Abstract Session
Periodic fever syndromes, autoinflammatory diseases, Still’s disease and MAS/HLH
Session: Abstracts: Miscellaneous Rheumatic & Inflammatory Diseases III (2569–2574)
2570: Thrombosis in Patients with VEXAS Syndrome: A Retrospective Cohort Study
Wednesday, November 15, 2023
9:30 AM - 9:40 AM PT
Location: Room 32A-B
- AG
Atefeh Ghorbanzadeh, MD (she/her/hers)
Mayo Clinic
Rochester, MN, United StatesDisclosure information not submitted.
Presenting Author(s)
Atefeh Ghorbanzadeh1, Yael Kusne2, Alina Dulau-Florea3, Ruba Shalhoub3, Pedro Alcedo3, Khanh Nghiem3, Marcela Ferrada4, alexander hines1, Sumith Panicker3, amanda ombrello5, Kaaren K. Reichard1, Ivana Darden3, Wendy Goodspeed3, Lorena wilson3, Horatiu Olteanu1, Terra L. Lasho1, Daniel Kastner6, Kenneth Warrington1, Abhishek A. Mangaonkar1, Ronald Go1, David Beck7, Mrinal S. Patnaik1, Neal S. Young8, Katherine R. Calvo3, Ana Casanegra9, Peter Grayson10, Matthew Koster1, Colin O Wu3, Damon E. Houghton9, Bhavisha Patel11, Yogen Kanthi12 and Emma M. Groarke13, 1Mayo Clinic, Rochester, MN, 2Mayo Clinic, Phoenix, AZ, 3National Institutes of Health (NIH), Bethesda, MD, 4National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), National Institutes of Health (NIH), Bethesda, MD, 5National Institutes of Health, Rockville, MD, 6National Human Genome Research Institute, Bethesda, MD, 7New York University, New York, NY, 8National Institutes of Health (NIH), Rochester, MD, 9Mayo Clinic, Rochester, MD, 10National Institutes of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), National Institutes of Health (NIH), Chevy Chase, MD, 11National Institutes of Health, Beltsville, MD, 12National Heart, Lung and Blood Institute, National Institutes of Health, Bethesda, MD, 13Hematology Branch, National Heart, Lung, and Blood Institute, National Institutes of Health (NIH), Bethesda, MD
Background/Purpose: VEXAS (vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic) syndrome, due to somatic mutations in the UBA1 gene, is an autoinflammatory disorder associated with an increased thrombotic risk. We investigated the frequency and characteristics of thrombotic events in patients with VEXAS syndrome, as well as their impact on survival.
Methods: This retrospective, multi-center study utilized electronic medical records from the National Institutes of Health (NIH) Clinical Center and Mayo Clinic, along with referred outside records, to identify patients with VEXAS syndrome who had a confirmed UBA1 somatic mutation and VEXAS clinical features. The time of symptom onset was used as the time of disease onset. Thrombosis was considered VEXAS disease onset if it occurred within two years of inflammatory symptom onset. SVT was excluded as a thrombotic event for analysis. Univariate and multivariate logistic regression analysis was used to identify predictors of thrombosis. Kaplan-Meier curve models were applied to estimate overall survival (OS). Statistical significance was P < .05. Lupus anticoagulant (LA) was assessed in 41 patients and other coagulation assays in 27.
Results: A total of 119 VEXAS patients were included. All patients (100%) were male with a median age of 65 years (39-86 years). Hematologic manifestations included macrocytic anemia in 107 (90%), myelodysplastic syndrome (MDS) in 36 (30%), and plasma cell dyscrasia (PCD) in 21 (18%).
