Abstract Session
Vasculitis
Session: Abstracts: Vasculitis – Non-ANCA-Associated & Related Disorders III: Innovation (2599–2604)
2603: Impact of IL-6 Receptor Small Nucleotide Polymorphism Asp358Ala on T Cell Activity and Clinical Outcomes in Patients with Giant Cell Arteritis
Wednesday, November 15, 2023
12:00 PM - 12:10 PM PT
Location: Room 33A-C
- CR
Christopher Redmond, MD, MSc (he/him/his)
West Virginia University
Hagerstown, MD, United StatesDisclosure(s): No financial relationships with ineligible companies to disclose
Presenting Author(s)
Christopher Redmond1, Robert Zorc2, McKella Sylvester3, Cameron Rankin4, Rebecca Kuan5, Kristina Wells6, Lily Dai7, Kaitlin Quinn8, Massimo Gadina9 and Peter Grayson10, 1National Institutes of Health, Hagerstown, MD, 2National Institute of Arthritis and Musculoskeletal and Skin Diseases/National Institutes of Health, Bethesda, MD, 3Colgate University, Bethesda, MD, 4NIAMS, NIH, Washington, DC, 5NIH, Bethesda, MD, 6University of California Davis, Bethesda, MD, 7NIH, Germantown, MD, 8National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), National Institutes of Health (NIH), Bethesda, MD, 9National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, MD, 10National Institutes of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), National Institutes of Health (NIH), Chevy Chase, MD
Background/Purpose: Giant cell arteritis (GCA) is a large vessel vasculitis in adults that commonly involves the aorta and branching arteries, resulting in multiple symptoms including vision loss. IL-6 has been identified as key to GCA pathogenesis, and treatment with IL6 receptor (IL-6R) antibody, tocilizumab, induces steroid-free disease remission in many patients with GCA. Not all patients with GCA have favorable clinical response to tocilizumab. A relatively common SNP in IL-6R, Asp358Ala, results in increased solubilization of the IL-6R that then binds the ubiquitous IL-6 co-receptor, gp130, leading to more pro-inflammatory soluble IL-6 signaling, known as trans signaling. This contrasts with classical membrane bound IL-6R signaling, found on hepatocytes and lymphocytes, that results in anti-inflammatory effects and release of C-reactive protein. We hypothesized that the Asp358Ala IL-6R variant in patients with GCA impact IL-6 mediated T cell activity and response to tocilizumab.
Methods: Samples and clinical data were obtained from patients who met the 2022 ACR/EULAR Classification Criteria for GCA. Genetic sequencing was completed to identify GCA patients with the Asp358Ala variant. Serum from patients was used to quantitate serum soluble IL-6R levels by ELISA. Peripheral blood mononuclear cells (PBMCs) evaluated for expression of IL-6R and its co-receptor, gp130, using flow cytometry. The same PBMCs were stimulated ex vivo with IL-6 and evaluated for downstream targets of IL-6, STAT3 phosphorylation and IL-17A expression, also using flow cytometry. Clinical symptoms, laboratory values, and response to tocilizumab was identified by chart review. Active disease was defined by clinical signs and symptoms.
