Abstract Session
Systemic lupus erythematosus (SLE)
Session: Abstracts: SLE – Diagnosis, Manifestations, & Outcomes III: Disease Activity (2551–2556)
2553: Prevalence, Determinants and Outcomes of Target Attainment in SLE Patients with Clinically Active Disease in a Large Multinational Prospective Lupus Cohort
Tuesday, November 14, 2023
4:30 PM - 4:40 PM PT
Location: Ballroom 20A
.png)
Mandana Nikpour, PhD, MBBS
The University of Sydney
Melbourne, Victoria, AustraliaDisclosure information not submitted.
Presenting Author(s)
Yanjie Hao1, Dylan Hansen2, Rangi Kandane-Rathnayake3, Worawit Louthrenoo4, Yi-Hsing Chen5, Jiacai Cho6, Aisha Lateef6, Laniyati Hamijoyo7, Shue-Fen Luo8, Yeong-Jian J Wu9, Sandra Navarra10, Leonid Zamora10, Zhanguo Li11, Sargunan Sockalingam12, Yasuhiro Katsumata13, Masayoshi Harigai13, Zhuoli Zhang14, Madelynn Chan15, Jun Kikuchi16, Tsutomu Takeuchi17, Sang-Cheol Bae18, Fiona Goldblatt19, Sean O’Neill20, Kristine Ng21, Annie Law22, BMDB Basnayake23, Nicola Tugnet24, Sunil Kumar25, Cherica Tee26, Michael Tee26, Yoshiya Tanaka27, Chak Sing Lau28, Vera Golder3, Alberta Hoi29, Eric Morand30, Shereen Oon31 and Mandana Nikpour32, 1The University of Melbourne Department of Medicine at St Vincents Hospital Melbourne, Melbourne, Australia, 2Department of Rheumatology, St Vincent’s Hospital Melbourne, Fitzroy, Australia, 3Monash University, Department of Medicine, Sub-faculty of Clinical and Molecular Medicine, Clayton, Australia, 4Chiang Mai University Hospital, Department of Internal Medicine, Faculty of Medicine, Chiang Mai, Thailand, 5Division of Allergy, Immunology and Rheumatology, Taichung Veterans General Hospital, Taichung, Taiwan, 6National University Hospital, Rheumatology