Poster Session B
Periodic fever syndromes, autoinflammatory diseases, Still’s disease and MAS/HLH
Session: (1124–1154) Miscellaneous Rheumatic & Inflammatory Diseases Poster II
1147: Blood-Based Biomarkers of Inflammation and Tissue Remodeling Can Discriminate Between Rheumatoid Arthritis, Psoriasis, and Psoriatic Arthritis and Are Associated with Hand Function
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- HP
Helena Port I Linares, MSc
Nordic Bioscience A/S / University of Copenhagen
Herlev, DenmarkDisclosure information not submitted.
Abstract Poster Presenter(s)
Helena Port I Linares1, Birte Coppers2, Sara Bayat2, Godonou Elie-Tino2, Larissa Valor-Mendez2, David Simon2, Filippo Fagni2, Giulia Corte2, Anne-Christine Bay-Jensen3, Koray Tascilar2, Axel Hueber4, Verena Schoenau2, Michael Sticherling5, Simon Heinrich6, Daniela Bohr2, Georg Schett7, Arnd Kleyer8, Sigrid Leyendecker6, Signe Holm Nielsen3 and Anna-Maria Liphardt9, 1Nordic Bioscience A/S / University of Copenhagen, Copenhagen, Denmark, 2Friedrich-Alexander-Universität (FAU) Erlangen-Nürnberg & Universitätsklinikum Erlangen, Department of Internal Medicine 3 - Rheumatology and Immunology / Deutsches Zentrum Immuntherapie, Erlangen, Germany, 3Nordic Bioscience, Herlev, Denmark, 4Division of Rheumatology, Klinikum Nürnberg, Paracelsus Medical University / Friedrich-Alexander-Universität (FAU) Erlangen-Nürnberg & Universitätsklinikum Erlangen, Department of Internal Medicine 3 - Rheumatology and Immunology, Nürnberg, Germany, 5Friedrich-Alexander-Universität (FAU) Erlangen-Nürnberg & Universitätsklinikum Erlangen, Department of Dermatology, Erlangen, Germany, 6Friedrich-Alexander-Universität (FAU) Erlangen-Nürnberg, Institute of Applied Dynamics, Erlangen, Germany, 7Friedrich-Alexander-Universität Erlangen-Nürnberg, Erlangen, Germany, 8University Hospital Erlangen, Erlangen, Germany, 9Friedrich-Alexander-University Erlangen-Nuremberg, Internal Medicine 3 - Rheumatology and Immunology, University Hospital Erlangen, Erlangen, Germany
Background/Purpose: Inflammatory musculoskeletal diseases including rheumatoid arthritis (RA) and psoriatic arthritis (PsA) are characterized by physical function impairment due to progressive inflammatory and structural changes. This becomes apparent even in the early disease stages before arthritis onset, such as in psoriasis (PsO). Chronic inflammation triggers an accelerated remodeling of the extracellular matrix (ECM), resulting in the release of specific degradation products of proinflammatory and collagen molecules that can be measured in blood. In this study, we explored whether levels of ECM biomarkers differ among patients with RA, PsA and PsO, and controls. In addition, we investigated the association between biomarkers and functional impairment by measures of hand function.
Methods: This is a secondary analysis of serum samples obtained from participants of three hand function studies with identical study conditions for sample collection and assessments (FAU ethics approval #125_16B, #357_20B). Serum samples from patients with RA, PsA, and PsO, and controls were obtained in the outpatient clinic of the Rheumatology and Dermatology Department, Universitätsklinikum Erlangen, Germany. Serum ECM catabolic markers (C1M, C2M, C3M, C4M, PRO-C4, C6M, ARG), formation markers (PRO-C1, PRO-C3, PRO-C6), and inflammation markers (VICM) were measured by immunoassay (Nordic Bioscience, Herlev, Denmark). Fine motor skills were assessed by the Moberg-Picking-Up Test (MPUT) and isometric grip strength was measured using a dynamometer. The Michigan Hand Questionnaire (MHQ) was used to evaluate patient-perceived hand function. The best out of two and three attempts was used for the analysis of MPUT and grip strength, respectively. Linear regression models with robust standard errors were used to compare biomarker levels between groups, adjusting for age and gender. Correlations between biomarker data and hand function were assessed by Spearman's rank correlation (ρ) and p-values were adjusted by false discovery rate method.
