Poster Session C
Vasculitis
Session: (2387–2424) Vasculitis – Non-ANCA-Associated & Related Disorders Poster III
2394: 18f-FDG-PET/CT for Giant Cell Arteritis Detection of Large Vessel Vasculitis: What Should We Take into Consideration? Analytical Study of the Arteser Registry
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- PE
Paula Valentina Estrada Alarcón, MD
H Moisès Broggi
Sant Joan Despí, Barcelona, SpainDisclosure information not submitted.
Abstract Poster Presenter(s)
Paula V. Estrada-Alarcón1, Marta Domínguez2, Rafael Benito Melero-Gonzalez3, Eugenio De Miguel4, Maria. T Silva-Diaz5, Jesús Alejandro Valero6, Ismael González7, Julio Sanchez Martin8, Javier Narvaez9, Eva Galindez-Agirregoikoa10, javier Mendizábal11, lydia Abasolo12, Javier Loricera8, Alejandro Muñoz13, Santos Castañeda14, Patricia Moya15, patricia Moran Alvarez16, Vanesa Navarro17, Carles Galisteo18, Ivette Casafont-Solé19, Jose Andres Roman Ivorra20, TAREK CARLOS SALMAN MONTE21, Margarida Rocha22, Carlota Laura Iñiguez23, Alicia García24, Cristina Campos25, María Alcalde26, Antonio Juan Mas27, Francisco Javier Prado28, Ricardo Blanco29 and On Behalf of ARTESER Working Group30, 1Hospital de San Juan Despí Moisès Broggi, Barcelona, Spain, 2Sociedad Española de Reumatología, Madrid, Spain, 3CHU Vigo, O Carballino, Spain, 4Hospital Universitario La Paz, Madrid, Spain, 5Complexo Hospitalario Universitario A Coruña, A Coruña, Spain, 6Department of Rheumatology, Hospital Universitario Donostia, San Sebastián, Spain, 7Hospital Universitario de León, León, Spain, 8Hospital Universitario Marqués de Valdecilla, Santander, Spain, 9Hospital Universitario de Bellvitge, Barcelona, Spain, 10Basurto University Hospital, Bilbao, Spain, 11Complejo Hospitalario de Navarra, Pamplona, Spain, 12Hospital Clínico San Carlos, Madrid, Spain, 13Hospital universitario Virgen del Rocío, El Viso de Alcor, Spain, 14Hospital Universitario de la Princesa, Madrid, Spain, 15Hospital de Santa Creu i Sant Pau, Barcelona, Spain, 16Hospital Ramón y Cajal, Madrid, Spain, 17H Moisès Broggi, Sant Joan Despí, Barcelona, Spain, 18Hospital Universitario Parc Taulí, Sabadell, Spain, 19Hospital Germans Trias i Pujol, Badalona, Spain, 20Hospital Universitari i Politècnic la Fe, Valencia, Spain, 21Hospital del Mar/Parc de Salut Mar-IMIM, Barcelona, Spain, 22Osakidetza, Bilbo, Spain, 23Hospital Universitario Lucus Augusti, Lugo, Spain, 24Hospital Universitario de Canarias, Islas Canarias, 25Rheumatology Unit, Hospital General Universitario de Valencia, Valencia, Spain, 26Hospital Severo Ochoa, Madrid, Spain, 27Hospital Universitario Son Llàtzer, Mallorca, Spain, 28Research department Hospital Infantil de México Federico Gómez, Mexico City, Mexico, 29Hospital Universitario Marqués de Valdecilla, IDIVAL, Santander, Spain, 30SER, Spain
Background/Purpose: Imaging studies in patients with giant cell arteritis (GCA) and suspected large vessel vasculitis (LVV) are sensitive, increasingly available, and less aggressive than temporal artery biopsy. Positron emission tomography/computed tomography (PET/CT) with 18-fluorodeoxyglucose (18F-FDG) allows for the evaluation of mural inflammation in extracranial arteries and supports the diagnosis of LVV. This study aimed to analyze in which patients with GCA the use of 18F-FDG-PET/CT is useful for detecting LVV.
Methods: ARTESER is a large Spanish registry promoted by the Spanish Society of Rheumatology, including patients with a new diagnosis of GCA, from June 2013 to March 2019. A selection of patients who underwent an 18F-FDG-PET/CT was carried out. The main variable of the study was the presence of vasculitis detected by this imaging technique. The diagnosis of LVV was based on expert opinion. Demographic variables, comorbidities, acute phase reactants, cumulative dose of glucocorticoids prior to the test, and the number of days elapsed before performing the test were considered. The clinical phenotypes were defined as cranial or extracranial, depending on the main symptom at the moment of diagnosis. Results are presented as a bivariable model to analyze differences between patients with positive and negative results, a regression model to approach the association between variables in those with a positive test, and a ROC area under the curve analysis.
