Poster Session A
Antiphospholipid Syndrome
Session: (0096–0116) Antiphospholipid Syndrome Poster
0108: Decoding Antiphospholipid Syndrome Laboratory Test Outcomes in a Large Multicenter Electronic Health Record Database
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- EB
Emily Balczewski, BA
University of Michigan
Ann Arbor, MI, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Emily Balczewski, Wenying Liang, Amala Ambati, Yu Zuo, Karandeep Singh and Jason Knight, University of Michigan, Ann Arbor, MI
Background/Purpose: Electronic health record (EHR) data provide an inexpensive, information-rich tool to study rare diseases like antiphospholipid syndrome (APS). Many such studies rely on structured EHR data, e.g., diagnostic codes, laboratory testing, medication information. As a first step toward assessing the feasibility of using structured EHR data to study APS, we evaluated the presence and positivity of APS antibody testing in a diverse, US-based cohort using the All of Us Research Network Database (AOU).
Methods: Possible APS patients were identified by the presence of SNOMED-CT codes: 26843008 (Antiphospholipid syndrome) and 19267009 (Lupus anticoagulant disorder). Among possible APS patients, APS laboratory tests – anticardiolipin (aCL), anti-beta-2 glycoprotein I (aβ2GPI), and lupus anticoagulant (LA; confirmatory tests only) – were identified with a hand-curated list of LOINC and SNOMED-CT codes. Tests were identified as positive if they had a) a text result indicating positivity (e.g. positive, high, abnormal), b) a numeric result exceeding the normal reported range for that test, or c) a numeric result exceeding 40 if the test lacked a range, but included units of GPL, MPL, SGU, or SMU; negative tests were identified analogously. Tests without text or numeric results, ranges, or units, or tests with units other than those referenced above were classified as indeterminate. T-tests were two-sided with Bonferroni correction.
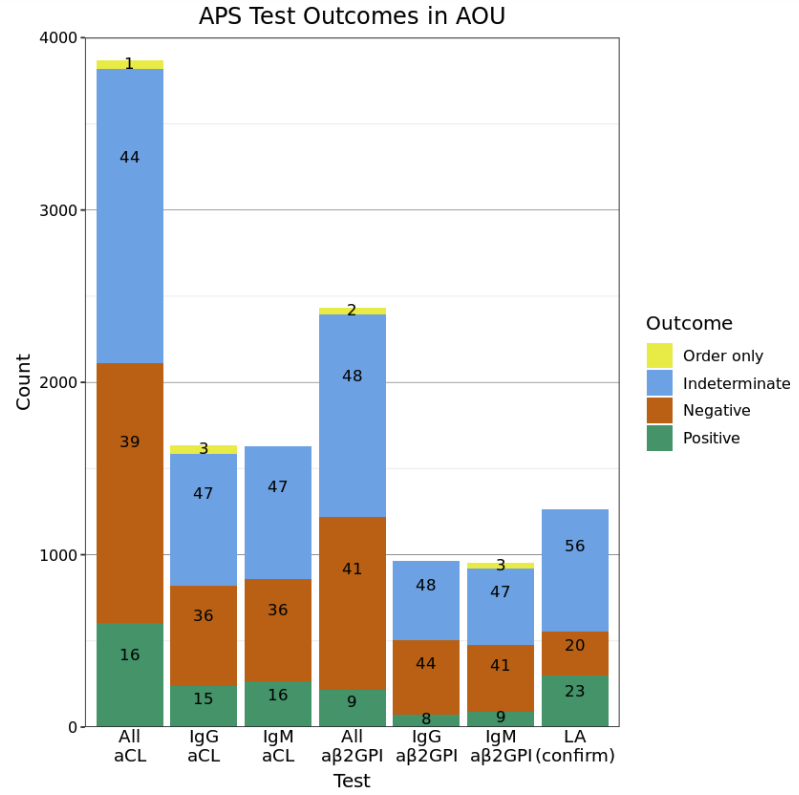
Results: Out of 372,082 total AOU patients, 883 (2.4 in 1,000) were identified as possible APS patients with demographic characteristics of 74% female, 55% White, and 17% Black, and a mean age of 55. Despite having testing information on 613 (70%) patients, only 434 (49%) had enough data to classify a test as positive or negative. Of these, 245 (28%) had any positive antiphospholipid test (Figure 1). Additionally, only 119 (13%) had two positive tests greater than 12 weeks apart, in accordance with APS classification criteria. Despite these low rates of positive tests, possible APS patients had on average 40 rows (i.e., orders and results) of APS test data compared to 1.7 rows among the non-APS patients. Percentages of positive tests were higher for LA (possibly because of its confirmatory nature) than aCL or aβ2GPI; percentages of indeterminate test results were high (44-56%) across all tests (Figure 2).
