Poster Session A
Fibrosing rheumatic diseases (scleroderma, MCTD, IgG4-related disease, scleroderma mimics)
Session: (0609–0672) Systemic Sclerosis & Related Disorders – Clinical Poster I: Research
0630: Prevalence and Risk Factors for Systemic Sclerosis Digital Ischemic Complications in the Collaborative National Quality and Efficacy Registry
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- MS
Marissa Savoie, MD
Massachusetts General Hospital
Boston, MA, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Marissa Savoie1, Monica Harding2, Jessica Alvey2, John VanBuren2, Shervin Assassi3, Elana Bernstein4, Lorinda Chung5, Luke Evnin6, Tracy Frech7, Jessica Gordon8, Faye Hant9, Laura Hummers10, Dinesh Khanna11, Kimberly Lakin8, Dorota Lebiedz-Odrobina12, Yiming Luo13, Ashima Makol14, Jerry Molitor15, Duncan Moore16, Carrie Richardson17, Nora Sandorfi18, Ami Shah19, Ankoor Shah20, Brian Skaug21, Virginia Steen22, Elizabeth Volkmann23 and Flavia Castelino1, 1Massachusetts General Hospital, Boston, MA, 2University of Utah, Salt Lake City, UT, 3University of Texas McGovern Medical School at Houston, Houston, TX, 4Division of Rheumatology, Department of Medicine, Vagelos College of Physicians and Surgeons, Columbia University Irving Medical Center, New York, NY, 5Department of Medicine, Division of Immunology and Rheumatology, Stanford University School of Medicine, Woodside, CA, 6Scleroderma Research Foundation, Brisbane, CA, 7Vanderbilt University Medical Center, Nashville, TN, 8Hospital for Special Surgery, New York, NY, 9Medical University of South Carolina, Charleston, SC, 10Johns Hopkins University, Baltimore, MD, 11University of Michigan, Ann Arbor, MI, 12University of Utah, Cottonwood Heights, UT, 13Division of Rheumatology, Department of Medicine, Columbia University Irving Medical Center, New York, NY, 14Mayo Clinic, Rochester, MN, Rochester, MN, 15University of Minnesota, Minneapolis, MN, 16Northwestern University, Department of Rheumatology, Chicago, IL, 17Northwestern University, Chicago, IL, 18University of Pennsylvania, Philadelphia, PA, 19Department of Medicine, Division of Rheumatology, Johns Hopkins University School of Medicine, Ellicott City, MD, 20Duke University, Durham, NC, 21Division of Rheumatology, University of Texas McGovern Medical School, Houston, TX, 22Georgetown University School of Medicine, Washington, DC, 23University of California Los Angeles, Los Angeles, CA
Background/Purpose: Digital ischemic ulcers (DIU) develop in 36-44% of patients with systemic sclerosis (SSc).1,2Prior international observational studies have evaluated characteristics of digital pitting and ulcers in their SSc patient populations; however, these studies do not necessarily reflect the US SSc patient population. We examined the prevalence of digital ischemic complications in a US-based longitudinal registry and evaluated clinical factors and patient reported outcomes associated with current digital pitting scars (DPS) and DIU.
Methods: We conducted a cross-sectional study utilizing the Collaborative National Quality and Efficacy Registry (CONQUER), a US-based, prospective, multi-center cohort of adults with SSc who meet 2013 ACR/EULAR Classification Criteria, with a disease duration ≤ 5 years from first non-Raynaud's symptom at enrollment. At study enrollment, clinicians recorded the presence of DPS and DIU, and demographic, clinical, laboratory variables, and patient reported outcomes were collected. Multivariable-adjusted logistic regression models were designed using directed acyclic graphs.
