Abstract Session
Periodic fever syndromes, autoinflammatory diseases, Still’s disease and MAS/HLH
Session: Abstracts: Miscellaneous Rheumatic & Inflammatory Diseases III (2569–2574)
2574: Are Patients Classified as IPAF More Likely to Also Meet SLE Criteria by 2019 EULAR/ACR Than by SLICC?: An Abstract
Wednesday, November 15, 2023
10:30 AM - 10:40 AM PT
Location: Room 32A-B

Gabriela Martinez Zayas, MD (she/her/hers)
UT Southwestern Medical Center
Dallas, TX, United StatesDisclosure information not submitted.
Presenting Author(s)
Gabriela Martinez Zayas1, David Karp1 and Elena Joerns2, 1UT Southwestern Medical Center, Dallas, TX, 2University of Texas Southwestern Medical Center, Dallas, TX
Background/Purpose: Interstitial pneumonia with autoimmune features (IPAF) is a research classification proposed by the European Respiratory Society and American Thoracic Society Task Force for those patients with forms of interstitial lung disease (ILD) that have autoimmune features but that do not classify as having systemic rheumatic diseases such as systemic lupus erythematous (SLE).(1)
In 2012 the Systemic Lupus International Collaborating Clinics (SLICC) published criteria to identify and classify patients with SLE. (2) Subsequently, in 2019 the European Alliance of Association of Rheumatology/American College of Rheumatology (EULAR/ACR) published updated criteria SLE criteria. Sensitivities were similar, but 2019 EULAR/ACR criteria had superior specificity. (3)
We hypothesized that patients initially classified as IPAF may meet SLE by 2019 ACR/EULAR criteria more frequently than by SLICC criteria. The purpose of our study was to review data of patients classified as IPAF and assess if there is a difference in classification of SLE by both sets of criteria.