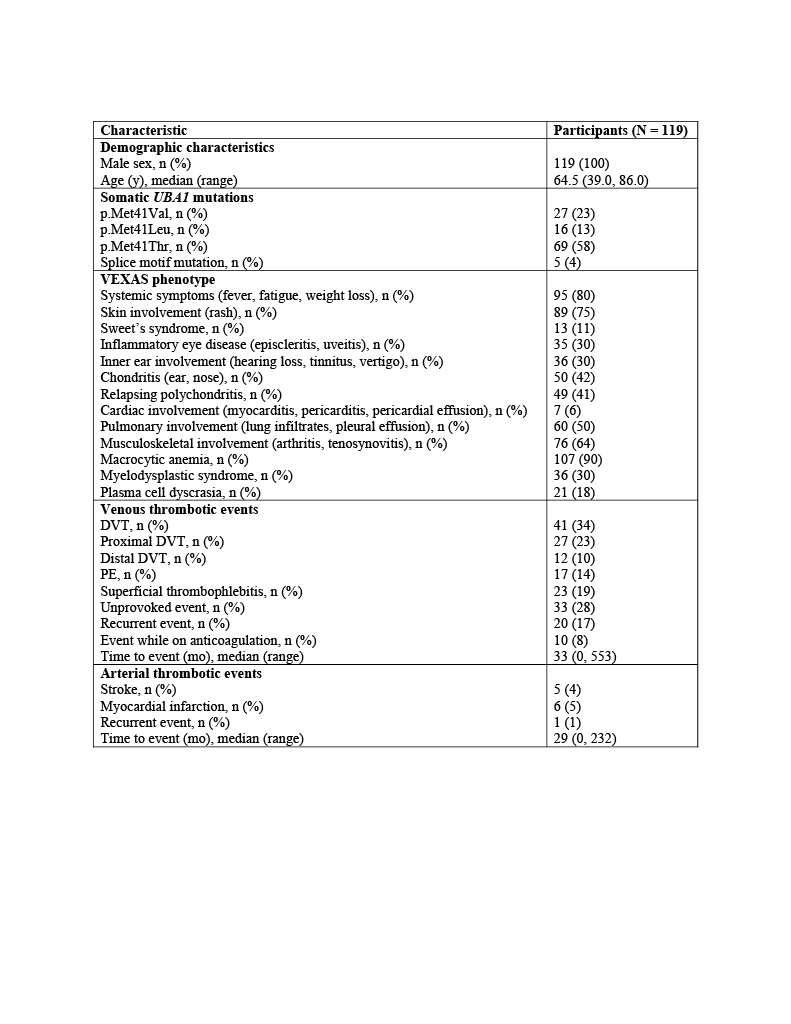
Of the 119 patients, 58/119 (49%) had a thrombotic event. Of those, 51 (43%) had VTE, and 9 (8%) had arterial events (AT), with a median time from disease onset of 33 and 29 months, respectively; seven patients (6%) had both venous and AT. The majority of VTE events were unprovoked 33 (28%) and over one-third 20 (17%) were recurrent, and 10 (8%) on anticoagulation (Table 1).
The cumulative incidence (CI) at a median follow-up of DVT, PE, and arterial thrombosis was 28.7%, 9.7%, and 8.7% respectively. CI at median follow-up of any thrombotic event was 22% for UBA1 M41L, 40% for M41T, and 37% for M41V, however, rising CI was seen with M41L over time while occurring early in M41T and M41V (Figure 1). The M41L UBA1 variant was positively associated with PE in univariate (OR: 4.58; p=0.02) and multivariate (OR: 16.94; p = 0.01) analyses but not with DVT or arterial thrombosis. The median OS from disease onset was 1740 days with no significant difference in OS between patients with (86.9%) or without (89.7%) thrombosis (Figure 2). In the univariate and multivariate Cox proportional hazard model, thrombosis was not associated with worse OS. The majority of VEXAS patients had high factor VIII levels (26/27; 96%) and VWF activity (16/27; 59%), while most had high VWF antigen (11/25; 44%), factor IX levels (12/27; 44%) and protein C activity (11/27; 41%). CRP and ESR were positively correlated with D-dimer and negatively correlated with VWF activity. LA was positive in 16/41 (40%) and correlated significantly with higher CRP (p< 0.01).
Conclusion: Patients with VEXAS syndrome are at high risk of VTE, which can recur despite anticoagulation therapy. Considering the high prevalence of thrombosis in VEXAS patients, it is crucial to assess their VTE risk carefully and consider prophylactic anticoagulation.
.jpg)
.jpg)
A. Ghorbanzadeh: None; Y. Kusne: None; A. Dulau-Florea: None; R. Shalhoub: None; P. Alcedo: None; K. Nghiem: None; M. Ferrada: None; a. hines: None; S. Panicker: None; a. ombrello: None; K. Reichard: None; I. Darden: None; W. Goodspeed: None; L. wilson: None; H. Olteanu: None; T. Lasho: None; D. Kastner: None; K. Warrington: Bristol-Myers Squibb(BMS), 5, Chemocentryx, 1, 6, Eli Lilly, 5, kiniksa, 5; A. Mangaonkar: None; R. Go: None; D. Beck: None; M. Patnaik: None; N. Young: None; K. Calvo: None; A. Casanegra: None; P. Grayson: None; M. Koster: None; C. Wu: None; D. Houghton: None; B. Patel: None; Y. Kanthi: None; E. Groarke: None.
