Poster Session A
Antiphospholipid Syndrome
Session: (0096–0116) Antiphospholipid Syndrome Poster
0111: Hippo-YAP1-CCN2 Signaling by Microvascular Endothelial Cells Licenses Vascular Smooth Muscle Cell Proliferation in Antiphospholipid Syndrome
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall

Eliza Pei-Suen Tsou, PhD
University of Michigan
Ann Arbor, MI, United StatesDisclosure(s): No financial relationships with ineligible companies to disclose
Abstract Poster Presenter(s)
Wenying Liang1, Allison Billi1, Srilakshmi Yalavarthi1, Christine Rysenga1, Claire Hoy1, Cyrus Sarosh1, Yu Zuo1, Eliza Pei-Suen Tsou1, Jason Knight1 and Hui Shi2, 1University of Michigan, Ann Arbor, MI, 2Department of Rheumatology and Immunology, Ruijin Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai, China
Background/Purpose: Some patients with antiphospholipid syndrome (APS) are afflicted by an insidious small-vessel vasculopathy that results in the accrual of organ damage over time. While the neotima formation that heralds this vasculopathy is most commonly visualized upon kidney biopsy, other organs such as the brain, heart, and skin may also be at risk. We previously used single-cell RNA sequencing of APS skin biopsies to identify the upregulation of various proliferation-supporting genes in microvascular endothelial cells (MVECs). The most notable example was CCN2 (also known as CTGF), a classic downstream target of Hippo-YAP1 signaling. Furthermore, the previous