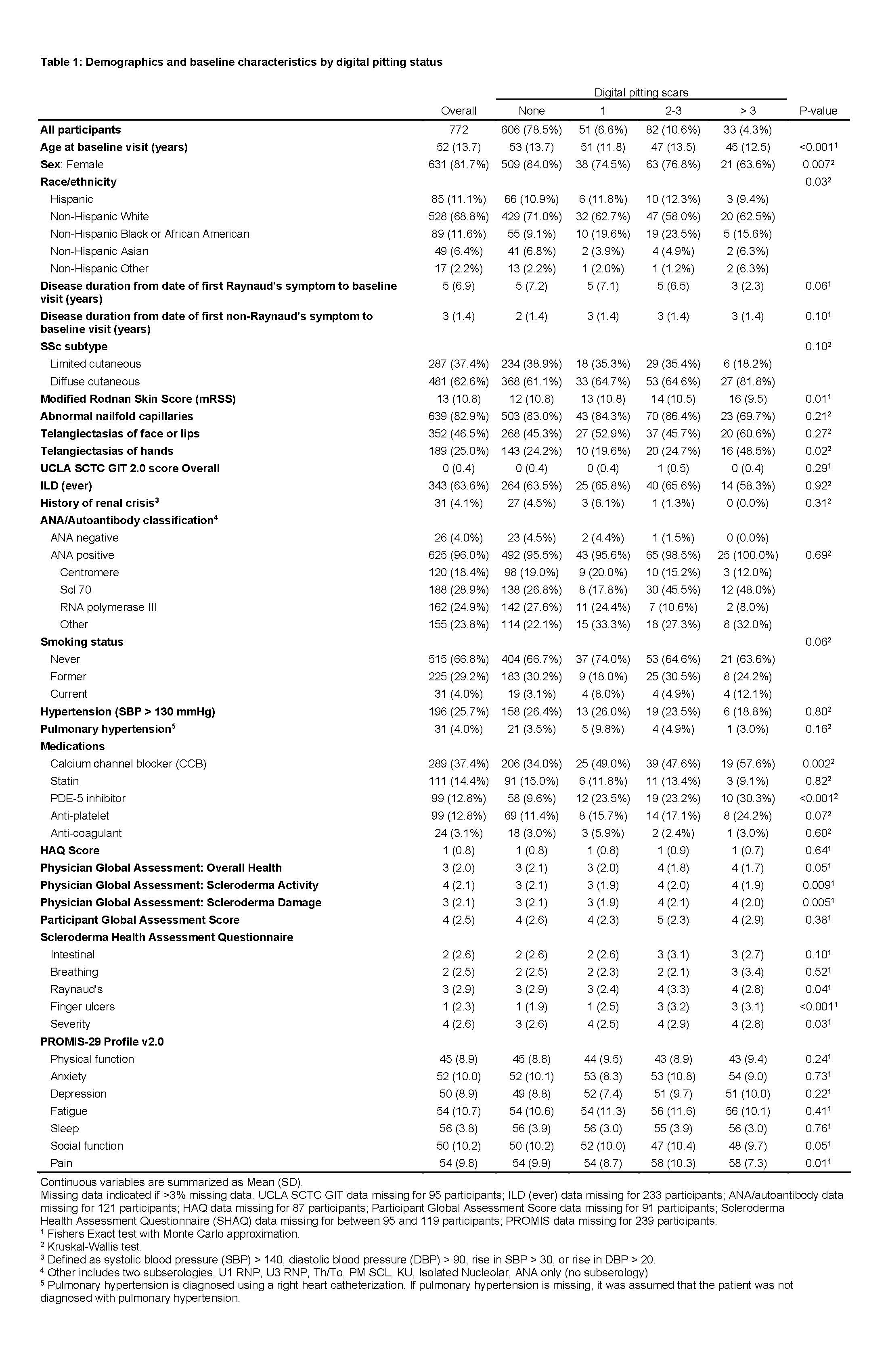
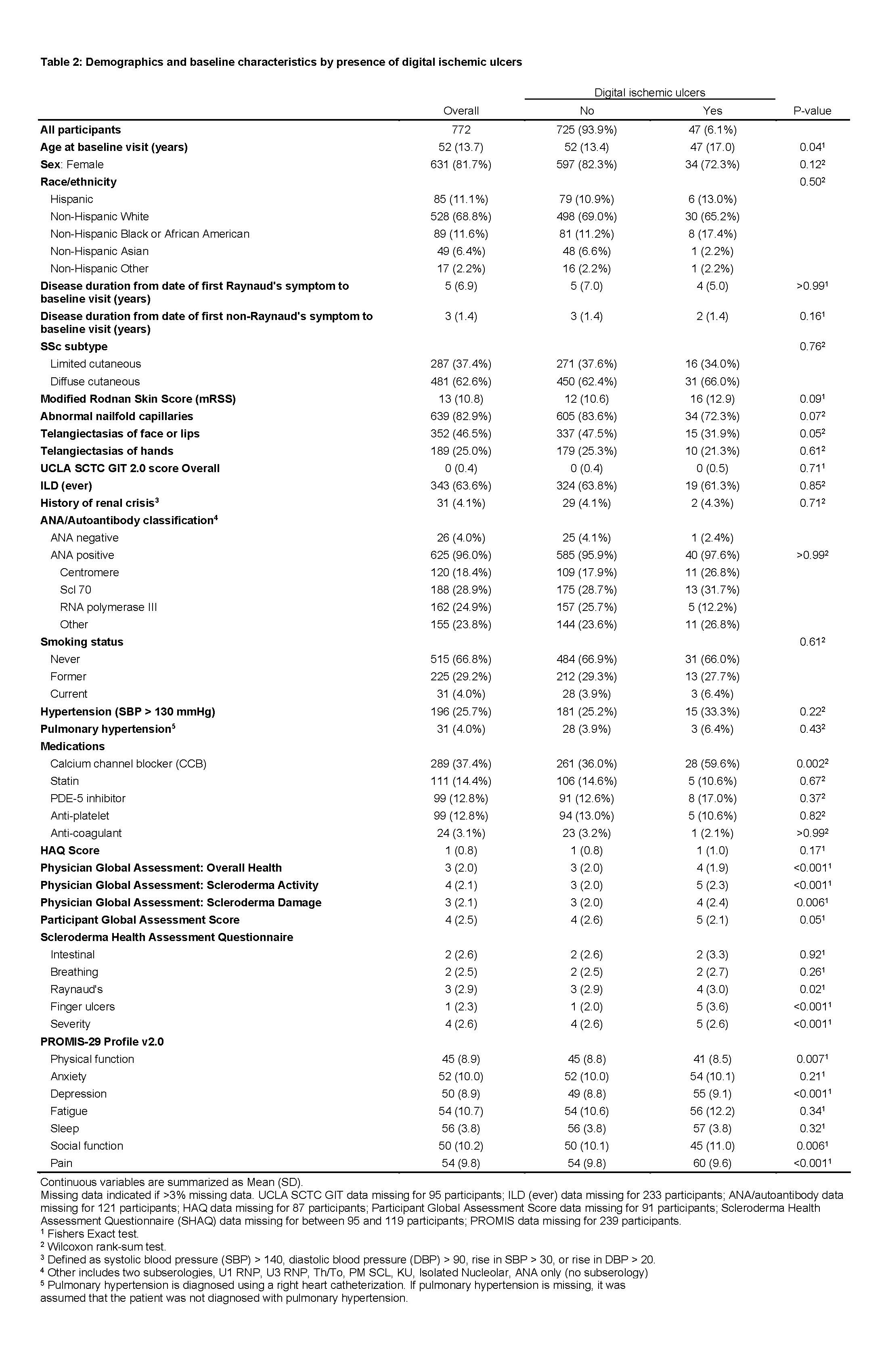
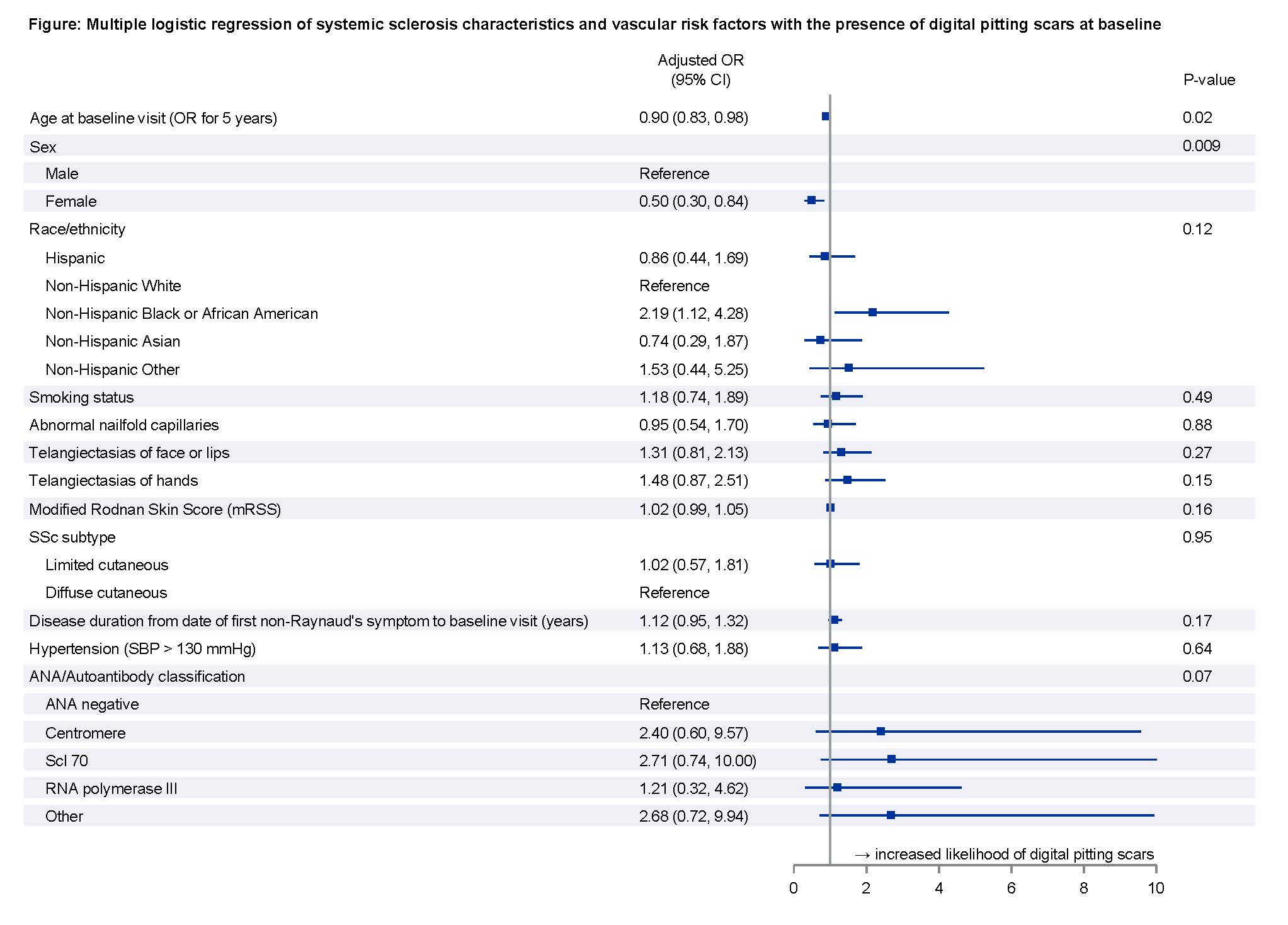
Results: Among 772 eligible CONQUER participants, 166 (22%) had at least one DPS and 47 (6%) had active DIU at enrollment (Tables 1 and 2). Half of the participants with DPS were prescribed calcium channel blockers. Participants with DPS had worse PROMIS-29 v2.0 social function and pain scores, and participants with DIU had worse physical function, depression, social function, and pain scores. In our multivariable logistic regression model, younger age and male sex were associated with DPS (OR for female sex 0.50, 95% CI: 0.30 to 0.84 and OR for increased age by 5-year increments 0.90, 95% CI: 0.83 to 0.98, Figure). Race, tobacco use, abnormal nailfold capillaries, presence of telangiectasias, mRSS, disease subtype, disease duration, hypertension, and autoantibody status were not associated with DPS.
