, 6, Novartis, 5, Pfizer, 5, 6, Sandoz, 5, 6; M. Østergaard: AbbVie, 2, 5, 6, Amgen, 5, Boehringer-Ingelheim, 2, 6, Bristol-Myers Squibb(BMS), 2, 5, 6, Celgene, 2, 5, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Gilead, 2, 6, Hospira, 2, 6, Janssen, 2, 6, MEDAC, 6, Merck, 2, 5, 6, Novartis, 2, 5, 6, Novo Nordisk, 2, 6, Orion, 2, 6, Pfizer, 2, 6, Regeneron, 2, 6, Roche, 2, 6, Sandoz, 2, 6, Sanofi, 2, 6, UCB, 2, 6; L. Ørnbjerg: Novartis, 5.
Background/Purpose: Patient reported outcomes (PROs) are important in the treatment evaluation of patients with spondyloarthritis, including axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA). For secukinumab, limited real world data on PROs is available – and no comparison between axSpA and PsA patients has been performed. In European patients with axSpA and PsA who initiated secukinumab, we aimed to determine: 1) the proportion of patients achieving 6-, 12- and 24-month pain, fatigue, patient global assessment (PGA) and health assessment questionnaire (HAQ) remission, and 2) the 24-month secukinumab retention rate.
Methods: The study was conducted within the European Spondyloarthritis Research Collaboration Network (EuroSpA)(1). From 16 European registries, patients with axSpA or PsA who initiated secukinumab between 2015 and 2021 were included. Based on the ASAS working group definition of partial remission in axSpA(2), we applied the following definitions of PRO remission: pain≤20, PGA≤20, fatigue≤20 (all on visual analogue scales 0-100 mm) and HAQ≤0.5 for both axSpA and PsA patients, to make comparisons feasible.PRO remission rates were calculated as crude and adjusted for secukinumab adherence (LUNDEX). Comparisons of remission rates were performed with univariable and multivariable (baseline covariates: age, gender, registry, and number of previous b/tsDMARDs)logistic regression analyses. Kaplan-Meier with log-rank test and Cox regression analyses were performed to assess and compare the 24-month secukinumab retention rate between axSpA and PsA patients.
Results: We included 3062 axSpA patients and 3217 PsA patients initiating secukinumab in routine care. At secukinumab treatment start (baseline), axSpA patients had a mean (SD) age of 46.8 (12.0) years, with 52.7% being male. PsA patients had a mean age (SD) of 51.9 (11.9) years and were predominantly female (56.9%). No clinically relevant differences in disease duration, physician global assessment (PhGA), number of previous b/tsDMARDs or PROs values were found at baseline (Table 1). The reductions in pain, fatigue, and PGA values from baseline to 24-month follow-up were greater in axSpA patients compared to PsA patients. While crude PRO remission rates (pain, fatigue, PGA and HAQ) were higher for axSpA than for PsA patients at all timepoints, LUNDEX-adjusted remission rates were similar (Table 2). After correction for multiple confounders no difference was found between the groups, except for a higher remission rate for fatigue in PsA at 24 months (OR= 1.8 [95% CI 1.1-3.0]) (Table 2). The 24-month retention rates were similar in axSpA and PsA (61.2% vs. 62.8%, HR= 0.92 [95% CI 0.84-1.02], p=0.14 in fully adjusted analyses) (Figure 1).
Conclusion: Our study supports the real-world effectiveness of secukinumab in both axSpA and PsA, as measured by PROs, and suggests that PROs (including pain, PGA and HAQ) and 24-month retention rate after secukinumab initiation are similar in axSpA and PsA patients. Further analyses, aiming to evaluate long-term effectiveness of axSpA and PsA are needed.
References
1. Anon. https://eurospa.eu/.
2. Anderson JJ et al. Arthritis and Rheumatism 2001;44:1876–1886.
.gif)
Table 1. Baseline characteristics of axSpA and PsA patients
.gif)
Table 2. PROs and PRO remission rates 6, 12 and 24 months after secukinumab initiation in European axSpA and PsA patients.

