Poster Session B
Crystal arthropathies
Session: (1100–1123) Metabolic & Crystal Arthropathies – Basic & Clinical Science Poster II
1121: Cardiovascular Safety of Febuxostat in Patients with Gout or Hyperuricemia: A Systematic Review of Randomized Controlled Trials
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
Abstract Poster Presenter(s)
Roba Ghossan1, Ouidade Aitisha Tabesh2, Fouad Fayad2, Pascal Richette3 and Thomas Bardin4, 1COCHIN HOSPITAL, Paris, France, 2Hotel Dieu de France, Beirut, Lebanon, 3Lariboisière Hospital, Paris, France, 4Université de Paris Cité, Paris, France
Background/Purpose: To this date, a causal relationship between febuxostat and cardiovascular disease remains controversial as comparison between trials can be challenging and may lead to misleading conclusions especially when facing heterogeneous cardiovascular outcomes. We aimed to compare the cardiovascular outcomes in the most pertinent trials of Febuxostat compared to controls.
Methods: We searched electronic databases using a PICOS-style approach search strategy of randomized controlled trials on cardiovascular outcomes of Febuxostat in patients with gout or hyperuricemia. We conducted a quality and risk of bias assessment of the included clinical trials. The definition of MACE as well as all reported cardiovascular outcomes were retrieved from every involved trial.
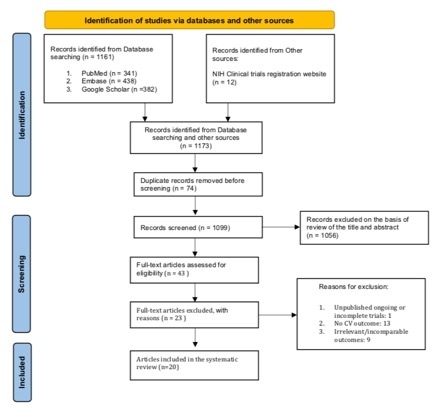
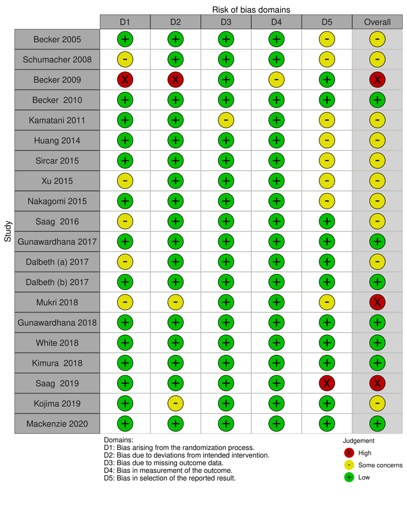
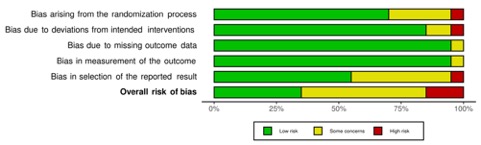
Results: Of the 1173 records identified from all sources, 20 RCTs were included in the analysis. Mean duration of follow-up was 69.7 ± 81.5 weeks and Febuxostat dose ranged from 10 to 240 mg with 80 mg being the most commonly used dosage. Overall, the quality of evidence deriving from all RCTs showed concerns in most studies (65%). Major cardiovascular event (MACE) was defined in 7 of the 20 RCTs (35%) and cardiovascular outcome reporting was very heterogeneous. Overall, data of cardiovascular safety of Febuxostat were reassuring.
Conclusion: Our systematic review showed no alarming increase of cardiovascular mortality and outcomes in Febuxostat treated patients except for the CARES trial in which the credibility of the results is biased by the high rate of drug discontinuation and of most importantly, withdrawal from follow-up. FAST trial results, backed up with real-world data cohort studies, were robust and reassuring with regard to the cardiovascular safety of Febuxostat in most white males with gout including elderly patients. This should lead the regulatory agencies to reconsider the limitations imposed on the use of Febuxostat in patients with non-severe CV burden
R. Ghossan: None; O. Aitisha Tabesh: None; F. Fayad: None; P. Richette: None; T. Bardin: None.
Background/Purpose: To this date, a causal relationship between febuxostat and cardiovascular disease remains controversial as comparison between trials can be challenging and may lead to misleading conclusions especially when facing heterogeneous cardiovascular outcomes. We aimed to compare the cardiovascular outcomes in the most pertinent trials of Febuxostat compared to controls.
Methods: We searched electronic databases using a PICOS-style approach search strategy of randomized controlled trials on cardiovascular outcomes of Febuxostat in patients with gout or hyperuricemia. We conducted a quality and risk of bias assessment of the included clinical trials. The definition of MACE as well as all reported cardiovascular outcomes were retrieved from every involved trial.
Results: Of the 1173 records identified from all sources, 20 RCTs were included in the analysis. Mean duration of follow-up was 69.7 ± 81.5 weeks and Febuxostat dose ranged from 10 to 240 mg with 80 mg being the most commonly used dosage. Overall, the quality of evidence deriving from all RCTs showed concerns in most studies (65%). Major cardiovascular event (MACE) was defined in 7 of the 20 RCTs (35%) and cardiovascular outcome reporting was very heterogeneous. Overall, data of cardiovascular safety of Febuxostat were reassuring.
Conclusion: Our systematic review showed no alarming increase of cardiovascular mortality and outcomes in Febuxostat treated patients except for the CARES trial in which the credibility of the results is biased by the high rate of drug discontinuation and of most importantly, withdrawal from follow-up. FAST trial results, backed up with real-world data cohort studies, were robust and reassuring with regard to the cardiovascular safety of Febuxostat in most white males with gout including elderly patients. This should lead the regulatory agencies to reconsider the limitations imposed on the use of Febuxostat in patients with non-severe CV burden

Figure 1: Flowchart of the included studies
NIH: National Institutes of Health; CV: Cardiovascular
NIH: National Institutes of Health; CV: Cardiovascular

Figure 2: Risk of bias of included randomized trials

Figure 2: Risk of bias of included randomized trials
R. Ghossan: None; O. Aitisha Tabesh: None; F. Fayad: None; P. Richette: None; T. Bardin: None.




