Poster Session A
Antiphospholipid Syndrome
Session: (0096–0116) Antiphospholipid Syndrome Poster
0114: Platelets Are Highly Activated and Could Participate in Immune Abnormality of APS
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall

Xiaofeng Zeng, MD (he/him/his)
Peking Union Medical College Hospital Department of Rheumatology and Clinical Immunology
Beijing, Beijing, ChinaDisclosure information not submitted.
Abstract Poster Presenter(s)
Yu Shi1, Xiaofan Luo2, Quan Chen3, Jiuliang zhao4, Mengtao Li4 and xiaofeng Zeng5, 1Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China, 2Nankai University, Tianjin, China, 3Nankai University, Beijing, China, 4Peking Union Medical College Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China, 5Department of Rheumatology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China
Background/Purpose: Platelets play a pivotal role in the process of coagulation and other biological process. Studies have shown multiple evidences that platelets are highly activated in APS, and have the ability to enhance the activity of other cells like endothelial cells, monocytes, and neutrophils. Emerging evidences have indicated that platelets are more than a part of coagulation process.
Methods: We included 35 patients who fulfilled the 2006 Sydney classification criteria of APS and 18 age, sex matched heathy controls (HC). Activation of platelet was measured by flow cytometry. Reactive oxygen species (ROS) production and mitochondrial respiration was measured. RNA-sequencing was performed.