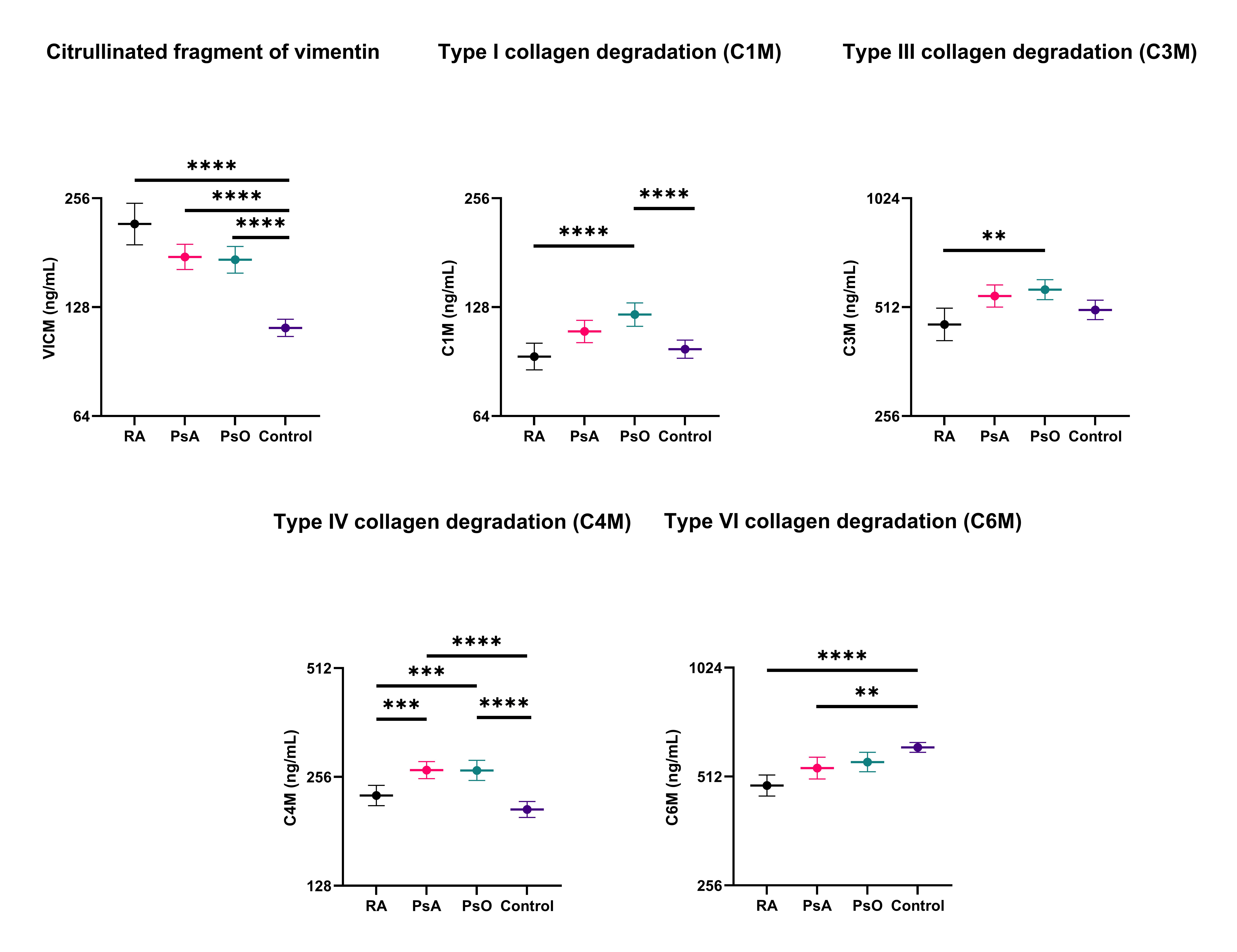
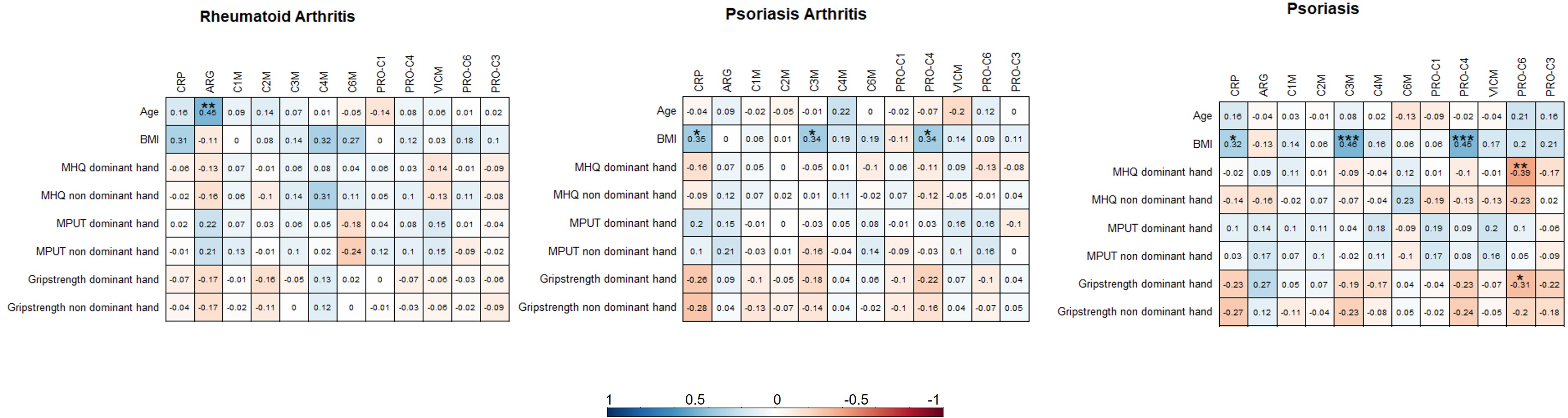
Results: 85 patients with RA, 115 with PsA, 102 with PsO and 110 controls (mean age, years: 58.4, 53.7, 45.8, 46.6; % male: 35.3, 49.6, 60.8, 45.0, respectively) were included. VICM levels were significantly higher in RA, PsA, and PsO than in controls (Figure 1, p< 0.0001). PsA and PsO showed significantly higher C4M levels compared to RA and controls, while C6M was lower in patients with RA and PsA than in controls (Figure 1, p< 0.001). C1M presented higher levels in PsO compared to controls, and together with C3M higher levels than RA (Figure 1, p< 0.004). The remaining biomarkers did not show any significant differences. Weak correlations were observed between the biomarkers and the hand function scores (all ρ<±0.2-0.3, Figure 2), while only PRO-C6 showed a significant negative correlation with MHQ (ρ=-0.39, p< 0.01) and grip strength (ρ=-0.31, p< 0.05) in patients with PsO.
Conclusion: Patients with RA, PsA, and PsO showed significant alterations in ECM remodeling biomarkers and specially PsA and PsO had higher levels of inflammatory biomarkers compared to RA and controls. Furthermore, predominantly in PsO, ECM formation biomarkers were associated with hand function impairment.