Division, Department of Medicine, Singapore, Singapore, 7Padjadjaran University/Hasan Sadikin General Hospital, Division of Rheumatology, Department of Internal Medicine, Faculty of Medicine, Bandung, Indonesia, 8Chang Gung University, Department of Rheumatology, Allergy and Immunology, Chang Gung Memorial Hospital, Taoyuan, Taiwan, 9Chang Gung University, Department of Rheumatology, Allergy and Immunology, Chang Gung Memorial Hospital, Keelung, Taiwan, 10University of Santo Tomas Hospital, Joint and Bone Center, Manila, Philippines, 11Peking University Health Science Center, Department of Rheumatology and Immunology, People's Hospital, Beijing, China, 12University of Malaya, Department of Medicine, Faculty of Medicine Building, Kuala Lumpur, Malaysia, 13Tokyo Women's Medical University, Division of Rheumatology, Department of Internal Medicine, Tokyo, Japan, 14Peking University First Hospital, Rheumatology and Immunology Department, Beijing, China, 15Tan Tock Seng Hospital, Department of Rheumatology, Allergy & Immunology, Singapore, Singapore, 16Keio University School of Medicine, Tokyo, Japan, 17Keio University School of Medicine and Saitama Medical University, Tokyo, Japan, 18Hanyang University Hospital for Rheumatic Diseases and Hanyang University Institute for Rheumatology Research, Department of Rheumatology, Seoul, South Korea, 19Royal Adelaide Hospital and Flinders Medical Centre, Adelaide, Australia, 20Department of Medicine, University of New South Wales, Kensington, Australia, 21Waitemata DHB, Auckland, New Zealand, 22Singapore General Hospital; Duke-NUS Medical School, Singapore, Singapore, 23Division of Nephrology, Teaching Hospital Kandy, Adelaide, Australia, 24Auckland District Health Board, Auckland, New