bioinformatic analysis demonstrated the potential for strong communication between secreted MVEC ligands such as CCN2 and vascular smooth muscle cells (VSMC) receptors—a potentially critical interaction given the important role of VSMC proliferation and migration in neointima formation. Here, we aimed to determine the potential role of the Hippo-YAP1-CCN2 axis in APS micro-vasculopathy.
Methods: Isolated healthy MVECs were cultured with APS patient serum or patient-derived IgG. CCN2 expression was measured by quantitative PCR and ELISA. Immunofluorescence (IF) microscopy and western blotting were used to assess YAP1 signaling. To study cellular communication, human VSMCs were cultured with conditioned media from APS-stimulated MVECs. The proliferation and migration of VSMCs were determined by BrdU-staining and wound-scratch assays, respectively. Finally, CCN2 expression was assessed in kidney biopsies from patients with APS nephropathy by IF microscopy.
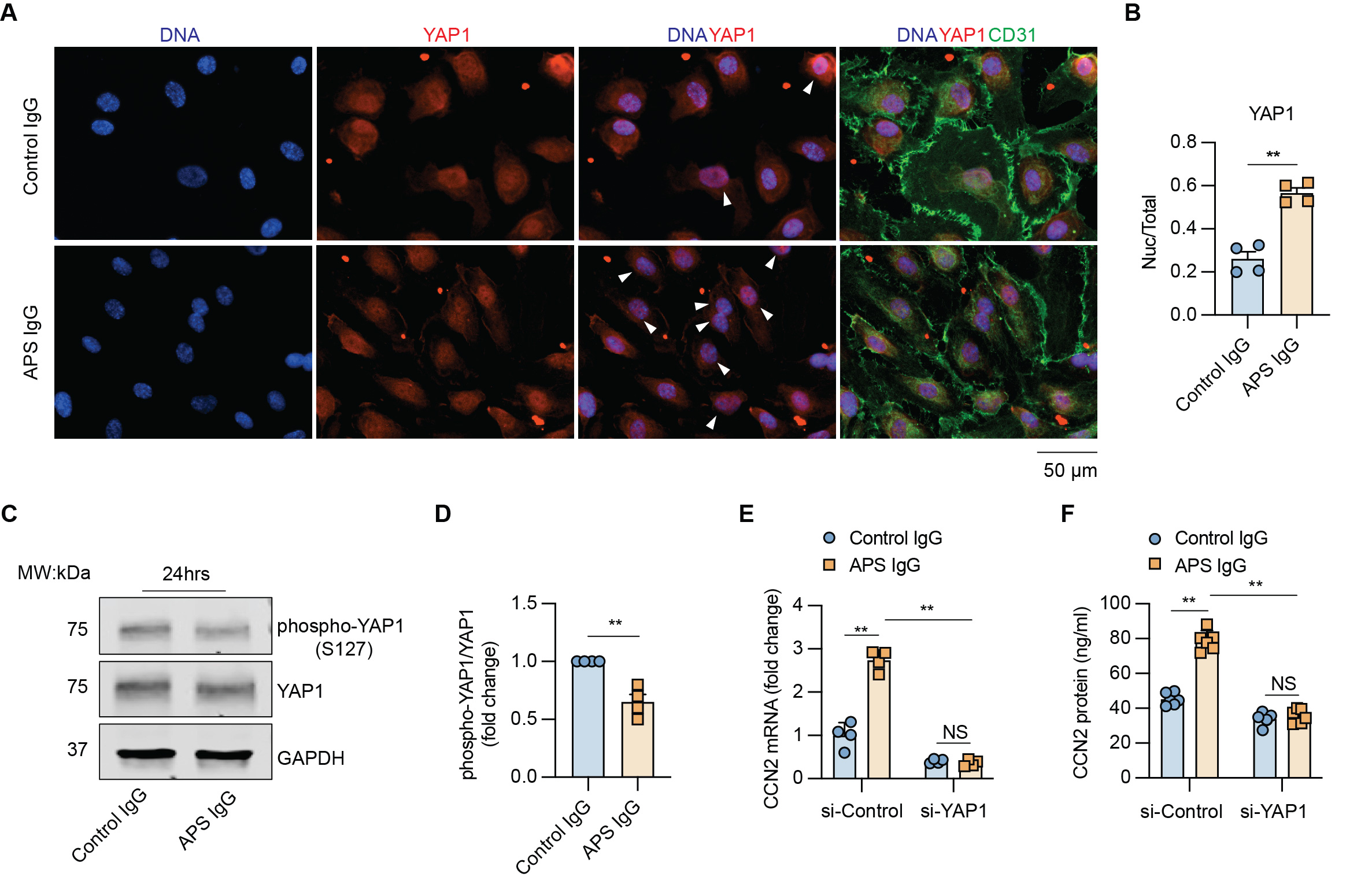
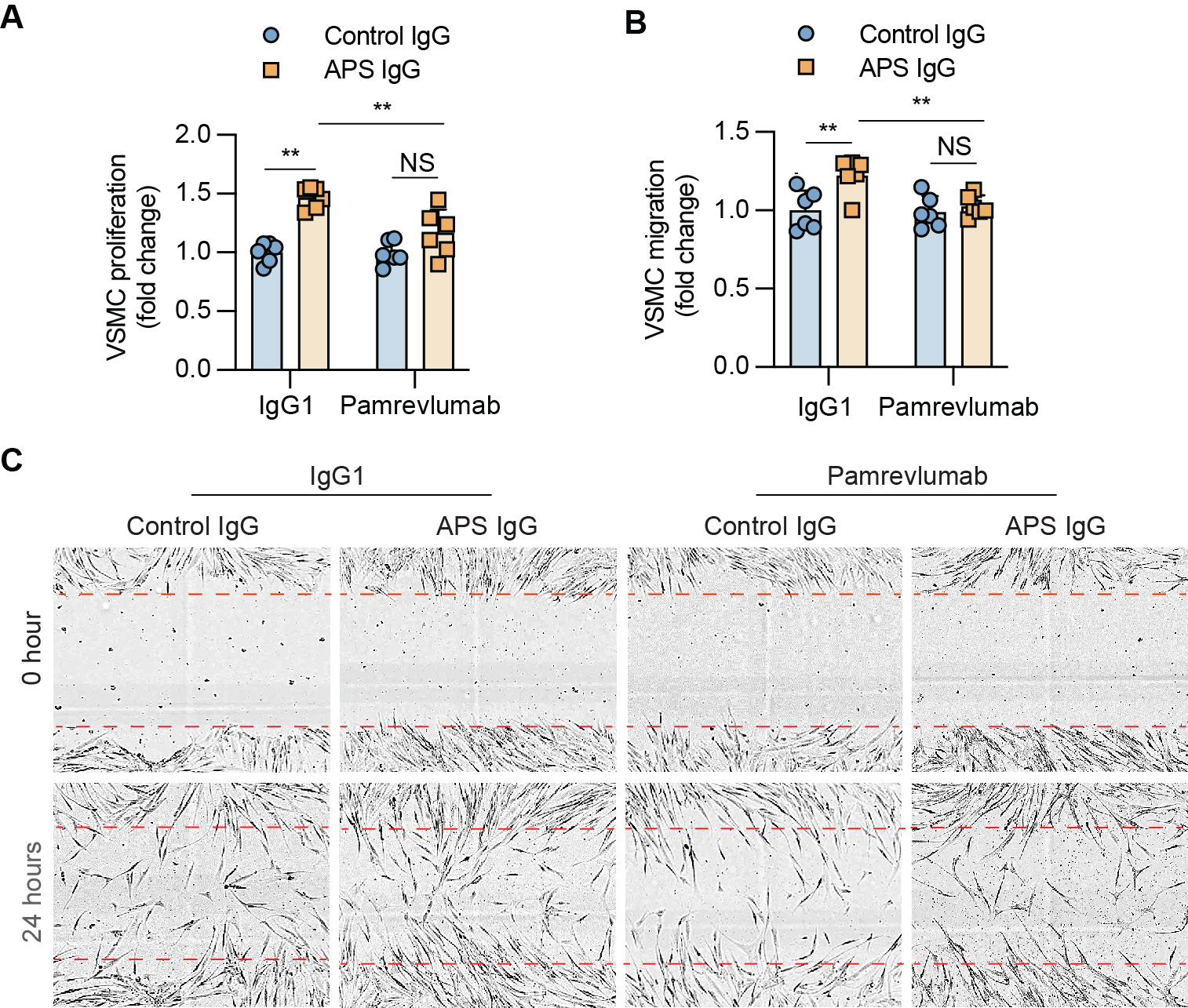
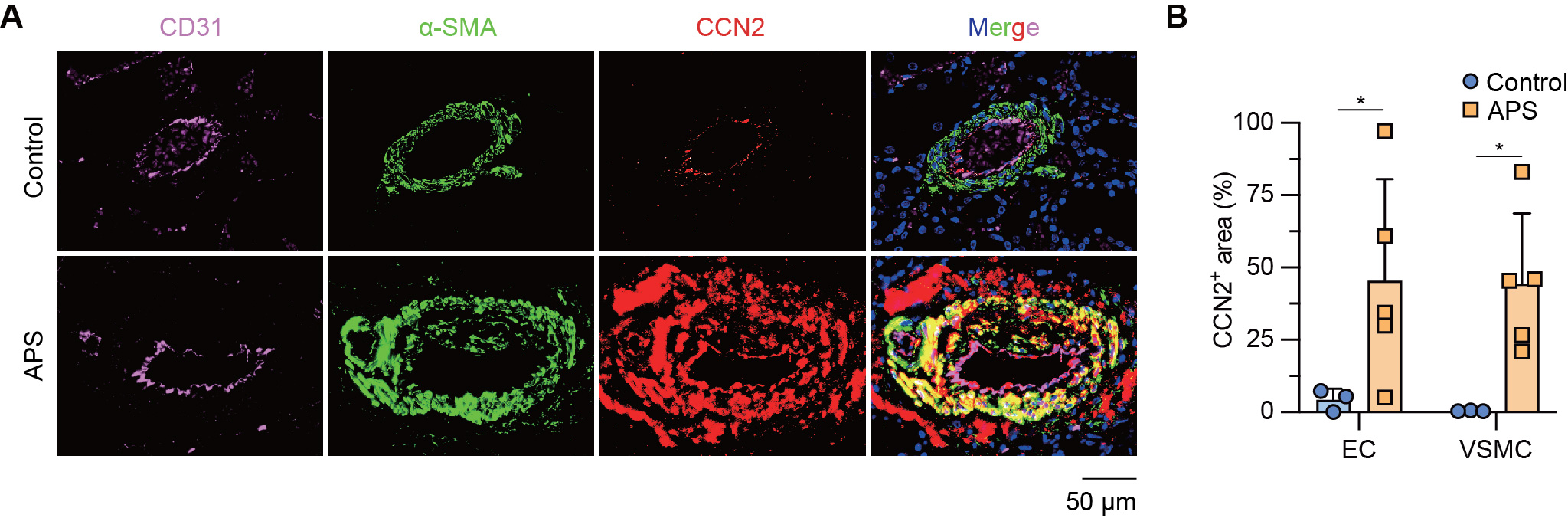
Results: Culture of MVECs with APS patient serum or patient-derived IgG led to upregulation of CCN2 mRNA (fold change 2.3 ± 0.3 and 2.8 ± 0.1, respectively, both p< 0.01). Concomitantly, secreted CCN2 protein in the supernatant was increased as assessed by ELISA (data not shown). In parallel studies, the activation of the Hippo-YAP1 pathway was confirmed by increased YAP1 nuclear