Poster Session B
Systemic lupus erythematosus (SLE)
Session: (1488–1512) SLE – Treatment Poster II
1502: Introduction of Belimumab Within Five Years of the Onset of Systemic Lupus Erythematosus (SLE) Contributes to the Better Therapeutic Response; A Multi-center Retrospective Cohort Study
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- KK
Kanako Kojima, MD
Nagasaki university graduate school
Nagasaki, JapanDisclosure information not submitted.
Abstract Poster Presenter(s)
Kanako Kojima1, Kunihiro Ichinose1, Toshimasa Shimizu1, Masataka Umeda1, Takahisa Suzuki2, Yoshikazu Nakashima3, Akitomo Okada4, Yoshiro Horai5, Keita Fujikawa6, Toshiyuki Aramaki7, Taiichiro Miyashita8, Masako Furuyama3, Naoki Matsuoka9 and Atsushi Kawakami10, 1Department of Immunology and Rheumatology, Nagasaki University Graduate School of Biomedical Sciences, Nagasaki, Japan, 2Japanese Red Cross Nagasaki Genbaku Hospital, Nagasaki, Japan, 3Nagasaki Kita Hospital, Nagasaki, Japan, 4National Hospital Organization Nagasaki Medical Center, Omura, Japan, 5Sasebo City General Hospital, Sasebo, Japan, 6Japan Community Healthcare Organization, Isahaya General Hospital, Isahaya, Japan, 7Sasebo Chuo Hospital, Sasebo, Japan, 8Miyashita Rheumatology Clinic, Omura, Japan, 9Nagasaki Medical Hospital of Rheumatology, Nagasaki, Japan, 10Nagasaki University, Nagasaki, Japan
Background/Purpose: Belimumab (BEL), B cell-targeted biologic agent for systemic lupus erythematosus (SLE), is cumulating evidence such as reducing glucocorticoids and preventing the progressionof organ damage. The BLISS-LN study evaluated the effect of BEL on lupus nephritis (LN) and found that BEL administered early in the course of LN improved proteinuria. There is ongoing debate regarding the optimal timing of administering BEL in the treatment of SLE in real-world settings. BAFF receptor expression decreases along with B cell differentiation, and the number of memory B cells is higher in SLE patients with longer disease duration. In addition, the shorter disease duration correlated with the higher blood BAFF concentration in SLE patients. Based on these reports, we hypothesized that BEL, which targeted BAFF, would be more effective in patients with shorter disease duration. In this study, we investigated whether the duration of SLE at the time of BEL introduction contributes to its treatment responsiveness.
Methods: Ninety-eight patients diagnosed with SLE according to the 1997 ACR Diagnostic Revision Criteria or 2012 SLICC Classification Criteria at Nagasaki University Hospital and its affiliated hospitals in Nagasaki Prefecture and who started BEL between December 2017 and August 2021 were included in the analysis. The primary objective of the study is to compare the decrease in SLEDAI scores after the introduction of BEL at different time points (3, 6, 9, and 12 months) between two groups categorized based on disease duration at BEL introduction (≤5 years and >5 years). The analysis will be adjusted using a mixed-effects model. Adjustment factors include age at induction, prednisolone (PSL) dose, SLEDAI at induction and hydroxychloroquine (HCQ), and the presence of lupus nephritis.
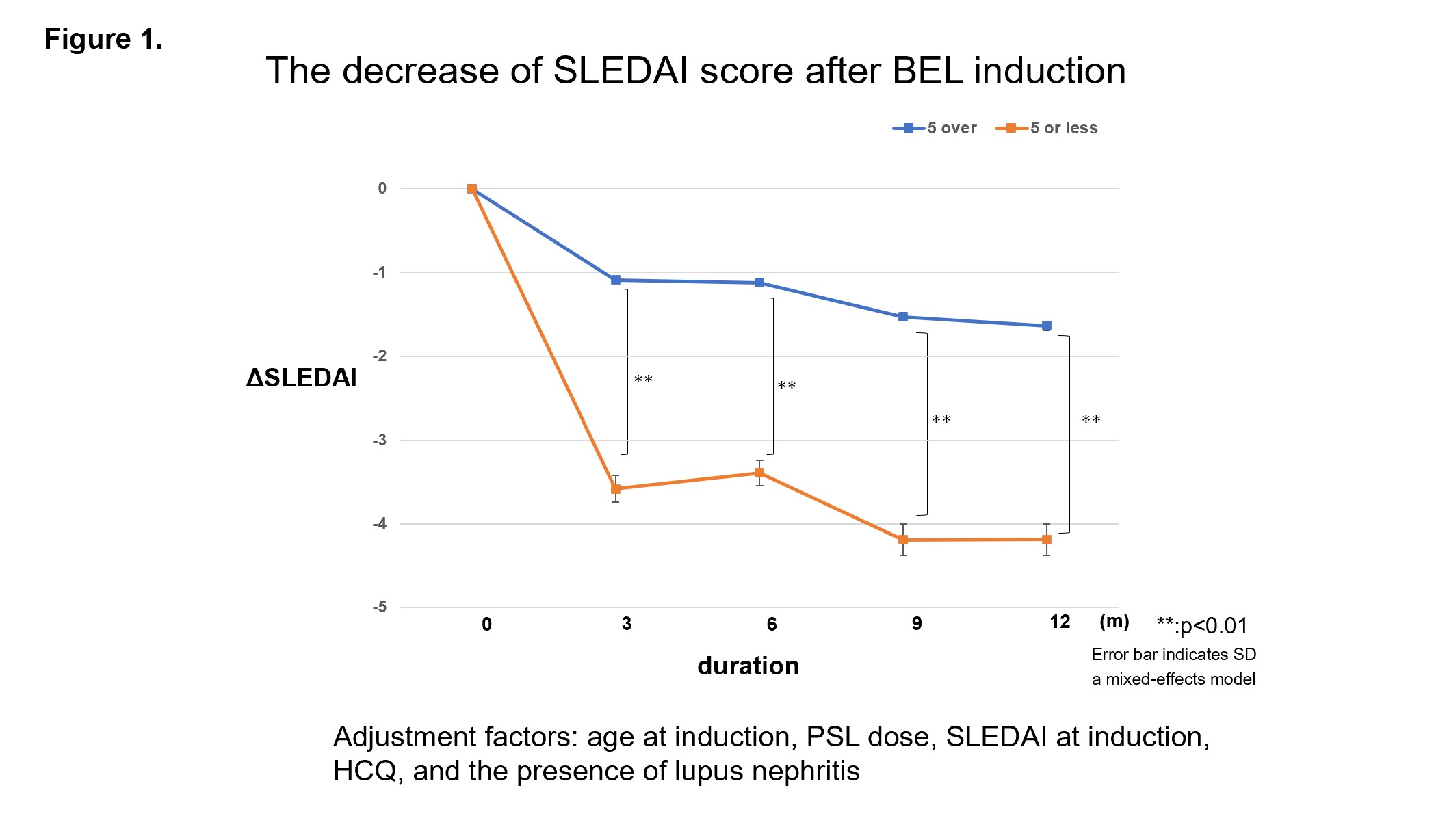
Results: In the overall patient population, the mean age at BEL introduction was 41 years, the mean SLEDAI score was 7, the mean dose of PSL was 9 mg/day, and HCQ was used in 50 patients (51%). The number of ≤5 years group was 20 patients and the number of >5 years was 78 patients. Both groups (≤5 years and >5 years) showed improvement in SLEDAI scores 3 months after the BEL introduction. In addition, the group receiving BEL at 5 years or less showed significantly greater improvement in SLEDAI than the group receiving BEL at more than 5 years at each evaluation time point after BEL introduction.