H. Port I Linares: None; B. Coppers: None; S. Bayat: EI Lilly, 5; G. Elie-Tino: None; L. Valor-Mendez: None; D. Simon: Janssen, 5; F. Fagni: Eli Lilly, 6, Galapagos, 6, Novartis, 6; G. Corte: None; A. Bay-Jensen: Nordic Bioscience, 3, 3, 8, 9; K. Tascilar: None; A. Hueber: EI Lilly, 5, 6, Janssen, 5, Novartis, 5, 6; V. Schoenau: None; M. Sticherling: EI Lilly, 5, El Lilly, 6, Janssen, 5, 6, Novartis, 5, 6; S. Heinrich: None; D. Bohr: None; G. Schett: None; A. Kleyer: None; S. Leyendecker: None; S. Holm Nielsen: Nordic Bioscience, 3, 8; A. Liphardt: Novartis Pharma GmbH, 5.
Background/Purpose: Inflammatory musculoskeletal diseases including rheumatoid arthritis (RA) and psoriatic arthritis (PsA) are characterized by physical function impairment due to progressive inflammatory and structural changes. This becomes apparent even in the early disease stages before arthritis onset, such as in psoriasis (PsO). Chronic inflammation triggers an accelerated remodeling of the extracellular matrix (ECM), resulting in the release of specific degradation products of proinflammatory and collagen molecules that can be measured in blood. In this study, we explored whether levels of ECM biomarkers differ among patients with RA, PsA and PsO, and controls. In addition, we investigated the association between biomarkers and functional impairment by measures of hand function.