Methods: This observational, single-center cohort study included consecutive patients who had initially been classified as having IPAF between December 1st, 2005-August 31st, 2019. Two authors (GM-Z, EKJ) independently and retrospectively reviewed patients' charts and assigned SLE criteria by SLICC or 2019 EULAR/ACR classifications. Fisher's exact test was used to assess for significant difference in number of patients' meeting SLE criteria by SLICC criteria versus 2019 EULAR/ACR criteria.
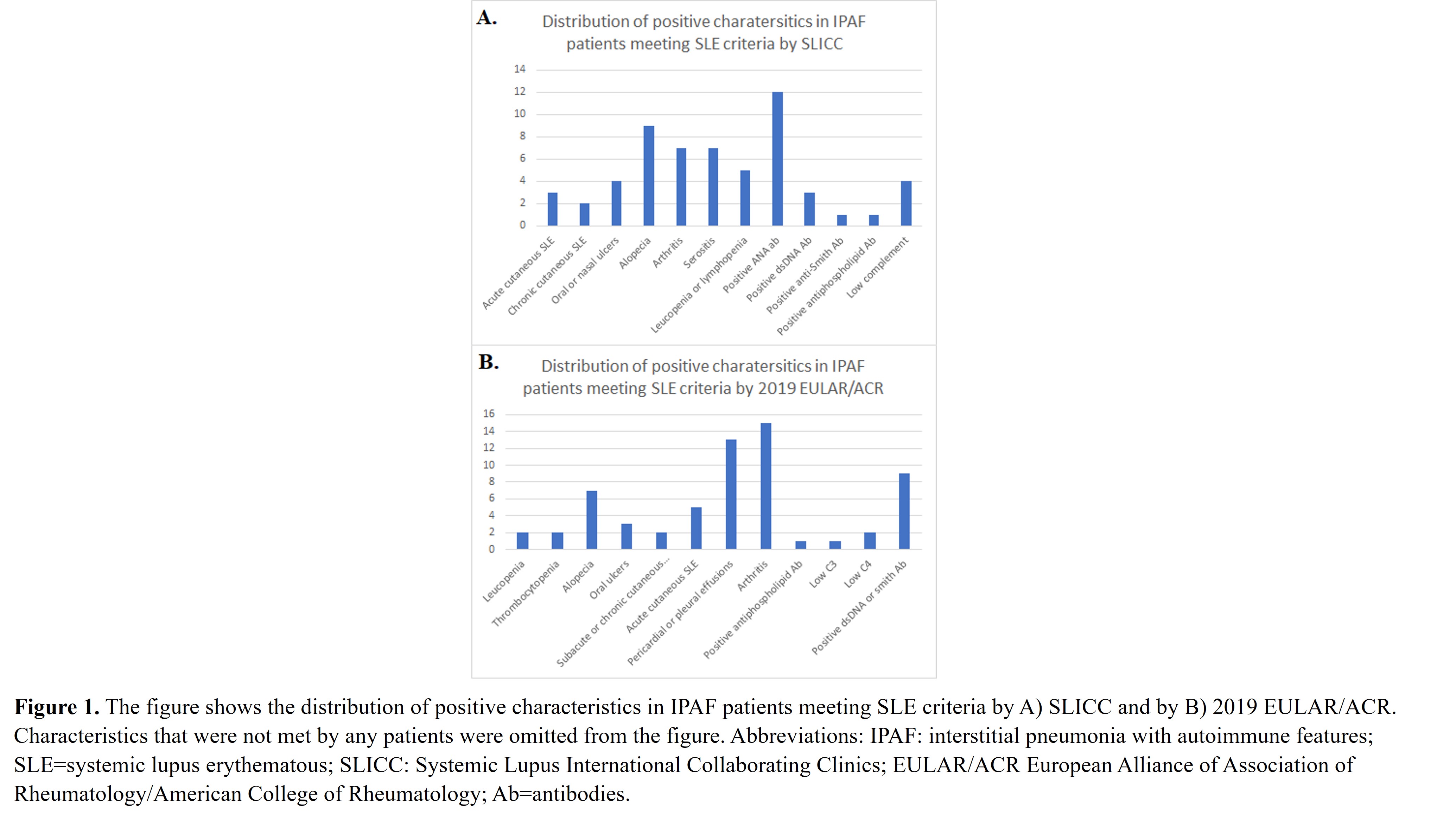
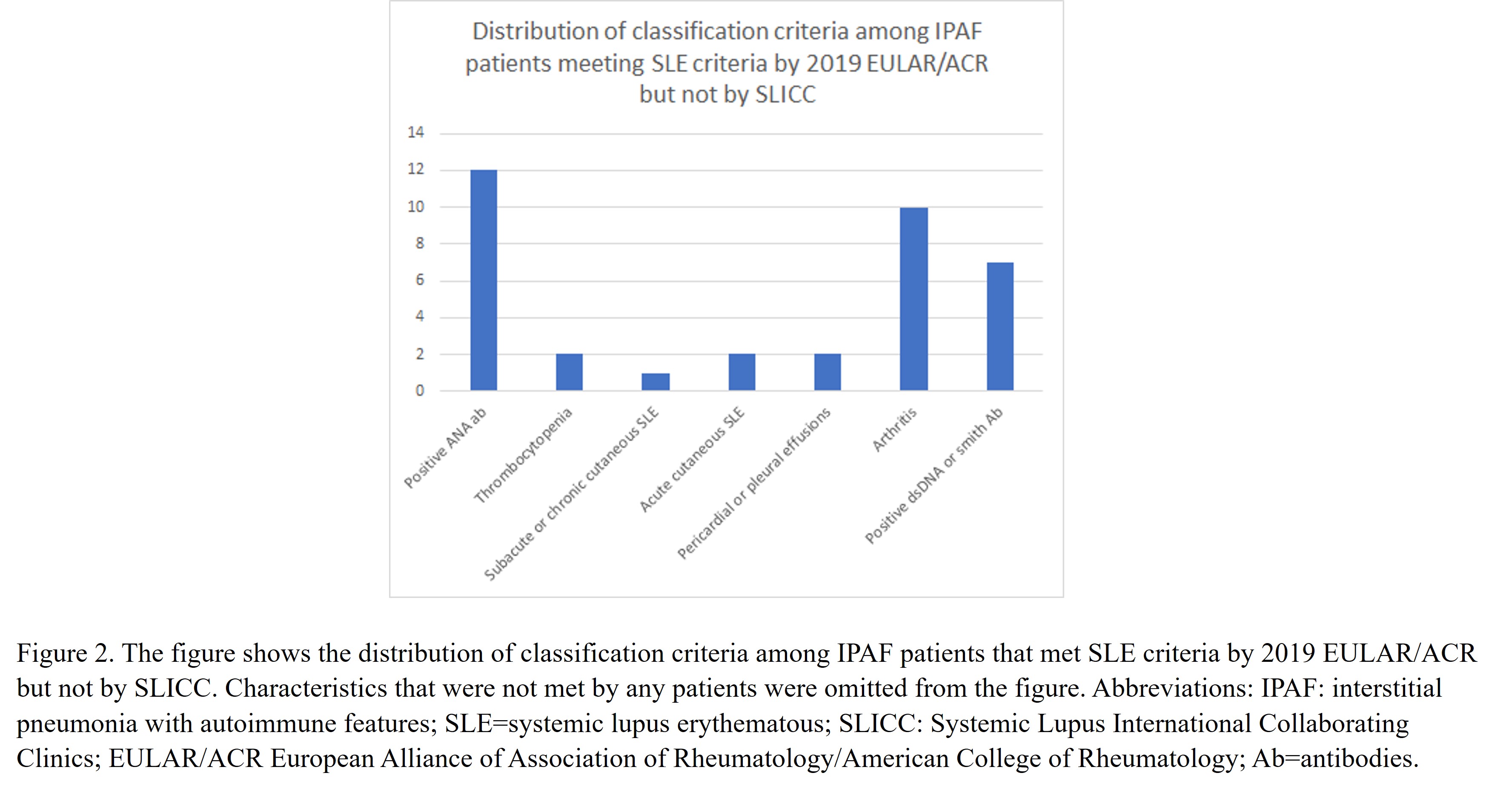
Results: A total of 197 patients that had been initially classified as IPAF were included. Out of these 197 patients, 12 met SLE criteria by SLICC and 23 by 2019 EULAR/ACR. All but three patients that met SLE criteria SLICC also met 2019 EULAR/ACR criteria. The difference in eligibility for these three patients was due to lymphopenia (Figure 1). The most frequent SLE inclusion characteristics by either criteria and by 2019 EULAR/ACR criteria alone are shown in Figures 1 and 2, respectively. The difference in meeting SLE criteria between groups was statistically significant (p< 0.001, Table 1).
Conclusion: The difference in the number of patients originally classified as IPAF that met SLE criteria by SLICC and by 2019 EULAR/ACR upon re-review in 2023 was statistically significant (12 vs 23 patients, respectively, p < 0.001, Table 1). Based on our results, we conclude that 2019 ACR/EULAR criteria are superior for SLE classification in patients with ILD. ILD may be an underrecognized manifestation of SLE. Patients with ILD who have features seen in SLE, particularly dsDNA/Smith, arthritis and serositis should undergo full work-up to evaluate for other SLE clinical characteristics.