Conclusion: Our investigating headlights and earlier introduction of BEL in SLE patients may contribute to a more remarkable improvement in disease activity. This finding could potentially impact the clinical decision-making process regarding the implementation of a treat-to-target strategy for patients with SLE.
K. Kojima: None; K. Ichinose: AstraZeneca, 6, GlaxoSmithKlein(GSK), 6; T. Shimizu: None; M. Umeda: None; T. Suzuki: None; Y. Nakashima: None; A. Okada: None; Y. Horai: None; K. Fujikawa: None; T. Aramaki: None; T. Miyashita: None; M. Furuyama: None; N. Matsuoka: None; A. Kawakami: None.
Background/Purpose: Belimumab (BEL), B cell-targeted biologic agent for systemic lupus erythematosus (SLE), is cumulating evidence such as reducing glucocorticoids and preventing the progressionof organ damage. The BLISS-LN study evaluated the effect of BEL on lupus nephritis (LN) and found that BEL administered early in the course of LN improved proteinuria. There is ongoing debate regarding the optimal timing of administering BEL in the treatment of SLE in real-world settings. BAFF receptor expression decreases along with B cell differentiation, and the number of memory B cells is higher in SLE patients with longer disease duration. In addition, the shorter disease duration correlated with the higher blood BAFF concentration in SLE patients. Based on these reports, we hypothesized that BEL, which targeted BAFF, would be more effective in patients with shorter disease duration. In this study, we investigated whether the duration of SLE at the time of BEL introduction contributes to its treatment responsiveness.
Methods: Ninety-eight patients diagnosed with SLE according to the 1997 ACR Diagnostic Revision Criteria or 2012 SLICC Classification Criteria at Nagasaki University Hospital and its affiliated hospitals in Nagasaki Prefecture and who started BEL between December 2017 and August 2021 were included in the analysis. The primary objective of the study is to compare the decrease in SLEDAI scores after the introduction of BEL at different time points (3, 6, 9, and 12 months) between two groups categorized based on disease duration at BEL introduction (≤5 years and >5 years). The analysis will be adjusted using a mixed-effects model. Adjustment factors include age at induction, prednisolone (PSL) dose, SLEDAI at induction and hydroxychloroquine (HCQ), and the presence of lupus nephritis.
Results: In the overall patient population, the mean age at BEL introduction was 41 years, the mean SLEDAI score was 7, the mean dose of PSL was 9 mg/day, and HCQ was used in 50 patients (51%). The number of ≤5 years group was 20 patients and the number of >5 years was 78 patients. Both groups (≤5 years and >5 years) showed improvement in SLEDAI scores 3 months after the BEL introduction. In addition, the group receiving BEL at 5 years or less showed significantly greater improvement in SLEDAI than the group receiving BEL at more than 5 years at each evaluation time point after BEL introduction.
Conclusion: Our investigating headlights and earlier introduction of BEL in SLE patients may contribute to a more remarkable improvement in disease activity. This finding could potentially impact the clinical decision-making process regarding the implementation of a treat-to-target strategy for patients with SLE.

The decrease of SLEDAI score after BEL induction
K. Kojima: None; K. Ichinose: AstraZeneca, 6, GlaxoSmithKlein(GSK), 6; T. Shimizu: None; M. Umeda: None; T. Suzuki: None; Y. Nakashima: None; A. Okada: None; Y. Horai: None; K. Fujikawa: None; T. Aramaki: None; T. Miyashita: None; M. Furuyama: None; N. Matsuoka: None; A. Kawakami: None.



