Poster Session A
Rheumatoid arthritis (RA)
Session: (0423–0459) RA – Treatment Poster I
0451: Long-term Clinical Profile of Filgotinib in Patients with Rheumatoid Arthritis by Cardiovascular Risk Factors: A Post Hoc Subgroup Analysis
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall

MAYA BUCH, MD, PhD
UNIVERSITY OF MANCHESTER
MANCHESTER, United KingdomDisclosure information not submitted.
Abstract Poster Presenter(s)
Maya Buch1, Jose A Gomez-Puerta2, Gerd Burmester3, Bernard G. Combe4, Vijay Rajendran5, Pieter-Jan Stiers6, Paul Van Hoek7, Katrien Van Beneden7, Jacques-Eric Gottenberg8, Yoshiya Tanaka9, Daniel Aletaha10, René Westhovens11 and Roberto F. Caporali12, 1University of Manchester and NIHR Manchester Biomedical Research Centre, Manchester, United Kingdom, 2Department of Rheumatology, Hospital Clinic of Barcelona, Barcelona, Spain, 3Charité – Universitätsmedizin Berlin, Department of Rheumatology and Clinical Immunology, Berlin, Germany, 4Department of Rheumatology, Montpellier University, Montpellier, France, 5Clinical Research, Galapagos NV, Mechelen, Belgium, 6Biostatistics, Galapagos NV, Mechelen, Belgium, 7Medical Safety, Galapagos NV, Mechelen, Belgium, 8Rheumatology Department, Strasbourg University Hospital, Strasbourg, France, 9University of Occupational and Environmental Health, Kitakyushu, Japan, 10Division of Rheumatology, Department of Medicine III, Medical University of Vienna, Vienna, Austria, Wien, Austria, 11Department of Rheumatology, KU Leuven, Skeletal Biology and Engineering Research Center, Leuven, Belgium, 12Department of Clinical Sciences and Community Health, University of Milan, and Department of Rheumatology and Medical Sciences, ASST Gaetano Pini-CTO, Milano, Italy
Background/Purpose: Filgotinib (FIL) is an oral Janus kinase 1 preferential inhibitor for the treatment of moderate to severe active RA. A previous pooled analysis reported long-term safety and efficacy for FIL 200 mg (FIL200) vs FIL 100 mg (FIL100) in patients (pts) aged ≥ 65 and < 65 y.1 The objective of this analysis was to report updated long-term safety and efficacy in 4 subgroups of pts with RA (aged < 65 y, ≥ 65 y, < 65 y without cardiovascular [CV] risk, and ≥ 65 y or with CV risk), treated with FIL200 vs FIL100.
Methods: This post hoc analysis pooled data from Phase 2 (DARWIN 1–3; NCT01888874, NCT01894516, NCT02065700) and Phase 3 (FINCH 1–4; NCT02889796, NCT02873936, NCT02886728, NCT03025308) trials. Data for long-term extensions (LTEs) were as of May 2, 2022 (DARWIN 3), and May 6, 2022 (FINCH 4). Analyses were by age (< 65 vs ≥ 65 y) and subgroup (< 65 y without CV risk factor vs ≥ 65 y or ≥1 CV risk factor). CV risk factors were: family history of CV disease; history of dyslipidemia, diabetes mellitus or CV disease; hypertension, ischemic vascular conditions, peripheral vascular disease, extra-articular manifestations of RA; or ever smoked. The as-treated analysis set included data for pts receiving ≥1 FIL dose. Exposure-adjusted incidence rates (EAIRs)/100 patient-years of exposure (PYE) of selected adverse events (AEs), and % of pts achieving Disease Activity Score in 28 joints using C-reactive protein (DAS28-CRP) of < 2.6 or ≤ 3.2 through Week 156 in FINCH 4, are reported.