Zealand, 25Middlemore Hospital, Auckland, New Zealand, 26University of the Philippines, Manila, Philippines, 27University of Occupational and Environmental Health, Kitakyushu, Japan, 28University of Hong Kong, Division of Rheumatology & Clinical Immunology, Department of Medicine, Queen Mary Hospital, Hong Kong, Hong Kong, 29Monash University, Department of Medicine, Sub-faculty of Clinical and Molecular Medicine, Melbourne, Australia, 30Monash University, Centre for Inflammatory Diseases, Melbourne, Australia, 31University of Melbourne at St Vincent’s Hospital, Departments of Rheumatology and Medicine, Fitzroy, Australia, 32The University of Melbourne at St. Vincent’s Hospital Melbourne, Departments of Medicine and Rheumatology, Melbourne, Australia
Background/Purpose: There is increasing interest in adopting the principle of treating to target (T2T) in systemic lupus erythematosus (SLE). Remission and low disease activity states have been demonstrated to have protective associations with adverse outcomes such as flare, damage accrual and mortality. However, knowledge gaps remain in relation to the timing and impact of target attainment. In this study, we aimed to identify (i) the proportion of patients achieving the lupus low disease activity state (LLDAS) and the definition of remission in SLE (DORIS-remission) subsequent to a visit with clinically active disease, (ii) time to attainment of LLDAS and DORIS-remission, (iii) determinants of attaining these targets, and (iv) frequency and time to flare and damage accrual following target attainment.