Methods: This is a secondary analysis of serum samples obtained from participants of three hand function studies with identical study conditions for sample collection and assessments (FAU ethics approval #125_16B, #357_20B). Serum samples from patients with RA, PsA, and PsO, and controls were obtained in the outpatient clinic of the Rheumatology and Dermatology Department, Universitätsklinikum Erlangen, Germany. Serum ECM catabolic markers (C1M, C2M, C3M, C4M, PRO-C4, C6M, ARG), formation markers (PRO-C1, PRO-C3, PRO-C6), and inflammation markers (VICM) were measured by immunoassay (Nordic Bioscience, Herlev, Denmark). Fine motor skills were assessed by the Moberg-Picking-Up Test (MPUT) and isometric grip strength was measured using a dynamometer. The Michigan Hand Questionnaire (MHQ) was used to evaluate patient-perceived hand function. The best out of two and three attempts was used for the analysis of MPUT and grip strength, respectively. Linear regression models with robust standard errors were used to compare biomarker levels between groups, adjusting for age and gender. Correlations between biomarker data and hand function were assessed by Spearman's rank correlation (ρ) and p-values were adjusted by false discovery rate method.
Results: 85 patients with RA, 115 with PsA, 102 with PsO and 110 controls (mean age, years: 58.4, 53.7, 45.8, 46.6; % male: 35.3, 49.6, 60.8, 45.0, respectively) were included. VICM levels were significantly higher in RA, PsA, and PsO than in controls (Figure 1, p< 0.0001). PsA and PsO showed significantly higher C4M levels compared to RA and controls, while C6M was lower in patients with RA and PsA than in controls (Figure 1, p< 0.001). C1M presented higher levels in PsO compared to controls, and together with C3M higher levels than RA (Figure 1, p< 0.004). The remaining biomarkers did not show any significant differences. Weak correlations were observed between the biomarkers and the hand function scores (all ρ<±0.2-0.3, Figure 2), while only PRO-C6 showed a significant negative correlation with MHQ (ρ=-0.39, p< 0.01) and grip strength (ρ=-0.31, p< 0.05) in patients with PsO.
Conclusion: Patients with RA, PsA, and PsO showed significant alterations in ECM remodeling biomarkers and specially PsA and PsO had higher levels of inflammatory biomarkers compared to RA and controls. Furthermore, predominantly in PsO, ECM formation biomarkers were associated with hand function impairment.

Figure 1. ECM biomarkers in patients with rheumatoid arthritis (RA), psoriatic arthritis (PsA) and psoriasis (PsO), and controls. Data is shown as mean ± 95% confidential intervals.Linear regression models using robust standard errors were performed to compare biomarker levels within the groups with age and gender as covariates. P-values were adjusted by Holm correction and significance threshold was set at 0.004 considering that 12 biomarkers were analyzed. Significance is shown as ** p< 0.004, ***p< 0.001 and ****p<0.0001.

Figure 2. Spearman’s correlations between serological metabolites and clinical scores were performed. Spearman’s rho (ρ) is shown. The significance of correlations are shown as * p< 0.05, **p< 0.01, and ***p< 0.001. Abbreviations: BMI, body mass index; MHQ, Michigan Hand Questionnaire; MPUT, Moberg Picking-Up Test; CRP, C-reactive protein; ARG, aggrecan ADAMTS degradation; C1M, MMP-2/9/13-degraded type I collagen; C2M, MMP (multiple) -degraded type II collagen; C3M, MMP-9-degraded type III collagen; C4M, MMP (multiple)-degraded type IV collagen; C6M, MMP (multiple)-degraded type IV collagen; PRO-C1, Type I collagen N-terminal propeptide; PRO-C4, Type IV 7S domain collagen; VICM, citrullinated and MMP-degraded vimentin; PRO-C6, Type VI collagen, alpha-3 chain, C5 domain; PRO-C3, type II collagen N-terminal propeptide.
H. Port I Linares: None; B. Coppers: None; S. Bayat: EI Lilly, 5; G. Elie-Tino: None; L. Valor-Mendez: None; D. Simon: Janssen, 5; F. Fagni: Eli Lilly, 6, Galapagos, 6, Novartis, 6; G. Corte: None; A. Bay-Jensen: Nordic Bioscience, 3, 3, 8, 9; K. Tascilar: None; A. Hueber: EI Lilly, 5, 6, Janssen, 5, Novartis, 5, 6; V. Schoenau: None; M. Sticherling: EI Lilly, 5, El Lilly, 6, Janssen, 5, 6, Novartis, 5, 6; S. Heinrich: None; D. Bohr: None; G. Schett: None; A. Kleyer: None; S. Leyendecker: None; S. Holm Nielsen: Nordic Bioscience, 3, 8; A. Liphardt: Novartis Pharma GmbH, 5.