Conclusion: Digital ischemic complications were associated with impaired quality of life among patients with SSc, specifically in the domains of social function and pain. Younger age and male sex were associated with presence of digital pitting scars.
References
M. Savoie: None; M. Harding: None; J. Alvey: None; J. VanBuren: None; S. Assassi: AstraZeneca, 2, aTyr, 2, Boehringer Ingelheim, 2, 5, CSL Behring, 2, Janssen, 5, Merck, 2, Momenta, 5, TeneoFour, 2; E. Bernstein: Boehringer Ingelheim, 2, 5, Kadmon, 5, Pfizer, 5; L. Chung: Eicos Science, 1, 2, Eli Lilly, 1, 2, Genentech, 1, 2, IgM biosciences, 1, 2, Janssen, 1, 2, Kyverna, 1, 2, Mitsubishi Tanabe, 1, 2; L. Evnin: None; T. Frech: None; J. Gordon: Cumberland Pharmaceuticals, 5, Prometheus Pharmaceuticals, 5; F. Hant: None; L. Hummers: AbbVie/Abbott, 1, Biotest, 2, Boehringer-Ingelheim, 1, 5, CSL Behring, 1, Cumberland Pharmaceuticals, 5, GlaxoSmithKlein(GSK), 5, Kadmon Corporation, 5, Medpace, 5, Mitsubishi Tanabe, 5, Prometheus, 5; D. Khanna: AbbVie, 12, DSMB, AstraZeneca, 2, Boehringer-Ingelheim, 2, Bristol-Myers Squibb, 2, 5, CSL Behring, 2, Genentech, 2, Horizon Therapeutics, 2, 5, Janssen, 2, 6, Pfizer, 5, Prometheus, 2; K. Lakin: None; D. Lebiedz-Odrobina: None; Y. Luo: None; A. Makol: Boehringer-Ingelheim, 1, Sanofi-Genzyme, 1; J. Molitor: None; D. Moore: None; C. Richardson: None; N. Sandorfi: None; A. Shah: Arena Pharmaceuticals, 5, Eicos Sciences, 5, Kadmon Corporation, 5, Medpace LLC, 5; A. Shah: None; B. Skaug: None; V. Steen: None; E. Volkmann: Boehringer-Ingelheim, 2, 5, 6, CSL Behring, 2, GlaxoSmithKline, 2, Horizon, 5, Prometheus, 5, Roche, 2; F. Castelino: Boehringer-Ingelheim, 2, Genentech, 5, Prometheus, 5.
Background/Purpose: Digital ischemic ulcers (DIU) develop in 36-44% of patients with systemic sclerosis (SSc).1,2Prior international observational studies have evaluated characteristics of digital pitting and ulcers in their SSc patient populations; however, these studies do not necessarily reflect the US SSc patient population. We examined the prevalence of digital ischemic complications in a US-based longitudinal registry and evaluated clinical factors and patient reported outcomes associated with current digital pitting scars (DPS) and DIU.
Methods: We conducted a cross-sectional study utilizing the Collaborative National Quality and Efficacy Registry (CONQUER), a US-based, prospective, multi-center cohort of adults with SSc who meet 2013 ACR/EULAR Classification Criteria, with a disease duration ≤ 5 years from first non-Raynaud's symptom at enrollment. At study enrollment, clinicians recorded the presence of DPS and DIU, and demographic, clinical, laboratory variables, and patient reported outcomes were collected. Multivariable-adjusted logistic regression models were designed using directed acyclic graphs.