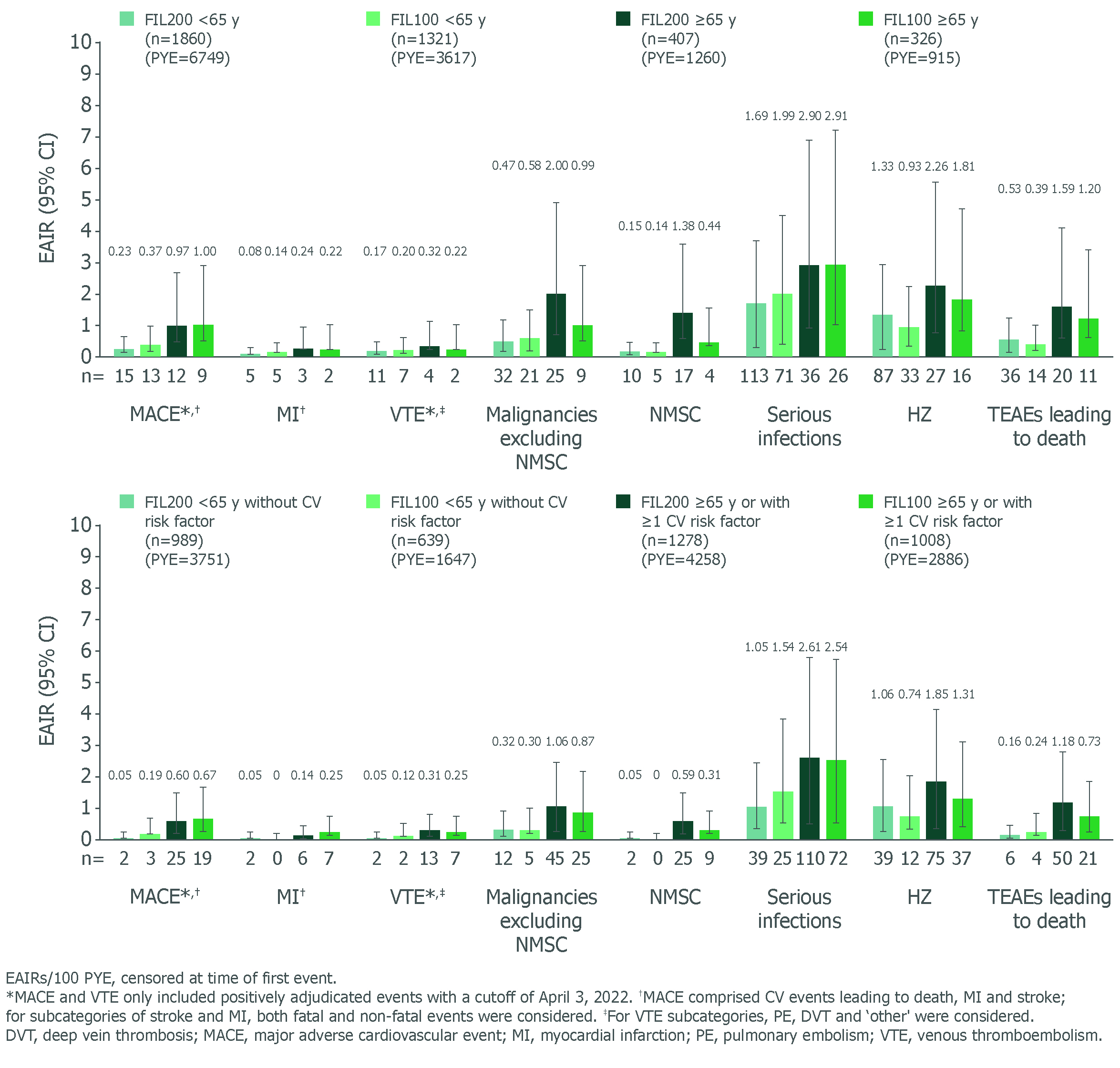
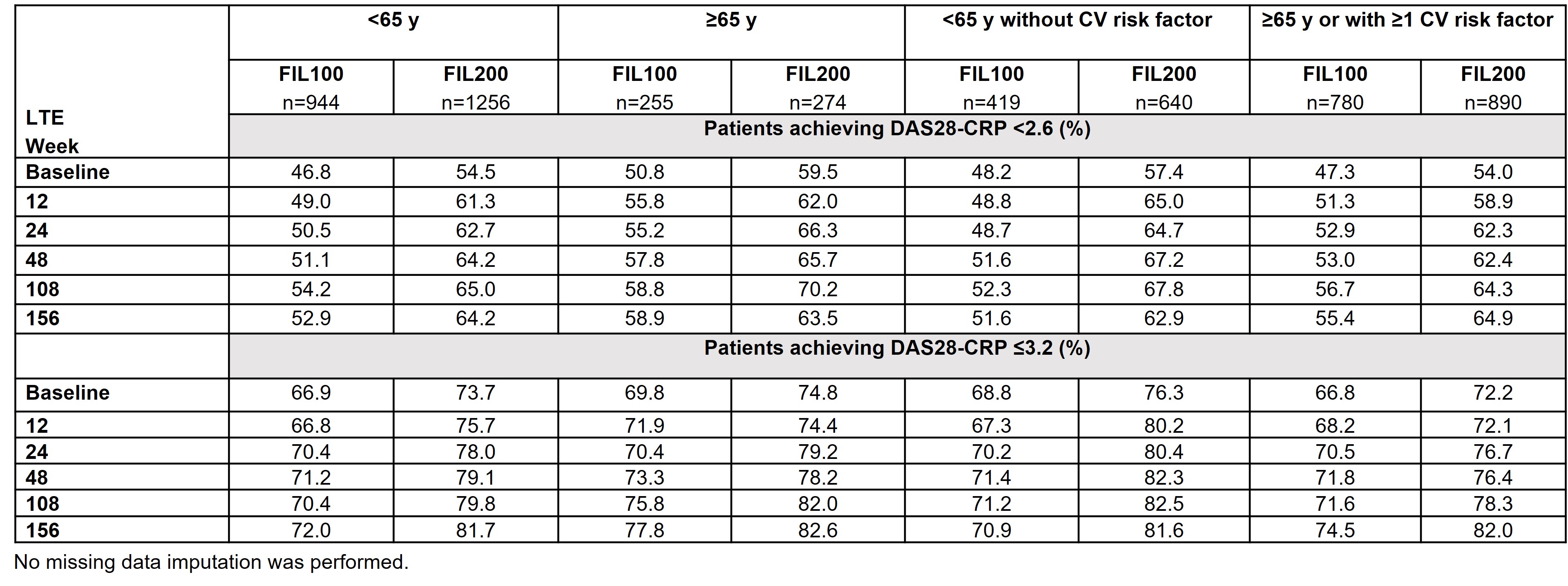
Results: Baseline characteristics by age have been reported.1 Baseline disease characteristics by age or CV risk factor were similar for disease severity and concurrent treatment. EAIRs for any treatment-emergent AEs (TEAEs) were generally higher in pts aged ≥ 65 (120.40) vs < 65 y (97.86), and higher in pts aged ≥ 65 y or with CV risk (120.45) vs pts aged < 65 y without CV risk (81.83). Pts aged ≥ 65 y, followed by the broader subgroup with CV risk factors (i.e. ≥ 65 y or ≥ 1 CV risk) had higher EAIRs of serious TEAEs and TEAEs leading to discontinuation vs other subgroups, indicating age is a key risk factor alongside other CV risk factors for developing AEs (data not shown). In pts aged ≥ 65 y, lower incidences of malignancies (excluding nonmelanoma skin cancer [NMSC]), NMSC, herpes zoster (HZ) and TEAEs leading to death were observed with FIL100 than FIL200 (Figure). In the broader subgroup with risk factors (i.e. aged ≥ 65 y or ≥ 1 CV risk factor), EAIRs of AEs were generally lower vs pts aged ≥ 65 y, indicating greater influence of age. EAIRs of AEs for subgroups (aged < 65 y without CV risk; ≥ 65 y or ≥ 1 CV risk) for FIL200 and FIL100 are shown (Figure). Rates of DAS28-CRP < 2.6 or ≤ 3.2 in all subgroups (FINCH 4) were maintained with FIL100 and FIL200 to Week 156 (Table).
Conclusion: This post hoc analysis (in pts ≥ 65 y or with ≥ 1 CV risk factor) suggests that age is an important risk factor in the evaluation of the FIL200 safety profile. Future studies are needed to address the relative contribution of age to traditional CV risk factors. In FINCH 4, efficacy was maintained in all subgroups.