Methods: Patients in the Asia Pacific Lupus Collaboration (APLC) cohort, followed between 2013 and 2020 who had any clinical disease activity according to the clinical domains of SLEDAI-2K and did not fulfill LLDAS or DORIS-remission were followed from their first time fulfilling these criteria. SELENA-SLEDAI flare index and the SLICC/ACR Damage Index were used to assess flare and damage accrual, respectively. We used Multivariable Cox regression models to analyse factors associated with attainment of LLDAS and DORIS-remission. Generalized estimating equation (GEE) models were used to identify factors associated with flare after attainment of target.
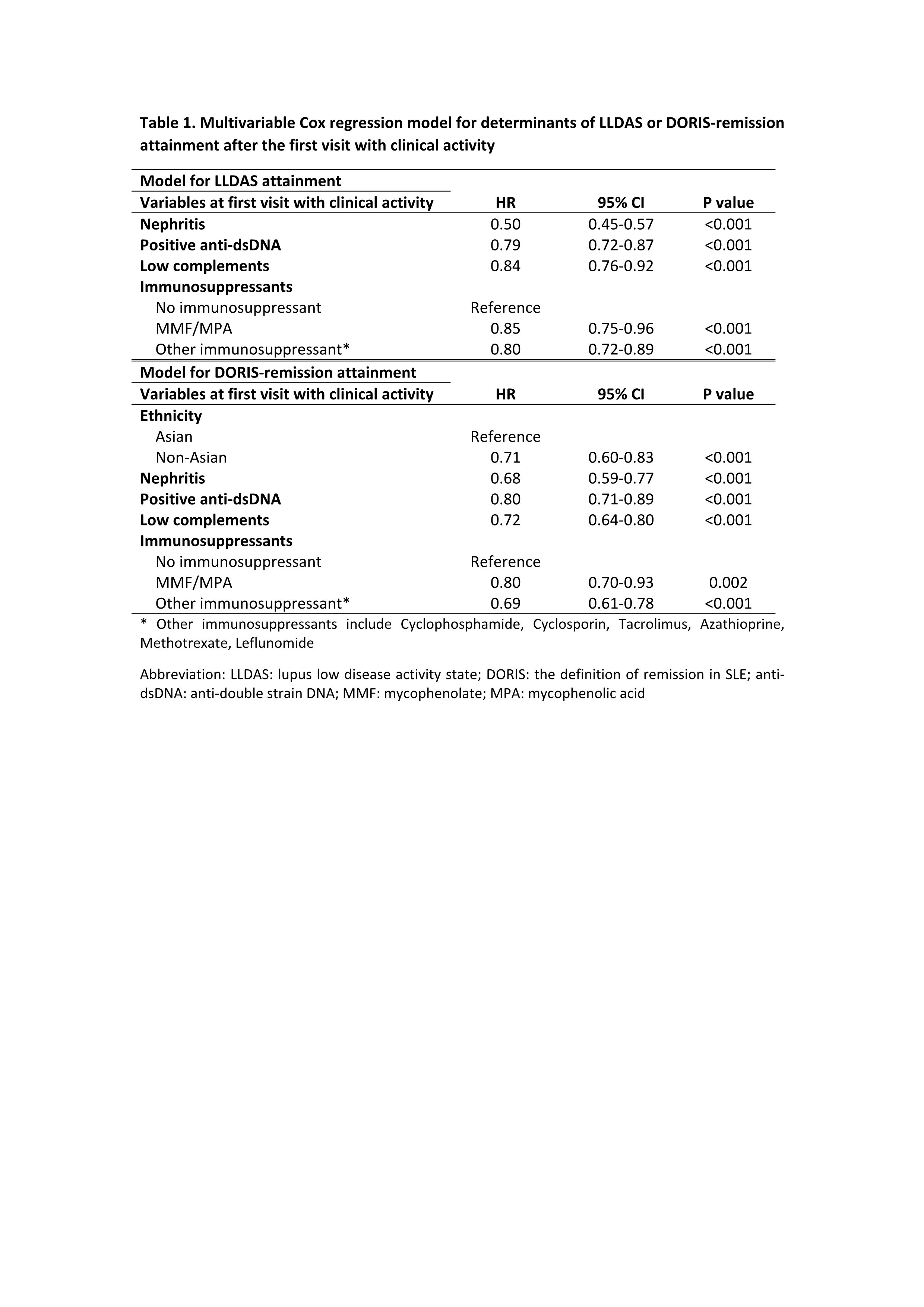
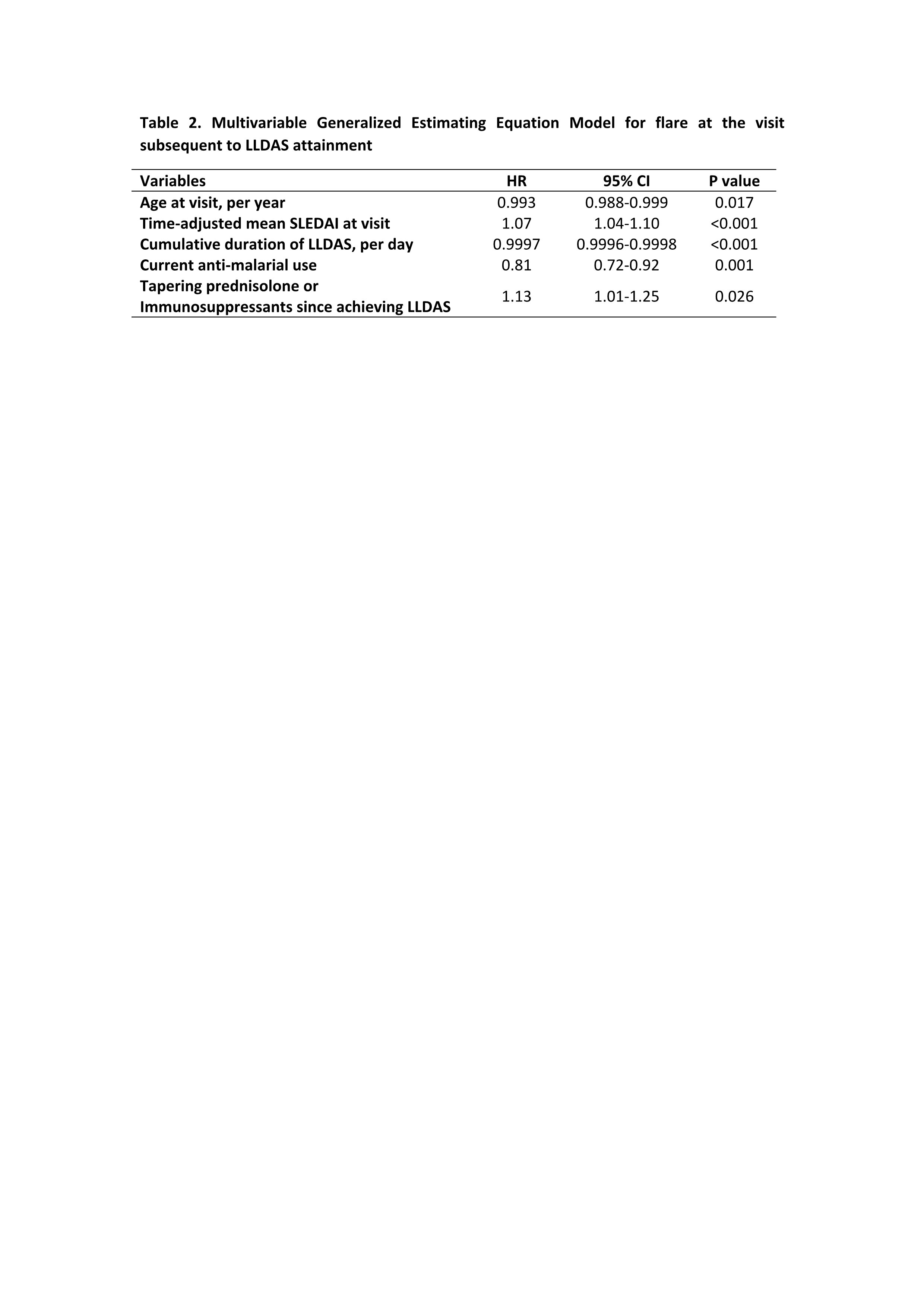
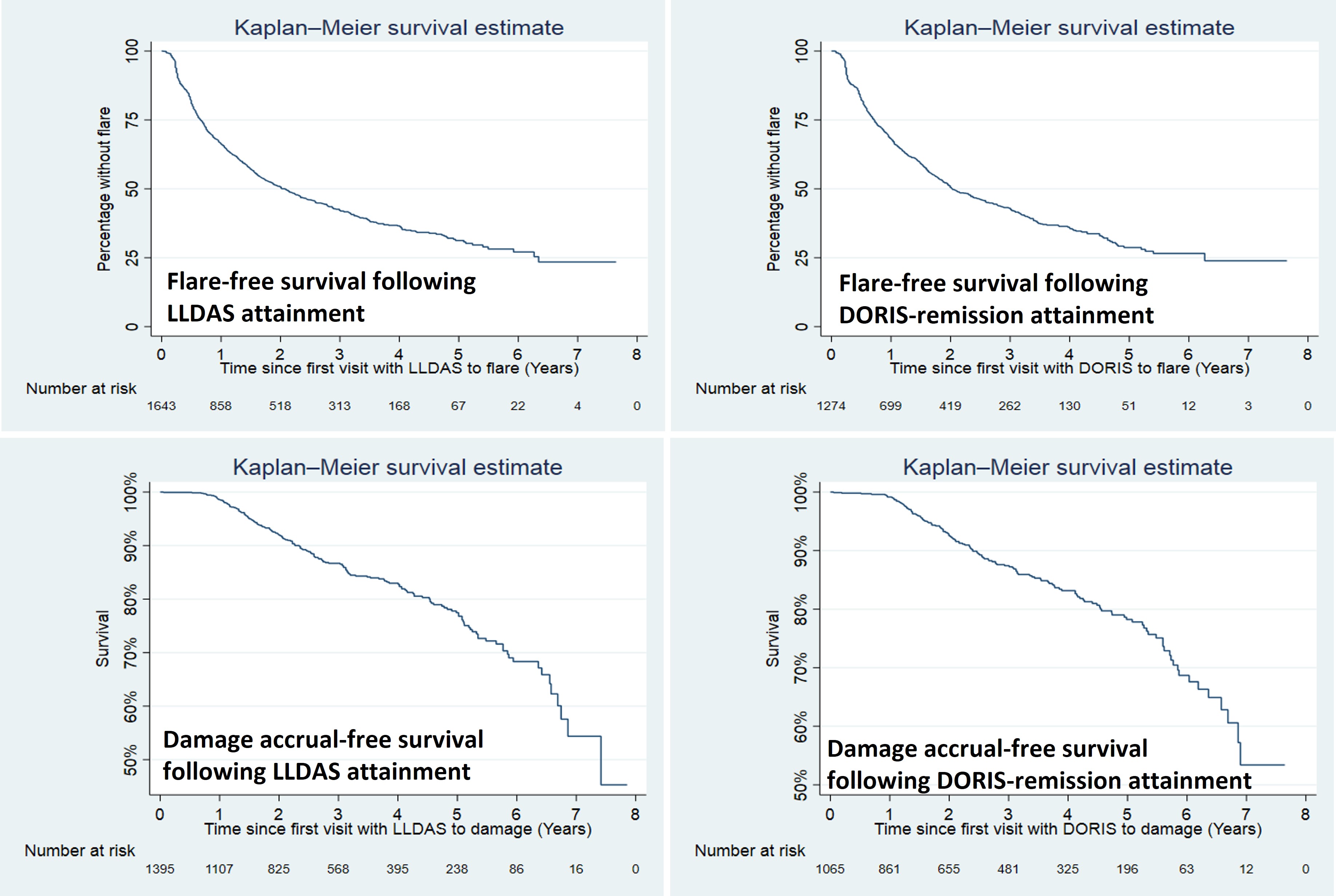
Results: 2852 patients (92.4% female) were followed for a median of 3.3 (1.1-5.7) years. The mean (standard deviations) SLEDAI and PGA at the first visit with clinical activity were 7.0±4.3 and 0.9±0.7, respectively. 1858 (65.2%) of the patients achieved LLDAS, with median (interquartile ranges) time to first attainment of LLDAS being 0.5 (0.3-1.2) years (Figure 1). The proportion and median time to first attainment of DORIS-remission were 49.3% and 0.7 (0.4-1.6) years, respectively. Multivariable Cox model showed that nephritis, positive anti-dsDNA and low complement were associated with a longer time to attainment of LLDAS and DORIS-remission, and in addition non-Asian ethnicity was associated with a longer time taken to attain DORIS-remission (Table 1). After the first attainment of LLDAS, 887/1852 (47.9%) of patients experienced flare(s) with a median time to first flare of 0.7 (0.3-1.4) years. 679/1405 (48.3%) patients had flare(s) after the first attainment of DORIS-remission within a median time of 0.8 (0.3-1.6) years (Figure 1). In multivariable GEE model, factors associated with flare post attainment of LLDAS included tapering of corticosteroids and immunosuppressants (Table 2). 110/960 (11.5%) and 77/750 (10.2%) patients had damage accrual within 24 months of first attainment of LLDAS and DORIS-remission, respectively (Figure 1).