Results: Among 772 eligible CONQUER participants, 166 (22%) had at least one DPS and 47 (6%) had active DIU at enrollment (Tables 1 and 2). Half of the participants with DPS were prescribed calcium channel blockers. Participants with DPS had worse PROMIS-29 v2.0 social function and pain scores, and participants with DIU had worse physical function, depression, social function, and pain scores. In our multivariable logistic regression model, younger age and male sex were associated with DPS (OR for female sex 0.50, 95% CI: 0.30 to 0.84 and OR for increased age by 5-year increments 0.90, 95% CI: 0.83 to 0.98, Figure). Race, tobacco use, abnormal nailfold capillaries, presence of telangiectasias, mRSS, disease subtype, disease duration, hypertension, and autoantibody status were not associated with DPS.
Conclusion: Digital ischemic complications were associated with impaired quality of life among patients with SSc, specifically in the domains of social function and pain. Younger age and male sex were associated with presence of digital pitting scars.
References
- Khimdas S, Harding S, Bonner A, et al. Associations with digital ulcers in a large cohort of systemic sclerosis: results from the Canadian Scleroderma Research Group registry. Arthritis Care Res (Hoboken) 2011;63(1):142–149.
- Meier FMP, Frommer KW, Dinser R, et al. Update on the profile of the EUSTAR cohort: an analysis of the EULAR Scleroderma Trials and Research group database. Ann Rheum Dis 2012;71(8):1355–1360.

Table 1: Demographics and baseline characteristics by digital pitting status

Table 2: Demographics and baseline characteristics by presence of digital ischemic ulcers

Figure: Multiple logistic regression of systemic sclerosis characteristics and vascular risk factors with the presence of digital pitting scars at baseline
M. Savoie: None; M. Harding: None; J. Alvey: None; J. VanBuren: None; S. Assassi: AstraZeneca, 2, aTyr, 2, Boehringer Ingelheim, 2, 5, CSL Behring, 2, Janssen, 5, Merck, 2, Momenta, 5, TeneoFour, 2; E. Bernstein: Boehringer Ingelheim, 2, 5, Kadmon, 5, Pfizer, 5; L. Chung: Eicos Science, 1, 2, Eli Lilly, 1, 2, Genentech, 1, 2, IgM biosciences, 1, 2, Janssen, 1, 2, Kyverna, 1, 2, Mitsubishi Tanabe, 1, 2; L. Evnin: None; T. Frech: None; J. Gordon: Cumberland Pharmaceuticals, 5, Prometheus Pharmaceuticals, 5; F. Hant: None; L. Hummers: AbbVie/Abbott, 1, Biotest, 2, Boehringer-Ingelheim, 1, 5, CSL Behring, 1, Cumberland Pharmaceuticals, 5, GlaxoSmithKlein(GSK), 5, Kadmon Corporation, 5, Medpace, 5, Mitsubishi Tanabe, 5, Prometheus, 5; D. Khanna: AbbVie, 12, DSMB, AstraZeneca, 2, Boehringer-Ingelheim, 2, Bristol-Myers Squibb, 2, 5, CSL Behring, 2, Genentech, 2, Horizon Therapeutics, 2, 5, Janssen, 2, 6, Pfizer, 5, Prometheus, 2; K. Lakin: None; D. Lebiedz-Odrobina: None; Y. Luo: None; A. Makol: Boehringer-Ingelheim, 1, Sanofi-Genzyme, 1; J. Molitor: None; D. Moore: None; C. Richardson: None; N. Sandorfi: None; A. Shah: Arena Pharmaceuticals, 5, Eicos Sciences, 5, Kadmon Corporation, 5, Medpace LLC, 5; A. Shah: None; B. Skaug: None; V. Steen: None; E. Volkmann: Boehringer-Ingelheim, 2, 5, 6, CSL Behring, 2, GlaxoSmithKline, 2, Horizon, 5, Prometheus, 5, Roche, 2; F. Castelino: Boehringer-Ingelheim, 2, Genentech, 5, Prometheus, 5.