Background/Purpose: VEXAS (vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic) syndrome, due to somatic mutations in the UBA1 gene, is an autoinflammatory disorder associated with an increased thrombotic risk. We investigated the frequency and characteristics of thrombotic events in patients with VEXAS syndrome, as well as their impact on survival.
Methods: This retrospective, multi-center study utilized electronic medical records from the National Institutes of Health (NIH) Clinical Center and Mayo Clinic, along with referred outside records, to identify patients with VEXAS syndrome who had a confirmed UBA1 somatic mutation and VEXAS clinical features. The time of symptom onset was used as the time of disease onset. Thrombosis was considered VEXAS disease onset if it occurred within two years of inflammatory symptom onset. SVT was excluded as a thrombotic event for analysis. Univariate and multivariate logistic regression analysis was used to identify predictors of thrombosis. Kaplan-Meier curve models were applied to estimate overall survival (OS). Statistical significance was P < .05. Lupus anticoagulant (LA) was assessed in 41 patients and other coagulation assays in 27.
Results: A total of 119 VEXAS patients were included. All patients (100%) were male with a median age of 65 years (39-86 years). Hematologic manifestations included macrocytic anemia in 107 (90%), myelodysplastic syndrome (MDS) in 36 (30%), and plasma cell dyscrasia (PCD) in 21 (18%).
Of the 119 patients, 58/119 (49%) had a thrombotic event. Of those, 51 (43%) had VTE, and 9 (8%) had arterial events (AT), with a median time from disease onset of 33 and 29 months, respectively; seven patients (6%) had both venous and AT. The majority of VTE events were unprovoked 33 (28%) and over one-third 20 (17%) were recurrent, and 10 (8%) on anticoagulation (Table 1).
The cumulative incidence (CI) at a median follow-up of DVT, PE, and arterial thrombosis was 28.7%, 9.7%, and 8.7% respectively. CI at median follow-up of any thrombotic event was 22% for UBA1 M41L, 40% for M41T, and 37% for M41V, however, rising CI was seen with M41L over time while occurring early in M41T and M41V (Figure 1). The M41L UBA1 variant was positively associated with PE in univariate (OR: 4.58; p=0.02) and multivariate (OR: 16.94; p = 0.01) analyses but not with DVT or arterial thrombosis. The median OS from disease onset was 1740 days with no significant difference in OS between patients with (86.9%) or without (89.7%) thrombosis (Figure 2). In the univariate and multivariate Cox proportional hazard model, thrombosis was not associated with worse OS. The majority of VEXAS patients had high factor VIII levels (26/27; 96%) and VWF activity (16/27; 59%), while most had high VWF antigen (11/25; 44%), factor IX levels (12/27; 44%) and protein C activity (11/27; 41%). CRP and ESR were positively correlated with D-dimer and negatively correlated with VWF activity. LA was positive in 16/41 (40%) and correlated significantly with higher CRP (p< 0.01).
Conclusion: Patients with VEXAS syndrome are at high risk of VTE, which can recur despite anticoagulation therapy. Considering the high prevalence of thrombosis in VEXAS patients, it is crucial to assess their VTE risk carefully and consider prophylactic anticoagulation.

Table 1. Demographic and clinical characteristics of patients with VEXAS syndrome and thrombotic events
.jpg)
.jpg)
A. Ghorbanzadeh: None; Y. Kusne: None; A. Dulau-Florea: None; R. Shalhoub: None; P. Alcedo: None; K. Nghiem: None; M. Ferrada: None; a. hines: None; S. Panicker: None; a. ombrello: None; K. Reichard: None; I. Darden: None; W. Goodspeed: None; L. wilson: None; H. Olteanu: None; T. Lasho: None; D. Kastner: None; K. Warrington: Bristol-Myers Squibb(BMS), 5, Chemocentryx, 1, 6, Eli Lilly, 5, kiniksa, 5; A. Mangaonkar: None; R. Go: None; D. Beck: None; M. Patnaik: None; N. Young: None; K. Calvo: None; A. Casanegra: None; P. Grayson: None; M. Koster: None; C. Wu: None; D. Houghton: None; B. Patel: None; Y. Kanthi: None; E. Groarke: None.