Conclusion: AOU offers one of the largest and most diverse datasets for studying the epidemiology and effects of APS. However, poor data quality can complicate the identification of a robust cohort of APS patients; only 1 in 8 patients with an APS diagnostic code in AOU met the laboratory criteria for APS, despite having an over-enrichment of APS test data compared to the whole database. This result is partially driven by half of all tests having an indeterminate result due to missing data. While improved standardization for testing and documentation could ameliorate data quality issues, creating a "digital phenotype" which combines many and different data types from the EHR will be needed to confidently identify likely APS patients for further study.
.jpg)
E. Balczewski: None; W. Liang: None; A. Ambati: None; Y. Zuo: None; K. Singh: Blue Cross Blue Shield of Michigan, 5, Flatiron Health, 1, National Institute of Diabetes and Digestive and Kidney Diseases, 5, Teva Pharmaceuticals, 5; J. Knight: Jazz Pharmaceuticals, 2.
Background/Purpose: Electronic health record (EHR) data provide an inexpensive, information-rich tool to study rare diseases like antiphospholipid syndrome (APS). Many such studies rely on structured EHR data, e.g., diagnostic codes, laboratory testing, medication information. As a first step toward assessing the feasibility of using structured EHR data to study APS, we evaluated the presence and positivity of APS antibody testing in a diverse, US-based cohort using the All of Us Research Network Database (AOU).
Methods: Possible APS patients were identified by the presence of SNOMED-CT codes: 26843008 (Antiphospholipid syndrome) and 19267009 (Lupus anticoagulant disorder). Among possible APS patients, APS laboratory tests – anticardiolipin (aCL), anti-beta-2 glycoprotein I (aβ2GPI), and lupus anticoagulant (LA; confirmatory tests only) – were identified with a hand-curated list of LOINC and SNOMED-CT codes. Tests were identified as positive if they had a) a text result indicating positivity (e.g. positive, high, abnormal), b) a numeric result exceeding the normal reported range for that test, or c) a numeric result exceeding 40 if the test lacked a range, but included units of GPL, MPL, SGU, or SMU; negative tests were identified analogously. Tests without text or numeric results, ranges, or units, or tests with units other than those referenced above were classified as indeterminate. T-tests were two-sided with Bonferroni correction.
Results: Out of 372,082 total AOU patients, 883 (2.4 in 1,000) were identified as possible APS patients with demographic characteristics of 74% female, 55% White, and 17% Black, and a mean age of 55. Despite having testing information on 613 (70%) patients, only 434 (49%) had enough data to classify a test as positive or negative. Of these, 245 (28%) had any positive antiphospholipid test (Figure 1). Additionally, only 119 (13%) had two positive tests greater than 12 weeks apart, in accordance with APS classification criteria. Despite these low rates of positive tests, possible APS patients had on average 40 rows (i.e., orders and results) of APS test data compared to 1.7 rows among the non-APS patients. Percentages of positive tests were higher for LA (possibly because of its confirmatory nature) than aCL or aβ2GPI; percentages of indeterminate test results were high (44-56%) across all tests (Figure 2).
Conclusion: AOU offers one of the largest and most diverse datasets for studying the epidemiology and effects of APS. However, poor data quality can complicate the identification of a robust cohort of APS patients; only 1 in 8 patients with an APS diagnostic code in AOU met the laboratory criteria for APS, despite having an over-enrichment of APS test data compared to the whole database. This result is partially driven by half of all tests having an indeterminate result due to missing data. While improved standardization for testing and documentation could ameliorate data quality issues, creating a "digital phenotype" which combines many and different data types from the EHR will be needed to confidently identify likely APS patients for further study.
.jpg)
Figure 1: Upset plot of APS antibody testing outcomes by patient. Possible APS patients are sorted into one of the following test outcome categories: no test data, no positive test (i.e., only negative or indeterminate results), or positive test for one or more different APS antibodies. A single patient may have multiple tests within a single category. Categories with fewer than 20 participants are excluded for patient privacy.

Figure 2: Per-test outcomes of antiphospholipid antibody testing. Outcomes of possible APS patient lab results in the All of Us Research Network Database (AOU). Indeterminate: insufficient data to assess positivity or negativity of test; order only, EHR order without reported results. Labels on bars correspond to a per-bar percentage.
E. Balczewski: None; W. Liang: None; A. Ambati: None; Y. Zuo: None; K. Singh: Blue Cross Blue Shield of Michigan, 5, Flatiron Health, 1, National Institute of Diabetes and Digestive and Kidney Diseases, 5, Teva Pharmaceuticals, 5; J. Knight: Jazz Pharmaceuticals, 2.



