Poster Session B
Rheumatoid arthritis (RA)
Session: (1308–1344) RA – Treatment Poster II
1338: The Advantage of Tight Control and Treat-to-Target in New-onset Rheumatoid Arthritis Patients in Daily Rheumatology Practice: Results from a Contemporary University Clinic Inception Cohort
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall

Elisabet Lindqvist, MD, PhD
Department of Rheumatology, Skane University Hospital, Lund
Lund, SwedenDisclosure information not submitted.
Abstract Poster Presenter(s)
Katarina Friberger Pajalic1, Jon Thorkel Einarsson1, Caroline Bengtsson2, Ellen Landgren3, Elisabeth Mogard4, Carmen Roseman5, Johan Karlsson Wallman4, Elisabet Lindqvist4, Tor Olofsson4 and Meliha Kapetanovic4, 1Lund University, Institution for clinical sciences, section for rheumatology Lund and Lund University Hospital, Lund, Sweden, 2Lund University Hospital, section for rheumatology, Lund, Sweden, 3Lund University, Institution for clinical sciences, section for rheumatology Lund and Skåne University Hospital, Lund, Sweden, 4Lund University and Skåne University Hospital, Lund, Sweden, 5Lund University, Institution for clinical sciences, section for rheumatology Lund and Lund University Hospital, Lund, Sweden
Background/Purpose: Since 2018, all patients with new-onset rheumatoid arthritis (RA) at the Department of Rheumatology, Skåne University Hospital, Lund, Sweden, are invited to participate in a ”tight control” and ”treat-to-target” (TC+T2T ) follow-up strategy. This strategy includes follow-up visits to a rheumatologist (at diagnosis and 3, 6, 12, 18, 24 months) plus physical/telephone consultations with a rheumatology nurse between physician visits – both with disease activity assessments and, if needed, adjustment of anti-rheumatic treatment, aiming for remission. The aim of the current study was to explore the possible advantages of implementing this TC+T2T strategy over routine care in reaching remission (DAS28 < 2.6, DAS28CRP < 2.4 or CDAI≤2.8) in clinical practice of new-onset RA patients.
Methods: RA patients with symptom duration < 24 months at diagnosis were eligible. Data on disease and treatment characteristics, as well as outcome measures during follow-up, were retrieved from the Swedish Rheumatology Quality register (SRQ). In total, 336 patients entered one of the follow-up strategies between January 1, 2018, and October 31, 2022. Of these, 193 were followed according to the TC+T2T strategy and 143 according to routine care. Percentage females/mean age/mean symptom duration at inclusion were 75%/56 years/4.5 months (TC+T2T) and 71%/62 years/7.6 months (routine care). Achievement of remission was compared between the two strategies using logistic regression, adjusted for sex, age, symptom duration, and DAS28/DAS28CRP/CDAI at diagnosis. In addition, changes in patient-reported outcomes (fatigue and pain) were assessed for the respective strategies.
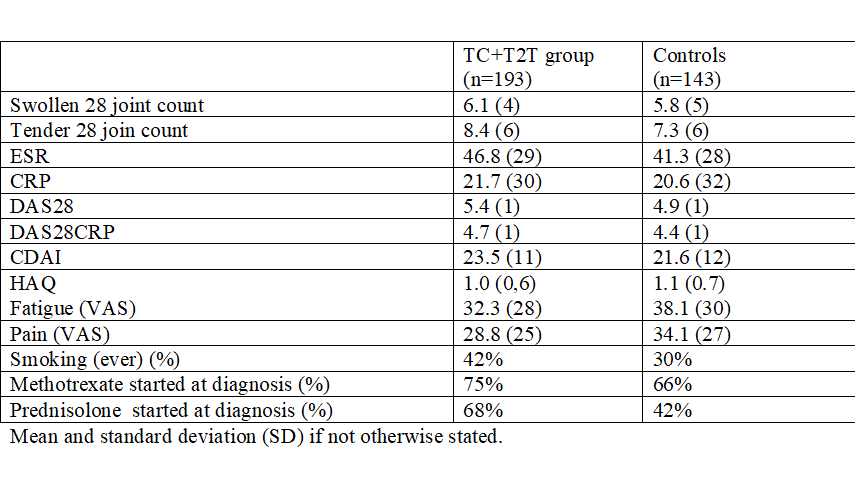
Results: Disease and treatment characteristics at diagnosis are summarized in Table1.