Reference:
1. Buch M, et al. Arthritis Rheumatol 2022;74(Suppl 9):0281
M. Buch: AbbVie, 2, 6, 12, All paid to host institution, Boehringer Ingelheim, 2, 6, 12, Paid to host institution, Galapagos, 2, 6, 12, Paid to host institution, Gilead, 2, 5, 6, 12, Paid to host institution, Lilly, 2, 6, 12, All paid to host institution, National Insitute for Health and Care Research (NIHR), 3, 12, Maya H Buch is a National Institute for Health and Care Research (NIHR) Senior Investigator. The views expressed are those of the authors and not nece, Pfizer, 2, 12, Paid to host institution; J. Gomez-Puerta: AstraZeneca, 6, Eli Lilly, 6, Galapagos, 6, GSK, 6, Janssen, 6, Pfizer, 6, Sanofi, 2; G. Burmester: AbbVie, 2, 6, Boehringer Ingelheim, 2, 6, Bristol-Myers Squibb, 2, 6, Chugai, 6, Galapagos, 2, 6, Lilly, 2, 6, Pfizer, 2, 6, Sanofi, 2, 6; B. Combe: AbbVie, 2, 6, BMS, 6, Celltrion, 2, Eli Lilly, 1, 2, 6, Galapagos, 2, 6, Gilead, 2, Janssen, 2, 6, MSD, 6, Nordic Pharma, 5, Novartis, 1, Pfizer, 6, Roche-Chugai, 2, 6; V. Rajendran: Galapagos, 3, 11; P. Stiers: Galapagos, 3, 11; P. Van Hoek: Galapagos, 7; K. Van Beneden: Galapagos, 3, 11; J. Gottenberg: AbbVie, 2, BMS, 2, 5, Galapagos, 2, Gilead, 2, Lilly, 2, MSD, 2, Novartis, 2, Pfizer, 2, 5; Y. Tanaka: AbbVie, 6, AstraZeneca, 6, BMS, 6, Boehringer-Ingelheim, 6, Chugai, 5, 6, Eisai, 5, 6, Eli Lilly, 6, Gilead, 6, GSK, 6, Mitsubishi-Tanabe, 5, Pfizer, 6, Taiho, 6, Taisho, 5, 6; D. Aletaha: AbbVie, 2, 5, 6, Amgen, 2, 5, 6, Janssen, 2, 6, Lilly, 2, 5, 6, Merck, 2, 6, Novartis, 2, 5, 6, Pfizer, 2, 6, Roche, 2, 5, 6, Sandoz, 2, 6, Sanofi, 5, Sobi, 5; R. Westhovens: Celltrion, 2, 6, Galapagos, 2, 6, Gilead, 2, 6; R. Caporali: AbbVie, 2, 6, Amgen, 2, 6, BMS, 2, 6, Celltrion, 2, 6, Fresenius Kabi, 2, Galapagos, 2, 6, Janssen, 2, 6, Lilly, 2, 6, MSD, 2, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 2, Sandoz, 2, 6, UCB, 2, 6.
Background/Purpose: Filgotinib (FIL) is an oral Janus kinase 1 preferential inhibitor for the treatment of moderate to severe active RA. A previous pooled analysis reported long-term safety and efficacy for FIL 200 mg (FIL200) vs FIL 100 mg (FIL100) in patients (pts) aged ≥ 65 and < 65 y.1 The objective of this analysis was to report updated long-term safety and efficacy in 4 subgroups of pts with RA (aged < 65 y, ≥ 65 y, < 65 y without cardiovascular [CV] risk, and ≥ 65 y or with CV risk), treated with FIL200 vs FIL100.
Methods: This post hoc analysis pooled data from Phase 2 (DARWIN 1–3; NCT01888874, NCT01894516, NCT02065700) and Phase 3 (FINCH 1–4; NCT02889796, NCT02873936, NCT02886728, NCT03025308) trials. Data for long-term extensions (LTEs) were as of May 2, 2022 (DARWIN 3), and May 6, 2022 (FINCH 4). Analyses were by age (< 65 vs ≥ 65 y) and subgroup (< 65 y without CV risk factor vs ≥ 65 y or ≥1 CV risk factor). CV risk factors were: family history of CV disease; history of dyslipidemia, diabetes mellitus or CV disease; hypertension, ischemic vascular conditions, peripheral vascular disease, extra-articular manifestations of RA; or ever smoked. The as-treated analysis set included data for pts receiving ≥1 FIL dose. Exposure-adjusted incidence rates (EAIRs)/100 patient-years of exposure (PYE) of selected adverse events (AEs), and % of pts achieving Disease Activity Score in 28 joints using C-reactive protein (DAS28-CRP) of < 2.6 or ≤ 3.2 through Week 156 in FINCH 4, are reported.
