Abstract Session
Periodic fever syndromes, autoinflammatory diseases, Still’s disease and MAS/HLH
Session: Abstracts: Miscellaneous Rheumatic & Inflammatory Diseases III (2569–2574)
2569: Efficacy and Safety of Therapeutic Interventions for the Treatment of Still’s Disease: A Systematic Review and Meta-analysis Informing the EULAR/PReS Recommendations for the Diagnosis and Management of Systemic Juvenile Idiopathic Arthritis and Adult-Onset Still’s Disease
Wednesday, November 15, 2023
9:15 AM - 9:25 AM PT
Location: Room 32A-B
- SB
Sara Bindoli, MD, PhD
Rheumatology Unit, Department of Medicine,University of Padova, Italy
Padova, Padua, ItalyDisclosure information not submitted.
Presenting Author(s)
Sara Bindoli1, Arianna De Matteis2, STEPHANE MITROVIC3, Bruno Fautrel4, Loreto Carmona5, Fabrizio De Benedetti6 and On Behalf Of The Eular/PreS QoC011 Task Force Member7, 1Rheumatology Unit, Dept of Medicine, University of Padova, Padova, Italy, 2IRCCS Ospedale Pediatrico Bambino Gesu', Rome, Italy, 3Pitie Salpetriere Hospital, Sorbonne University, APHP, Paris, France, 4Sorbonne Université APHP, Paris, France, 5Instituto de Salud Musculoesquelética (InMusc), Madrid, Spain, 6Bambino Gesu Children's Hospital, Division of Rheumatology, Rome, Italy, 7EULAR/PReS QoC011 Task Force members are: Jordi Anton, Alexandre Belot, Claudia Bracaglia, Tamas Constantin, Lorenzo Dagna, Alessandro De Bartolo, Eugen Feist, Dirk Foell, Marco Gattorno, Sophie Georgin-Lavialle, Roberto Giacomelli, Alexei Grom, Yvan Jamilloux, Katarina Laskari, Calin Lazar, Francesca Minoia, Peter Nigrovic, Filipa Oliveira Ramos, Seza Ozen, Pierre Quartier-dit-Maire, Piero Ruscitti, Erdal Sag, Sinisa Savic, Marie-Elise Truchetet, Bas Vastert, Tanita Wilhelmer, Carine Wouters., Paris, France
Background/Purpose: To investigate the efficacy and safety of therapies currently in use and under evaluation for systemic Juvenile Idiopathic Arthritis (sJIA) and Adult-Onset Still's disease (AOSD).