Percentages of patients reaching DAS28, DAS28CRP and CDAI remission criteria increased in both groups over time, but this was more pronounced in the TC+T2T group (Figure). A similar pattern was seen in the dynamics of fatigue and pain (Figure). Follow-up according to the TC+T2T was associated with a numerical higher odds ratio of achieving remission at all follow-up times regardless the remission criteria used and a significantly higher odds ratio of CDAI remission at 6 months (after adjustment for sex, age, symptom duration and CDAI at diagnosis) (Table 2).
Conclusion: Compared to routine rheumatology care, the implementation of a ”tight control” and ”treat-to-target” strategy resulted in numerically larger proportions of patients reaching remission at majority of follow-up visits over two years. Similarly, both strategies showed an early and sustained numerical improvement in patient-reported outcomes. Our results suggest that this type of strategy should be integrated into daily clinical practice of new-onset RA.
.gif)
.gif)
K. Friberger Pajalic: None; J. Einarsson: None; C. Bengtsson: None; E. Landgren: None; E. Mogard: AbbVie/Abbott, 6, Novartis, 6; C. Roseman: None; J. Karlsson Wallman: AbbVie, 5, 6, Amgen, 5, 6, Eli Lilly, 5, Novartis, 5, Pfizer, 5; E. Lindqvist: None; T. Olofsson: Merck/MSD, 2, UCB, 2; M. Kapetanovic: None.
Background/Purpose: Since 2018, all patients with new-onset rheumatoid arthritis (RA) at the Department of Rheumatology, Skåne University Hospital, Lund, Sweden, are invited to participate in a ”tight control” and ”treat-to-target” (TC+T2T ) follow-up strategy. This strategy includes follow-up visits to a rheumatologist (at diagnosis and 3, 6, 12, 18, 24 months) plus physical/telephone consultations with a rheumatology nurse between physician visits – both with disease activity assessments and, if needed, adjustment of anti-rheumatic treatment, aiming for remission. The aim of the current study was to explore the possible advantages of implementing this TC+T2T strategy over routine care in reaching remission (DAS28 < 2.6, DAS28CRP < 2.4 or CDAI≤2.8) in clinical practice of new-onset RA patients.
Methods: RA patients with symptom duration < 24 months at diagnosis were eligible. Data on disease and treatment characteristics, as well as outcome measures during follow-up, were retrieved from the Swedish Rheumatology Quality register (SRQ). In total, 336 patients entered one of the follow-up strategies between January 1, 2018, and October 31, 2022. Of these, 193 were followed according to the TC+T2T strategy and 143 according to routine care. Percentage females/mean age/mean symptom duration at inclusion were 75%/56 years/4.5 months (TC+T2T) and 71%/62 years/7.6 months (routine care). Achievement of remission was compared between the two strategies using logistic regression, adjusted for sex, age, symptom duration, and DAS28/DAS28CRP/CDAI at diagnosis. In addition, changes in patient-reported outcomes (fatigue and pain) were assessed for the respective strategies.
Results: Disease and treatment characteristics at diagnosis are summarized in Table1.
Percentages of patients reaching DAS28, DAS28CRP and CDAI remission criteria increased in both groups over time, but this was more pronounced in the TC+T2T group (Figure). A similar pattern was seen in the dynamics of fatigue and pain (Figure). Follow-up according to the TC+T2T was associated with a numerical higher odds ratio of achieving remission at all follow-up times regardless the remission criteria used and a significantly higher odds ratio of CDAI remission at 6 months (after adjustment for sex, age, symptom duration and CDAI at diagnosis) (Table 2).
Conclusion: Compared to routine rheumatology care, the implementation of a ”tight control” and ”treat-to-target” strategy resulted in numerically larger proportions of patients reaching remission at majority of follow-up visits over two years. Similarly, both strategies showed an early and sustained numerical improvement in patient-reported outcomes. Our results suggest that this type of strategy should be integrated into daily clinical practice of new-onset RA.

Disease and treatment characteristics at diagnosis
.gif)
.gif)
Achievement of remission between "treat -to-target" strategy and routine rheumatology care ( logistic regression analysis)
K. Friberger Pajalic: None; J. Einarsson: None; C. Bengtsson: None; E. Landgren: None; E. Mogard: AbbVie/Abbott, 6, Novartis, 6; C. Roseman: None; J. Karlsson Wallman: AbbVie, 5, 6, Amgen, 5, 6, Eli Lilly, 5, Novartis, 5, Pfizer, 5; E. Lindqvist: None; T. Olofsson: Merck/MSD, 2, UCB, 2; M. Kapetanovic: None.