Results: Baseline characteristics by age have been reported.1 Baseline disease characteristics by age or CV risk factor were similar for disease severity and concurrent treatment. EAIRs for any treatment-emergent AEs (TEAEs) were generally higher in pts aged ≥ 65 (120.40) vs < 65 y (97.86), and higher in pts aged ≥ 65 y or with CV risk (120.45) vs pts aged < 65 y without CV risk (81.83). Pts aged ≥ 65 y, followed by the broader subgroup with CV risk factors (i.e. ≥ 65 y or ≥ 1 CV risk) had higher EAIRs of serious TEAEs and TEAEs leading to discontinuation vs other subgroups, indicating age is a key risk factor alongside other CV risk factors for developing AEs (data not shown). In pts aged ≥ 65 y, lower incidences of malignancies (excluding nonmelanoma skin cancer [NMSC]), NMSC, herpes zoster (HZ) and TEAEs leading to death were observed with FIL100 than FIL200 (Figure). In the broader subgroup with risk factors (i.e. aged ≥ 65 y or ≥ 1 CV risk factor), EAIRs of AEs were generally lower vs pts aged ≥ 65 y, indicating greater influence of age. EAIRs of AEs for subgroups (aged < 65 y without CV risk; ≥ 65 y or ≥ 1 CV risk) for FIL200 and FIL100 are shown (Figure). Rates of DAS28-CRP < 2.6 or ≤ 3.2 in all subgroups (FINCH 4) were maintained with FIL100 and FIL200 to Week 156 (Table).
Conclusion: This post hoc analysis (in pts ≥ 65 y or with ≥ 1 CV risk factor) suggests that age is an important risk factor in the evaluation of the FIL200 safety profile. Future studies are needed to address the relative contribution of age to traditional CV risk factors. In FINCH 4, efficacy was maintained in all subgroups.
Reference:
1. Buch M, et al. Arthritis Rheumatol 2022;74(Suppl 9):0281

Figure. EAIR of AEs by age and CV risk (safety analysis set, as treated)

Table. DAS28-CRP <2.6 and ≤3.2 from LTE baseline to Week 156, by age, CV risk and FIL dose (safety analysis set, observed values)
M. Buch: AbbVie, 2, 6, 12, All paid to host institution, Boehringer Ingelheim, 2, 6, 12, Paid to host institution, Galapagos, 2, 6, 12, Paid to host institution, Gilead, 2, 5, 6, 12, Paid to host institution, Lilly, 2, 6, 12, All paid to host institution, National Insitute for Health and Care Research (NIHR), 3, 12, Maya H Buch is a National Institute for Health and Care Research (NIHR) Senior Investigator. The views expressed are those of the authors and not nece, Pfizer, 2, 12, Paid to host institution; J. Gomez-Puerta: AstraZeneca, 6, Eli Lilly, 6, Galapagos, 6, GSK, 6, Janssen, 6, Pfizer, 6, Sanofi, 2; G. Burmester: AbbVie, 2, 6, Boehringer Ingelheim, 2, 6, Bristol-Myers Squibb, 2, 6, Chugai, 6, Galapagos, 2, 6, Lilly, 2, 6, Pfizer, 2, 6, Sanofi, 2, 6; B. Combe: AbbVie, 2, 6, BMS, 6, Celltrion, 2, Eli Lilly, 1, 2, 6, Galapagos, 2, 6, Gilead, 2, Janssen, 2, 6, MSD, 6, Nordic Pharma, 5, Novartis, 1, Pfizer, 6, Roche-Chugai, 2, 6; V. Rajendran: Galapagos, 3, 11; P. Stiers: Galapagos, 3, 11; P. Van Hoek: Galapagos, 7; K. Van Beneden: Galapagos, 3, 11; J. Gottenberg: AbbVie, 2, BMS, 2, 5, Galapagos, 2, Gilead, 2, Lilly, 2, MSD, 2, Novartis, 2, Pfizer, 2, 5; Y. Tanaka: AbbVie, 6, AstraZeneca, 6, BMS, 6, Boehringer-Ingelheim, 6, Chugai, 5, 6, Eisai, 5, 6, Eli Lilly, 6, Gilead, 6, GSK, 6, Mitsubishi-Tanabe, 5, Pfizer, 6, Taiho, 6, Taisho, 5, 6; D. Aletaha: AbbVie, 2, 5, 6, Amgen, 2, 5, 6, Janssen, 2, 6, Lilly, 2, 5, 6, Merck, 2, 6, Novartis, 2, 5, 6, Pfizer, 2, 6, Roche, 2, 5, 6, Sandoz, 2, 6, Sanofi, 5, Sobi, 5; R. Westhovens: Celltrion, 2, 6, Galapagos, 2, 6, Gilead, 2, 6; R. Caporali: AbbVie, 2, 6, Amgen, 2, 6, BMS, 2, 6, Celltrion, 2, 6, Fresenius Kabi, 2, Galapagos, 2, 6, Janssen, 2, 6, Lilly, 2, 6, MSD, 2, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 2, Sandoz, 2, 6, UCB, 2, 6.