translocation and reduced YAP1 phosphorylation (Figure 1A-D). Notably, the knockdown of YAP1 in MVECs abolished APS IgG-induced CCN2 expression and secretion (Figure 1E-F). Regarding MVEC-VSMC communication, conditioned media from APS IgG-stimulated MVECs triggered a noncontractile phenotype in VSMCs, accompanied by increased proliferation and migration. These phenotypes were mitigated by the humanized CCN2-blocking antibody, pamrevlumab (Figure 2). Finally, kidney biopsies from patients with APS nephropathy showed markedly enhanced CCN2 expression in the thickened intima and media of microvessels, indicating the in vivo relevance of this pathway (Figure 3).
Conclusion: We found that APS serum and patient-derived IgG triggered YAP1 nuclear translocation in MVECs, which was accompanied by increased CCN2 expression and secretion. The secreted CCN2 was then able to trigger a phenotypic switch in VSMCs toward the migratory and proliferative characteristics that are necessary for neointima formation. Blocking either YAP1 or CCN2 might be a novel approach for the treatment of APS-associated micro-vasculopathies.
W. Liang: None; A. Billi: None; S. Yalavarthi: None; C. Rysenga: None; C. Hoy: None; C. Sarosh: None; Y. Zuo: None; E. Tsou: None; J. Knight: Jazz Pharmaceuticals, 2; H. Shi: None.