Figure 1. Secukinumab retention rates in axSpA and PsA patients, with unadjusted and adjusted hazard ratios
M. Pons: Novartis, 5; S. Horskjær Rasmussen: Novartis, 5; S. Nysom Christiansen: Novartis, 5, 6; B. Michelsen: Novartis, 5; B. Glintborg: AbbVie/Abbott, 5, Pfizer, 5, Sandoz, 5; B. Gudbjornsson: Nordic-Pharma, 6, Novartis, 2, 6; G. Grondal: None; J. Vencovsky: Argenx, 2, Eli Lilly, 6, Galapagos, 2, Horizon, 2, Merck, 2; A. Loft: Ucb, 1, 6, 12, Congress participation; Z. Rotar: None; K. Perdan Pirkmajer: None; M. Nissen: AbbVie/Abbott, 2, Eli Lilly, 2, 12, Involved in Clinical Trial, Janssen, 2, Novartis, 6, 12, research funding paid to institution, Pfizer, 6, UCB, 2, 12, funding support to attend EULAR 2023, paid to institution; B. Moeller: None; G. Macfarlane: None; G. Jones: Amgen, 5; F. Iannone: Abbvie, 2, 5, BMS, 2, 5, Janssen, 2, 5, Lilly, 2, 5, MSD, 2, 5, Novartis, 2, 5, Pfizer, 2, 5, Roche, 2, 5, UCB, 2, 5; R. Caporali: AbbVie, 2, 6, Amgen, 2, 6, BMS, 2, 6, Celltrion, 2, 6, Fresenius Kabi, 2, Galapagos, 2, 6, Janssen, 2, 6, Lilly, 2, 6, MSD, 2, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 2, Sandoz, 2, 6, UCB, 2, 6; K. Laas: None; S. Vorobjov: None; D. Di Giuseppe: None; B. Nihan Coskun: None; B. Yagız: None; S. Provan: None; K. Fagerli: None; I. Castrejon: Bristol Myers Squibb, 1, 6, Galapagos, 2, GlaxoSmithKline, 1, 6, Lilly, 1, 6, Merck Sharp & Dohme, 6, Pfizer, 1, 2, 6; L. Otero-Valera: None; M. van de Sande: AbbVie, 2, Eli Lilly, 5, Janssen, 6, Novartis, 2, 5, 6, UCB Pharma, 2, 5, 6; I. van der Horst-Bruinsma: Abbvie, 2, 5, 5, Lilly, 2, MSD, 2, 5, Novartis, 2, Pfizer, 5, UCB, 2, 5, 6; D. Nordstrom: AbbVie/Abbott, 2, BMS, 2, Lilly, 2, MSD, 2, Novartis, 2, Pfizer, 2, UCB, 2; L. Kuusalo: None; E. Vieira-Sousa: Abbvie, 6, Celgene, 6, MSD, 5, 6, Novartis, 6, Pfizer, 5, UCB, 5, 6; M. Bernardes: None; T. Olofsson: Merck Sharp & Dohme, 2, UCB Pharma, 2; J. Baranová: None; M. Hetland: AbbVie/Abbott, 1, 5, Bristol-Myers Squibb(BMS), 5, Danbio, 12, Chari of Danbio registry, Eli Lilly, 5, MEDAC, 6, Novartis, 5, Pfizer, 5, 6, Sandoz, 5, 6; M. Østergaard: AbbVie, 2, 5, 6, Amgen, 5, Boehringer-Ingelheim, 2, 6, Bristol-Myers Squibb(BMS), 2, 5, 6, Celgene, 2, 5, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Gilead, 2, 6, Hospira, 2, 6, Janssen, 2, 6, MEDAC, 6, Merck, 2, 5, 6, Novartis, 2, 5, 6, Novo Nordisk, 2, 6, Orion, 2, 6, Pfizer, 2, 6, Regeneron, 2, 6, Roche, 2, 6, Sandoz, 2, 6, Sanofi, 2, 6, UCB, 2, 6; L. Ørnbjerg: Novartis, 5.