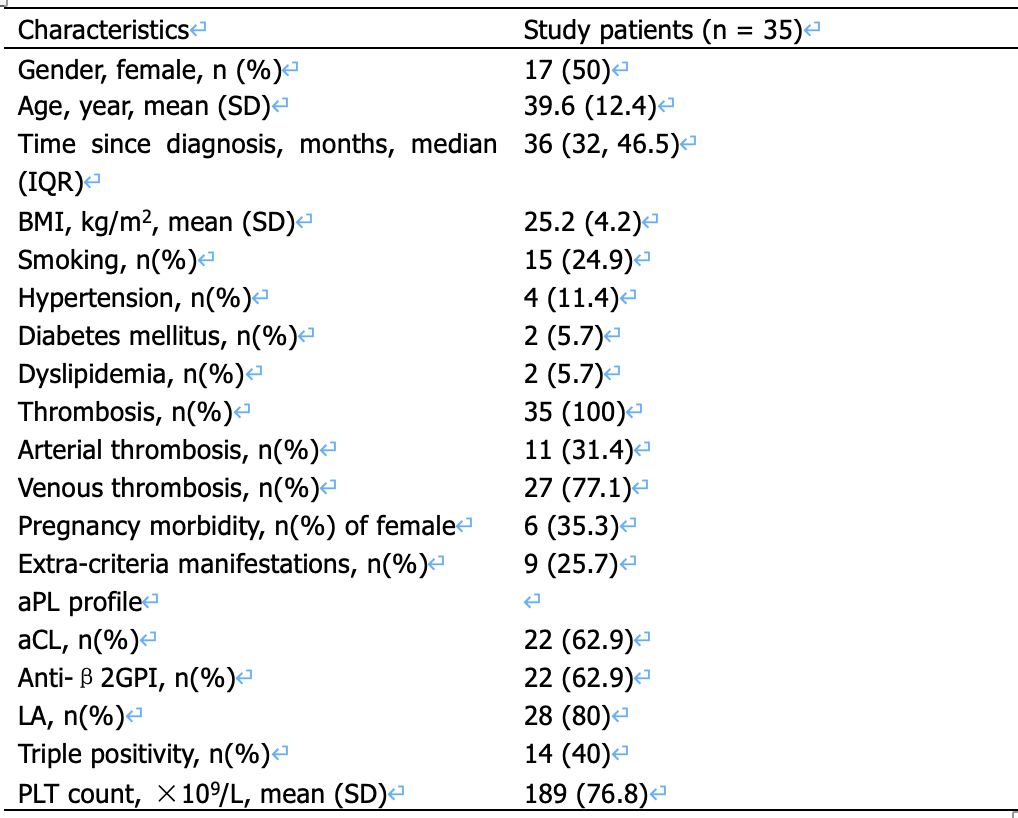
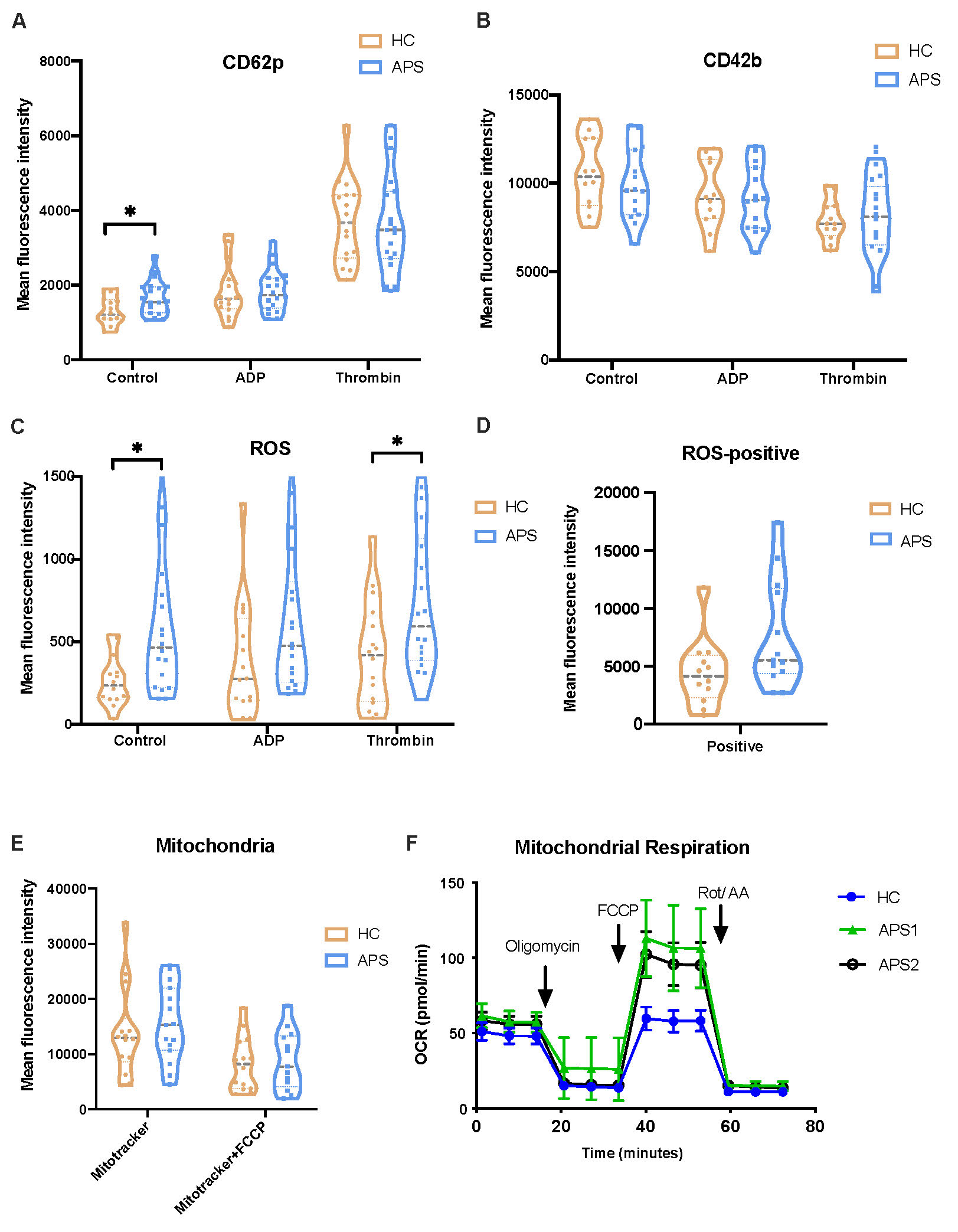
Results: The baseline characteristics of studied patients are shown in Table 1. Platelet activation was detected by the surface expression of CD62p after gating with CD41a. As presented in Figure 1A, the mean fluorescent intensity of CD62p was significantly increased in APS patients compared with HC (p< 0.05) . After stimulated with adenosine diphosphate (ADP, 0.1U/ml) and thrombin (2U/ml), the activation was similar between patients and HC. Previous research showed platelets treated with immune thrombocytopenia plasma was induced to shed GPIbα(CD42b), so we examined the surface expression of CD42b and found no statistic difference (Figure 1B). Then we detected the ROS production (Figure 1C-D)and found that platelets of APS patients both in original state and activated with thrombin produced significantly more ROS than the HC. Then we want to explore if platelets from APS patients contain more mitochondria or it is because the metabolism is more activated. Mitotracker was used to label mitochondria in platelets. As shown in Figure1E, the quantity of mitochondria was similar. We used seahorse analysis to examine mitochondrial respiration and found that maximum respiration was evidently higher in platelets from APS patients (Figure 1F).