Background/Purpose: Some patients with antiphospholipid syndrome (APS) are afflicted by an insidious small-vessel vasculopathy that results in the accrual of organ damage over time. While the neotima formation that heralds this vasculopathy is most commonly visualized upon kidney biopsy, other organs such as the brain, heart, and skin may also be at risk. We previously used single-cell RNA sequencing of APS skin biopsies to identify the upregulation of various proliferation-supporting genes in microvascular endothelial cells (MVECs). The most notable example was CCN2 (also known as CTGF), a classic downstream target of Hippo-YAP1 signaling. Furthermore, the previous bioinformatic analysis demonstrated the potential for strong communication between secreted MVEC ligands such as CCN2 and vascular smooth muscle cells (VSMC) receptors—a potentially critical interaction given the important role of VSMC proliferation and migration in neointima formation. Here, we aimed to determine the potential role of the Hippo-YAP1-CCN2 axis in APS micro-vasculopathy.
Methods: Isolated healthy MVECs were cultured with APS patient serum or patient-derived IgG. CCN2 expression was measured by quantitative PCR and ELISA. Immunofluorescence (IF) microscopy and western blotting were used to assess YAP1 signaling. To study cellular communication, human VSMCs were cultured with conditioned media from APS-stimulated MVECs. The proliferation and migration of VSMCs were determined by BrdU-staining and wound-scratch assays, respectively. Finally, CCN2 expression was assessed in kidney biopsies from patients with APS nephropathy by IF microscopy.