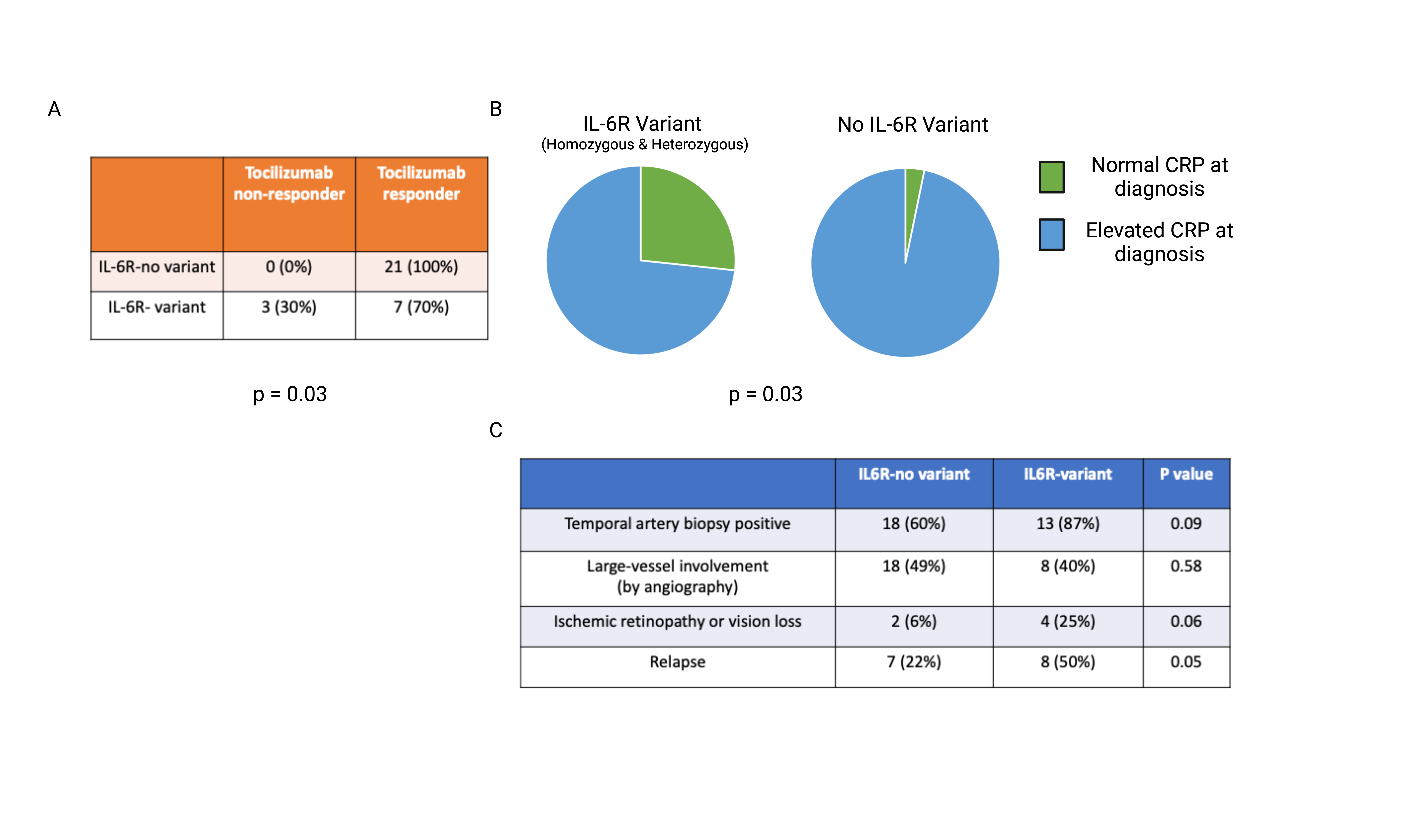
Results: Patients with GCA and the Asp358Ala IL-6R variant had increased serum soluble IL-6 receptor levels (Fig 1A) as well as higher lymphocyte expression of surface gp130 (Fig 1B). When stimulated ex vivo with IL-6, CD4+ T cells in patients with Asp358Ala had increased STAT3 phosphorylation and IL17A expression (Fig 1C and 1D). Similar associations were not seen in other leukocyte subsets or in CD4+ T cells from healthy controls by variant status. Clinically, patients with the Asp358Ala SNP had a higher likelihood of treatment failure to tocilizumab (Fig 2A), lower CRP values at diagnosis (Fig 2B), and a higher rate of relapse (Fig 2C).
Conclusion: The Asp358Ala IL6R variant may result in increased pro-inflammatory trans IL-6 signaling via increased soluble IL-6R and surface gp130 expression, leading to more robust STAT3 phosphorylation and IL-17A production in T cells. IL-6R inhibition via tocilizumab in patients with the Asp358Ala variant may be insufficient to achieve disease control. This variant may be a useful biomarker to predict treatment response to tocilizumab and to identify patients who may respond more favorably to alternative therapeutic approaches.
.jpg)
C. Redmond: None; R. Zorc: None; M. Sylvester: None; C. Rankin: None; R. Kuan: Colgate-Palmolive Company, 5; K. Wells: None; L. Dai: None; K. Quinn: None; M. Gadina: None; P. Grayson: None.