Methods: A systematic review (SR) was performed. Medline, Embase, and the Cochrane Library databases were searched up to October 2022 for clinical trials (randomised, RCT, and quasi-controlled, CCT), longitudinal observational studies (retrospective, LOR, and prospective, LOP) and SRs published after 2013. The research question was formulated according to the PICO format. Population: sJIA (fulfilling ILAR criteria) or AOSD (fulfilling either Yamaguchi's and/or Fautrel's criteria) patients; Intervention: any pharmacological treatment (in use or under evaluation for sJIA/AOSD); Comparator: any other active drug or placebo; Outcomes: any relevant efficacy and safety outcome. The risk of bias (RoB) of the included clinical studies was assessed with the Cochrane RoB tool, while AMSTAR-2 was used to critically assess SRs.
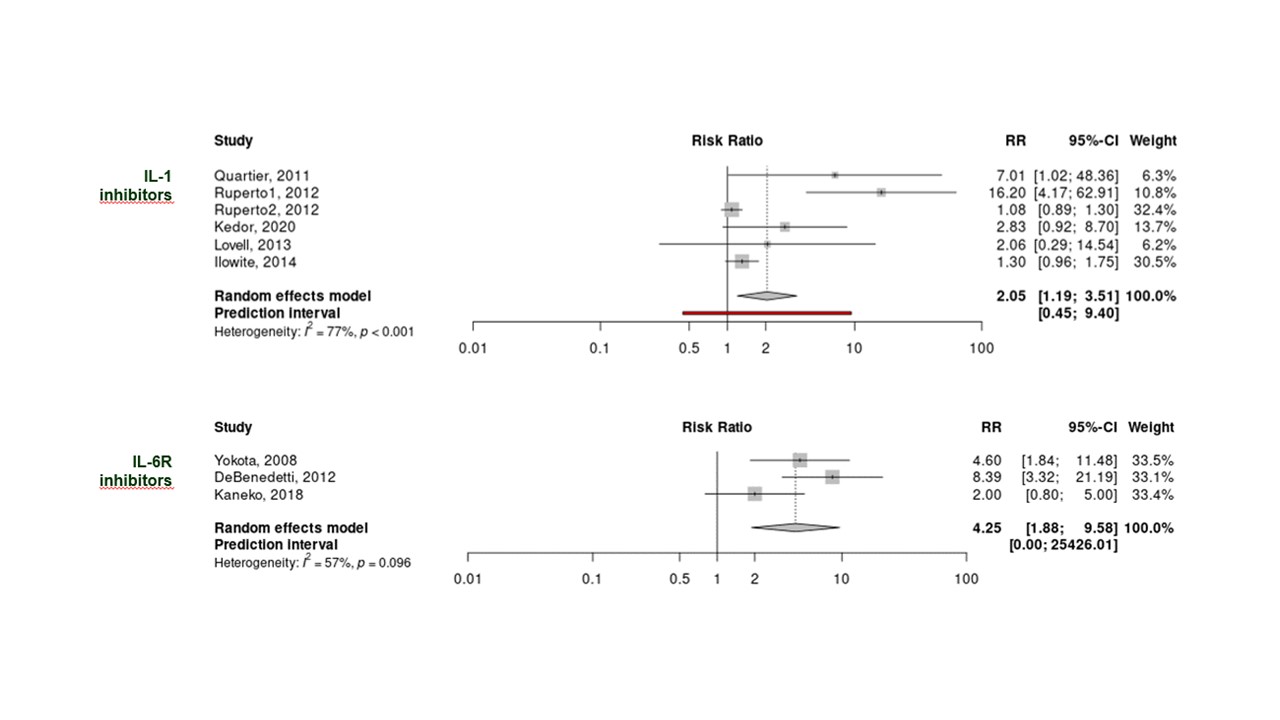
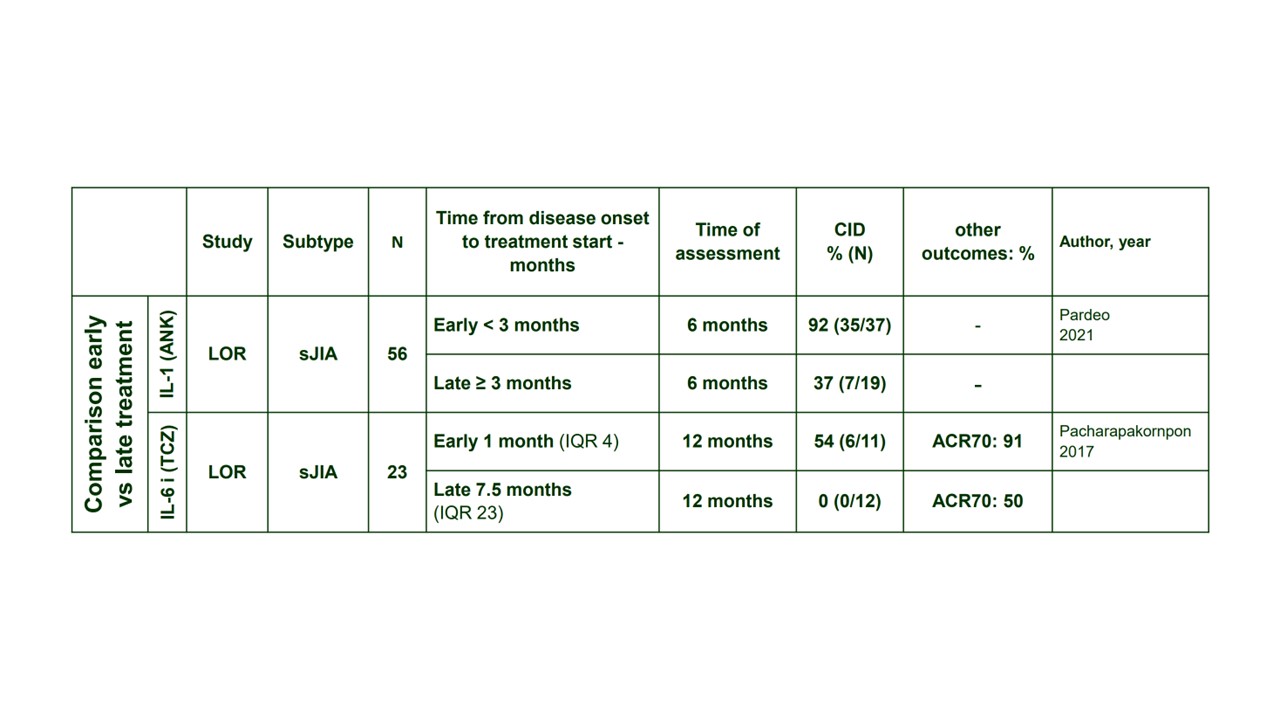
Results: Of 3,941 records, 115 full texts were finally included, representing 25 RCTs, 10 SRs published after 2013 and 80 LOR or LOP studies. Studies on glucocorticoids (GCs) and conventional synthetic DMARDs were mainly observational and displayed high RoB for the majority of them. Biologic DMARD (bDMARD) targeting IL-1 (IL-1i) or IL-6R (IL-6Ri) were the drugs with the highest level of evidence (6 clinical trials for IL-1i and 3 for IL-6Ri). A meta-analysis was conducted accordingly, using adapted ACR50 response as the outcome measure, and identified significant effect of both bDMARDs with RR 2.05 (95%CI 1.19, 3.51) and 4.25 (95%CI 1.88, 9.58) for IL1i and IL-6Ri respectively (Figure). Additionally, 2 retrospective longitudinal studies have demonstrated that the benefit of early bDMARD introduction, i.e. < 3 months, was associated with higher rates of clinically inactive disease (Table). This is consistent with data from other non-comparative studies in which clinically inactive disease (CID) achievement rates are between 59 to 100% when IL-1 or IL-6 inhibitors are initiated within 3 to 6 months, and 23 to 32% when they are started later on. Data on tsDMARDs (JAK-inhibitors), with promising findings, were limited to a few small case series.