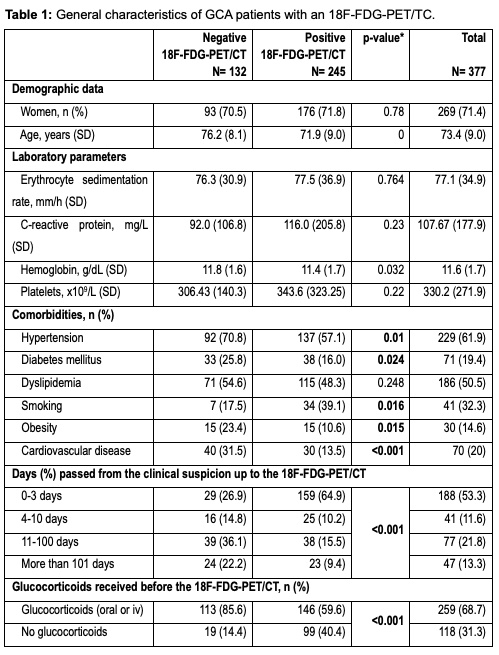
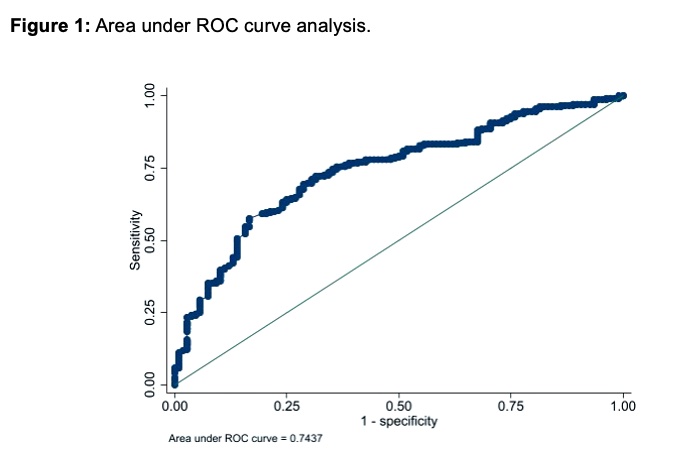
Results: From a total of 1675 GCA patients, included in ARTESER registry, 377 met the inclusion criteria of having an 18F-FDG-PET/CT done during the diagnostic process. Table 1 shows the bivariable analysis. 67.4% of patients presented as a cranial-GCA phenotype and 29.1% as extracranial-GCA variant, 18 patients could not be classified as either phenotype. Sixty-five percent of patients in our registry had LVV, as detected by 18F-FDG-PET/CT (n=245). In those patients with a positive PET/TC, the vascular territory most frequently affected was the thoracic aorta (85.7%), followed by supra-aortic vessels (78.8%), and abdominal aorta (57.6%). Cardiovascular risk factors were more frequent in patients with a negative 18F-FDG-PET/CT (Table 1). The regression model (Table 2) shows a negative association with a positive 18F-FDG-PET/CT for older patients, patients with diabetes mellitus and cardiovascular disease and, the odds ratio for having a positive 18F-FDG-PET/TC is lower, as days go by. No variables were found to have a positive association with a positive 18F-FDG-PET/TC. Depending on the cumulative dosage of glucocorticoids, 18F-FDG-PET/CT had an area under the curve of 0.74 (Figure 1).
Conclusion: Younger patients have a higher probability of presenting a LVV detected by 18F-FDG-PET/CT. Patients with cardiovascular risk factors (hypertension, diabetes mellitus, smoking), have a higher probability of a negative 18F-FDG-PET/CT. The most frequent large vessel affected was the thoracic aorta. As days go off, and the cumulative glucocorticoid dose is higher, vascular wall uptake of 18F-FDG is reduced, the reason why 18F-FDG-PET/CT should be performed as soon as possible.
.jpg)
P. Estrada-Alarcón: None; M. Domínguez: None; R. Melero-Gonzalez: None; E. De Miguel: None; M. Silva-Diaz: None; J. Valero: None; I. González: None; J. Sanchez Martin: None; J. Narvaez: None; E. Galindez-Agirregoikoa: None; j. Mendizábal: None; l. Abasolo: None; J. Loricera: None; A. Muñoz: None; S. Castañeda: None; P. Moya: None; p. Moran Alvarez: None; V. Navarro: None; C. Galisteo: None; I. Casafont-Solé: None; J. Roman Ivorra: None; T. SALMAN MONTE: None; M. Rocha: None; C. Iñiguez: None; A. García: None; C. Campos: None; M. Alcalde: None; A. Mas: None; F. Prado: None; R. Blanco: AbbVie, 5, 6, Amgen, 6, AstraZeneca, 2, BMS, 6, Eli Lilly, 6, Galapagos, 2, 6, Janssen, 2, 6, MSD, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 5, 6, Sanofi, 6; O. ARTESER Working Group: None.