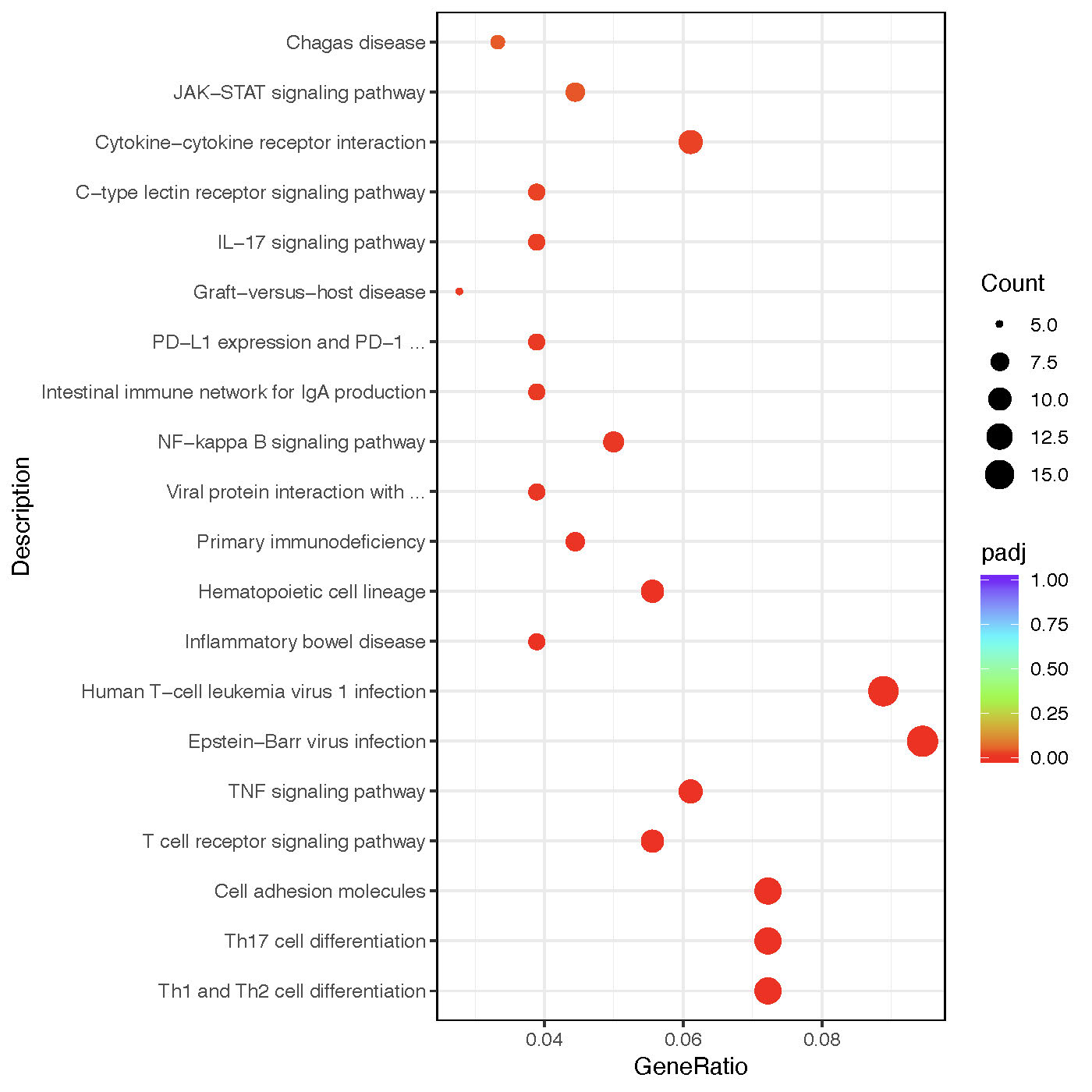
We performed RNA sequencing in 14 APS patients and 6 age, sex matched HC. 748 differently expressed genes with adjusted p-value < 0.01 and |log2FoldChange| > 1.5 were screened and tested enrichment in KEGG pathways (Figure 2). It is evident that multiple immune related pathways were enriched including inflammatory signaling pathways (JAK-STAT, IL-17 signaling pathways) and T cell associated pathways (Th1, Th2, and Th17 differentiation).