G. Martinez Zayas: None; D. Karp: Ample, 2, Biogen, 5, Bristol myers, 5, Celgene, 5, Eli Lilly, 5, Genetech, 5, Prevention Bio, 1, Rilite, 5, UCB, 5; E. Joerns: None.
Background/Purpose: Interstitial pneumonia with autoimmune features (IPAF) is a research classification proposed by the European Respiratory Society and American Thoracic Society Task Force for those patients with forms of interstitial lung disease (ILD) that have autoimmune features but that do not classify as having systemic rheumatic diseases such as systemic lupus erythematous (SLE).(1)
In 2012 the Systemic Lupus International Collaborating Clinics (SLICC) published criteria to identify and classify patients with SLE. (2) Subsequently, in 2019 the European Alliance of Association of Rheumatology/American College of Rheumatology (EULAR/ACR) published updated criteria SLE criteria. Sensitivities were similar, but 2019 EULAR/ACR criteria had superior specificity. (3)
We hypothesized that patients initially classified as IPAF may meet SLE by 2019 ACR/EULAR criteria more frequently than by SLICC criteria. The purpose of our study was to review data of patients classified as IPAF and assess if there is a difference in classification of SLE by both sets of criteria.
Methods: This observational, single-center cohort study included consecutive patients who had initially been classified as having IPAF between December 1st, 2005-August 31st, 2019. Two authors (GM-Z, EKJ) independently and retrospectively reviewed patients' charts and assigned SLE criteria by SLICC or 2019 EULAR/ACR classifications. Fisher's exact test was used to assess for significant difference in number of patients' meeting SLE criteria by SLICC criteria versus 2019 EULAR/ACR criteria.
Results: A total of 197 patients that had been initially classified as IPAF were included. Out of these 197 patients, 12 met SLE criteria by SLICC and 23 by 2019 EULAR/ACR. All but three patients that met SLE criteria SLICC also met 2019 EULAR/ACR criteria. The difference in eligibility for these three patients was due to lymphopenia (Figure 1). The most frequent SLE inclusion characteristics by either criteria and by 2019 EULAR/ACR criteria alone are shown in Figures 1 and 2, respectively. The difference in meeting SLE criteria between groups was statistically significant (p< 0.001, Table 1).
Conclusion: The difference in the number of patients originally classified as IPAF that met SLE criteria by SLICC and by 2019 EULAR/ACR upon re-review in 2023 was statistically significant (12 vs 23 patients, respectively, p < 0.001, Table 1). Based on our results, we conclude that 2019 ACR/EULAR criteria are superior for SLE classification in patients with ILD. ILD may be an underrecognized manifestation of SLE. Patients with ILD who have features seen in SLE, particularly dsDNA/Smith, arthritis and serositis should undergo full work-up to evaluate for other SLE clinical characteristics.

Figure 1. The figure shows the distribution of positive characteristics in IPAF patients meeting SLE criteria by A) SLICC and by B) 2019 EULAR/ACR

Figure 2. The figure shows the distribution of classification criteria among IPAF patients that met SLE criteria by 2019 EULAR/ACR but not by SLICC.

Table 1. IPAF Patients Meeting SLE Criteria by SLICC and 2019 EULAR/ACR.
G. Martinez Zayas: None; D. Karp: Ample, 2, Biogen, 5, Bristol myers, 5, Celgene, 5, Eli Lilly, 5, Genetech, 5, Prevention Bio, 1, Rilite, 5, UCB, 5; E. Joerns: None.