Background/Purpose: Imaging studies in patients with giant cell arteritis (GCA) and suspected large vessel vasculitis (LVV) are sensitive, increasingly available, and less aggressive than temporal artery biopsy. Positron emission tomography/computed tomography (PET/CT) with 18-fluorodeoxyglucose (18F-FDG) allows for the evaluation of mural inflammation in extracranial arteries and supports the diagnosis of LVV. This study aimed to analyze in which patients with GCA the use of 18F-FDG-PET/CT is useful for detecting LVV.
Methods: ARTESER is a large Spanish registry promoted by the Spanish Society of Rheumatology, including patients with a new diagnosis of GCA, from June 2013 to March 2019. A selection of patients who underwent an 18F-FDG-PET/CT was carried out. The main variable of the study was the presence of vasculitis detected by this imaging technique. The diagnosis of LVV was based on expert opinion. Demographic variables, comorbidities, acute phase reactants, cumulative dose of glucocorticoids prior to the test, and the number of days elapsed before performing the test were considered. The clinical phenotypes were defined as cranial or extracranial, depending on the main symptom at the moment of diagnosis. Results are presented as a bivariable model to analyze differences between patients with positive and negative results, a regression model to approach the association between variables in those with a positive test, and a ROC area under the curve analysis.
Results: From a total of 1675 GCA patients, included in ARTESER registry, 377 met the inclusion criteria of having an 18F-FDG-PET/CT done during the diagnostic process. Table 1 shows the bivariable analysis. 67.4% of patients presented as a cranial-GCA phenotype and 29.1% as extracranial-GCA variant, 18 patients could not be classified as either phenotype. Sixty-five percent of patients in our registry had LVV, as detected by 18F-FDG-PET/CT (n=245). In those patients with a positive PET/TC, the vascular territory most frequently affected was the thoracic aorta (85.7%), followed by supra-aortic vessels (78.8%), and abdominal aorta (57.6%). Cardiovascular risk factors were more frequent in patients with a negative 18F-FDG-PET/CT (Table 1). The regression model (Table 2) shows a negative association with a positive 18F-FDG-PET/CT for older patients, patients with diabetes mellitus and cardiovascular disease and, the odds ratio for having a positive 18F-FDG-PET/TC is lower, as days go by. No variables were found to have a positive association with a positive 18F-FDG-PET/TC. Depending on the cumulative dosage of glucocorticoids, 18F-FDG-PET/CT had an area under the curve of 0.74 (Figure 1).
Conclusion: Younger patients have a higher probability of presenting a LVV detected by 18F-FDG-PET/CT. Patients with cardiovascular risk factors (hypertension, diabetes mellitus, smoking), have a higher probability of a negative 18F-FDG-PET/CT. The most frequent large vessel affected was the thoracic aorta. As days go off, and the cumulative glucocorticoid dose is higher, vascular wall uptake of 18F-FDG is reduced, the reason why 18F-FDG-PET/CT should be performed as soon as possible.

*p-value refers to the statistical difference between patients with the listed characteristics, to those who do not present them.
.jpg)

P. Estrada-Alarcón: None; M. Domínguez: None; R. Melero-Gonzalez: None; E. De Miguel: None; M. Silva-Diaz: None; J. Valero: None; I. González: None; J. Sanchez Martin: None; J. Narvaez: None; E. Galindez-Agirregoikoa: None; j. Mendizábal: None; l. Abasolo: None; J. Loricera: None; A. Muñoz: None; S. Castañeda: None; P. Moya: None; p. Moran Alvarez: None; V. Navarro: None; C. Galisteo: None; I. Casafont-Solé: None; J. Roman Ivorra: None; T. SALMAN MONTE: None; M. Rocha: None; C. Iñiguez: None; A. García: None; C. Campos: None; M. Alcalde: None; A. Mas: None; F. Prado: None; R. Blanco: AbbVie, 5, 6, Amgen, 6, AstraZeneca, 2, BMS, 6, Eli Lilly, 6, Galapagos, 2, 6, Janssen, 2, 6, MSD, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 5, 6, Sanofi, 6; O. ARTESER Working Group: None.