Conclusion: Platelet activation and mitochondrial respiration is highly increased in APS. The platelet transcriptome of APS is significantly different from HC. Platelets could interact as immune cells to modulate T cell activity.
Y. Shi: None; X. Luo: None; Q. Chen: None; J. zhao: None; M. Li: None; x. Zeng: None.
Background/Purpose: Platelets play a pivotal role in the process of coagulation and other biological process. Studies have shown multiple evidences that platelets are highly activated in APS, and have the ability to enhance the activity of other cells like endothelial cells, monocytes, and neutrophils. Emerging evidences have indicated that platelets are more than a part of coagulation process.
Methods: We included 35 patients who fulfilled the 2006 Sydney classification criteria of APS and 18 age, sex matched heathy controls (HC). Activation of platelet was measured by flow cytometry. Reactive oxygen species (ROS) production and mitochondrial respiration was measured. RNA-sequencing was performed.
Results: The baseline characteristics of studied patients are shown in Table 1. Platelet activation was detected by the surface expression of CD62p after gating with CD41a. As presented in Figure 1A, the mean fluorescent intensity of CD62p was significantly increased in APS patients compared with HC (p< 0.05) . After stimulated with adenosine diphosphate (ADP, 0.1U/ml) and thrombin (2U/ml), the activation was similar between patients and HC. Previous research showed platelets treated with immune thrombocytopenia plasma was induced to shed GPIbα(CD42b), so we examined the surface expression of CD42b and found no statistic difference (Figure 1B). Then we detected the ROS production (Figure 1C-D)and found that platelets of APS patients both in original state and activated with thrombin produced significantly more ROS than the HC. Then we want to explore if platelets from APS patients contain more mitochondria or it is because the metabolism is more activated. Mitotracker was used to label mitochondria in platelets. As shown in Figure1E, the quantity of mitochondria was similar. We used seahorse analysis to examine mitochondrial respiration and found that maximum respiration was evidently higher in platelets from APS patients (Figure 1F).
We performed RNA sequencing in 14 APS patients and 6 age, sex matched HC. 748 differently expressed genes with adjusted p-value < 0.01 and |log2FoldChange| > 1.5 were screened and tested enrichment in KEGG pathways (Figure 2). It is evident that multiple immune related pathways were enriched including inflammatory signaling pathways (JAK-STAT, IL-17 signaling pathways) and T cell associated pathways (Th1, Th2, and Th17 differentiation).
Conclusion: Platelet activation and mitochondrial respiration is highly increased in APS. The platelet transcriptome of APS is significantly different from HC. Platelets could interact as immune cells to modulate T cell activity.

Table 1. Baseline demographic, clinical, and laboratory characteristics of study patients.

Figure 1. Platelet flow cytometry and mitochondrial respiration of study patients.

Figure 2. KEGG pathway enrichment of differently expressed genes from platelet RNA sequencing.
Y. Shi: None; X. Luo: None; Q. Chen: None; J. zhao: None; M. Li: None; x. Zeng: None.