Results: Culture of MVECs with APS patient serum or patient-derived IgG led to upregulation of CCN2 mRNA (fold change 2.3 ± 0.3 and 2.8 ± 0.1, respectively, both p< 0.01). Concomitantly, secreted CCN2 protein in the supernatant was increased as assessed by ELISA (data not shown). In parallel studies, the activation of the Hippo-YAP1 pathway was confirmed by increased YAP1 nuclear translocation and reduced YAP1 phosphorylation (Figure 1A-D). Notably, the knockdown of YAP1 in MVECs abolished APS IgG-induced CCN2 expression and secretion (Figure 1E-F). Regarding MVEC-VSMC communication, conditioned media from APS IgG-stimulated MVECs triggered a noncontractile phenotype in VSMCs, accompanied by increased proliferation and migration. These phenotypes were mitigated by the humanized CCN2-blocking antibody, pamrevlumab (Figure 2). Finally, kidney biopsies from patients with APS nephropathy showed markedly enhanced CCN2 expression in the thickened intima and media of microvessels, indicating the in vivo relevance of this pathway (Figure 3).
Conclusion: We found that APS serum and patient-derived IgG triggered YAP1 nuclear translocation in MVECs, which was accompanied by increased CCN2 expression and secretion. The secreted CCN2 was then able to trigger a phenotypic switch in VSMCs toward the migratory and proliferative characteristics that are necessary for neointima formation. Blocking either YAP1 or CCN2 might be a novel approach for the treatment of APS-associated micro-vasculopathies.

Figure 1. YAP1 mediates APS antibody-induced endothelial cell CCN2 expression. A, Representative images of YAP1 expression (red) in control IgG- or APS IgG-stimulated MVECs (both 100 µg/ml), accompanied by markers of endothelial cells (CD31, green) and nuclear DNA (Hoechst, blue). Arrowheads indicate cells with YAP1 nuclear translocation. Scale bar=50 µm. B, The ratio of cells with YAP1 nuclear translocation in the indicated groups. C, Representative Western blot of phospho-YAP1 (S127) and total YAP1 in control IgG- or APS IgG-stimulated MVECs. D, The band intensity of phospho-YAP1/total YAP1 from four independent western blots was quantified and normalized with GAPDH. MVECs stimulated with control IgG were set as 1. E-F, The relative mRNA and secreted protein levels of CCN2 in control or YAP1-knockdown MVECs stimulated with control or APS IgG. Data are presented as mean ± SEM. Unpaired t-test for B and D. Two-way ANOVA followed by Sidak post hoc analysis for E and F. n=4/group. si-Control, nontargeting small interfering RNA control. si-YAP1, small interfering RNA targeting YAP1. **p < 0.01, NS: not significant.

Figure 2. Communication between microvascular endothelial cells (MVECs) and vascular smooth muscle cells (VSMCs) in APS. A-B, Proliferation (A) and migration (B) of VSMCs were quantified and presented as the relative fold change compared to the group stimulated by control IgG1 and conditioned media from control IgG-stimulated MVECs. Migration was quantified by wound confluence (%). C, Representative images of each group at 0 or 24 hours after scratch. Data are represented as mean ± SEM and p values were calculated by two-way ANOVA followed by Sidak post hoc analysis. n=6/group. **p < 0.01, NS: not significant.

Figure 3. High expression of vascular CCN2 in patients with APS nephropathy. A, Representative images of CCN2 expression (red) in the kidney vessels from healthy controls or APS nephropathy patients. Endothelial cells (CD31+ area) and vascular smooth muscle cells (α-SMA+ area) were labeled in purple and green, respectively. Nuclear DNA was labeled by Hoechst (blue). Scale bar=50 µm. B, The percentage of CCN2+ area in kidney endothelium of indicated groups was calculated as CCN2+CD31+ area/CD31+ area. The percentage of CCN2+ area in kidney vascular smooth muscle cells of indicated groups was calculated as CCN2+α-SMA+ area/α-SMA+ area. Data are presented as mean ± SEM and p values were calculated by two-way ANOVA followed by Sidak post hoc analysis. n=3 for control, n=5 for APS nephropathy patients. *p<0.05. EC, endothelial cell. VSMC, vascular smooth muscle cell.
W. Liang: None; A. Billi: None; S. Yalavarthi: None; C. Rysenga: None; C. Hoy: None; C. Sarosh: None; Y. Zuo: None; E. Tsou: None; J. Knight: Jazz Pharmaceuticals, 2; H. Shi: None.