Background/Purpose: Giant cell arteritis (GCA) is a large vessel vasculitis in adults that commonly involves the aorta and branching arteries, resulting in multiple symptoms including vision loss. IL-6 has been identified as key to GCA pathogenesis, and treatment with IL6 receptor (IL-6R) antibody, tocilizumab, induces steroid-free disease remission in many patients with GCA. Not all patients with GCA have favorable clinical response to tocilizumab. A relatively common SNP in IL-6R, Asp358Ala, results in increased solubilization of the IL-6R that then binds the ubiquitous IL-6 co-receptor, gp130, leading to more pro-inflammatory soluble IL-6 signaling, known as trans signaling. This contrasts with classical membrane bound IL-6R signaling, found on hepatocytes and lymphocytes, that results in anti-inflammatory effects and release of C-reactive protein. We hypothesized that the Asp358Ala IL-6R variant in patients with GCA impact IL-6 mediated T cell activity and response to tocilizumab.
Methods: Samples and clinical data were obtained from patients who met the 2022 ACR/EULAR Classification Criteria for GCA. Genetic sequencing was completed to identify GCA patients with the Asp358Ala variant. Serum from patients was used to quantitate serum soluble IL-6R levels by ELISA. Peripheral blood mononuclear cells (PBMCs) evaluated for expression of IL-6R and its co-receptor, gp130, using flow cytometry. The same PBMCs were stimulated ex vivo with IL-6 and evaluated for downstream targets of IL-6, STAT3 phosphorylation and IL-17A expression, also using flow cytometry. Clinical symptoms, laboratory values, and response to tocilizumab was identified by chart review. Active disease was defined by clinical signs and symptoms.
Results: Patients with GCA and the Asp358Ala IL-6R variant had increased serum soluble IL-6 receptor levels (Fig 1A) as well as higher lymphocyte expression of surface gp130 (Fig 1B). When stimulated ex vivo with IL-6, CD4+ T cells in patients with Asp358Ala had increased STAT3 phosphorylation and IL17A expression (Fig 1C and 1D). Similar associations were not seen in other leukocyte subsets or in CD4+ T cells from healthy controls by variant status. Clinically, patients with the Asp358Ala SNP had a higher likelihood of treatment failure to tocilizumab (Fig 2A), lower CRP values at diagnosis (Fig 2B), and a higher rate of relapse (Fig 2C).
Conclusion: The Asp358Ala IL6R variant may result in increased pro-inflammatory trans IL-6 signaling via increased soluble IL-6R and surface gp130 expression, leading to more robust STAT3 phosphorylation and IL-17A production in T cells. IL-6R inhibition via tocilizumab in patients with the Asp358Ala variant may be insufficient to achieve disease control. This variant may be a useful biomarker to predict treatment response to tocilizumab and to identify patients who may respond more favorably to alternative therapeutic approaches.
.jpg)
Figure 1. A) Serum samples from GCA patients were measured by ELISA for soluble IL-6R levels. sIL-6R levels from patients with the Asp358Ala variant (homozygous and heterozygous) were compared to those without the variant. B) PBMCs were analyzed by flow cytometry for gp130 expression on different T cell subsets. C) PBMCs were stimulated with IL-6 (100ng/ml) for 15mins and then analyzed for STAT3 phosphorylation. C) PBMCs from patients with GCA were stimulated with 50ng/ml of IL-6 and costimulatory molecules (CD3, CD28, anti-IL4, and anti-IFNy) for 3 days and then re-stimulated with PMA/Ionomycin.

Figure 2. Clinical characteristics of a prospective observational cohort of patients with GCA. A) Tocilizumab non-response was identified as clinical signs or symptoms of active disease following treatment with tocilizumab. Response and non-response was compared between GCA patients with and without the IL6-R variant (Asp358Ala) B) CRP at time of diagnosis was compared between GCA patients with and without the IL6-R variant (Asp358Ala), normal range was determined by clinical laboratory standards. 3) Prospective chart review as conducted to identify clinical characteristics and complications commonly associated with GCA, before, during, or after treatment with tocilizumab.
C. Redmond: None; R. Zorc: None; M. Sylvester: None; C. Rankin: None; R. Kuan: Colgate-Palmolive Company, 5; K. Wells: None; L. Dai: None; K. Quinn: None; M. Gadina: None; P. Grayson: None.