Conclusion: Besides glucocorticoids, IL-1 and IL-6 inhibitors yielded the highest level of evidence for the treatment of sJIA and AOSD and their use early during the disease course was markedly efficacious.
S. Bindoli: None; A. De Matteis: None; S. MITROVIC: Eli Lilly, Pfitzer, BMS, SOBI, 2; B. Fautrel: AbbVie, 2, BMS, 2, Chugai, 2, Fresenius Kabi, 2, Galapagos, 2, Lilly, 2, Medac, 2, Nordic Pharma, 2, Novartis, 2, Pfizer, 2, Sobi, 2, UCB, 2; L. Carmona: Amgen, Fresenius Kabi Espana, Galapagos, Gilead, Pfizer, Lilly, Meda Pharma, MSD, Novartis, Roche, Sanofi Aventis, Upjohn, BMS, Novo Nordisk, and Sand, 5; F. De Benedetti: Abbvie, Novimmune, Novartis, Roche, Sanofi-Aventis, Sobi, Regeneron, Elixiron and Zydus, 5; O. Task Force Member: None.
Background/Purpose: To investigate the efficacy and safety of therapies currently in use and under evaluation for systemic Juvenile Idiopathic Arthritis (sJIA) and Adult-Onset Still's disease (AOSD).
Methods: A systematic review (SR) was performed. Medline, Embase, and the Cochrane Library databases were searched up to October 2022 for clinical trials (randomised, RCT, and quasi-controlled, CCT), longitudinal observational studies (retrospective, LOR, and prospective, LOP) and SRs published after 2013. The research question was formulated according to the PICO format. Population: sJIA (fulfilling ILAR criteria) or AOSD (fulfilling either Yamaguchi's and/or Fautrel's criteria) patients; Intervention: any pharmacological treatment (in use or under evaluation for sJIA/AOSD); Comparator: any other active drug or placebo; Outcomes: any relevant efficacy and safety outcome. The risk of bias (RoB) of the included clinical studies was assessed with the Cochrane RoB tool, while AMSTAR-2 was used to critically assess SRs.
Results: Of 3,941 records, 115 full texts were finally included, representing 25 RCTs, 10 SRs published after 2013 and 80 LOR or LOP studies. Studies on glucocorticoids (GCs) and conventional synthetic DMARDs were mainly observational and displayed high RoB for the majority of them. Biologic DMARD (bDMARD) targeting IL-1 (IL-1i) or IL-6R (IL-6Ri) were the drugs with the highest level of evidence (6 clinical trials for IL-1i and 3 for IL-6Ri). A meta-analysis was conducted accordingly, using adapted ACR50 response as the outcome measure, and identified significant effect of both bDMARDs with RR 2.05 (95%CI 1.19, 3.51) and 4.25 (95%CI 1.88, 9.58) for IL1i and IL-6Ri respectively (Figure). Additionally, 2 retrospective longitudinal studies have demonstrated that the benefit of early bDMARD introduction, i.e. < 3 months, was associated with higher rates of clinically inactive disease (Table). This is consistent with data from other non-comparative studies in which clinically inactive disease (CID) achievement rates are between 59 to 100% when IL-1 or IL-6 inhibitors are initiated within 3 to 6 months, and 23 to 32% when they are started later on. Data on tsDMARDs (JAK-inhibitors), with promising findings, were limited to a few small case series.
Conclusion: Besides glucocorticoids, IL-1 and IL-6 inhibitors yielded the highest level of evidence for the treatment of sJIA and AOSD and their use early during the disease course was markedly efficacious.

Figure: Meta-analysis of controlled clinical trials having assessed the efficacy of IL-1 and IL-6R inhibitors in Still’s disease, based on ACR50 response.

Table: Studies having compared early (< 3 months) versus late (≥3 months) introduction of IL-1 and IL-6R inhibitors for the treatment of people with Still’s disease.
S. Bindoli: None; A. De Matteis: None; S. MITROVIC: Eli Lilly, Pfitzer, BMS, SOBI, 2; B. Fautrel: AbbVie, 2, BMS, 2, Chugai, 2, Fresenius Kabi, 2, Galapagos, 2, Lilly, 2, Medac, 2, Nordic Pharma, 2, Novartis, 2, Pfizer, 2, Sobi, 2, UCB, 2; L. Carmona: Amgen, Fresenius Kabi Espana, Galapagos, Gilead, Pfizer, Lilly, Meda Pharma, MSD, Novartis, Roche, Sanofi Aventis, Upjohn, BMS, Novo Nordisk, and Sand, 5; F. De Benedetti: Abbvie, Novimmune, Novartis, Roche, Sanofi-Aventis, Sobi, Regeneron, Elixiron and Zydus, 5; O. Task Force Member: None.