Conclusion: In patients with clinically active disease, LLDAS and DORIS-remission are both achievable goals. However, flares still occur in almost 50% of patients, and 10-11.5% of patients still accrue damage over two years following LLDAS or DORIS-remission attainment, suggesting that further research is needed to inform monitoring and treatment adjustment in SLE patients to maintain states associated with optimal outcomes.
Y. Hao: None; D. Hansen: None; R. Kandane-Rathnayake: None; W. Louthrenoo: None; Y. Chen: None; J. Cho: None; A. Lateef: None; L. Hamijoyo: None; S. Luo: None; Y. Wu: None; S. Navarra: Astellas, 6, AstraZeneca, 6, Biogen, 2, Boehringer-Ingelheim, 2, GSK, 6, Novartis, 6, Pfizer, 6; L. Zamora: None; Z. Li: Abbott, 2, 9, Abbvie, 2, 9, BMS, 2, 9, Celgene, 2, 9, Eli Lilly, 2, 9, GSK, 2, 9, Janssen, 2, 9, MSD, 2, 9, Novartis, 2, 9, Pfizer, 2, 9, Roche, 2, 9, UCB Pharma, 2, 9; S. Sockalingam: AstraZeneca, 2, 12, Other - for attending meetings and/or travel, Pfizer, 2, 12, Other - for attending meetings and/or travel, ZP Therapeutics, 2; Y. Katsumata: Asahi Kasei Pharma, 6, Astella, 6, AstraZeneca, 6, Chugai, 6, GlaxoSmithKlein(GSK), 6, Janssen, 6, Mitsubishi Tanabe Pharma Corporation, 6, Pfizer, 6; M. Harigai: Astellas Pharma, 6, AstraZeneca, 6, GlaxoSmithKlein(GSK), 6, 12, Post marketing surveillence, Novartis, 5; Z. Zhang: None; M. Chan: None; J. Kikuchi: None; T. Takeuchi: AbbVie, 2, 5, 6, AYUMI, 5, Bristol-Myers Squibb, 6, Chugai, 2, 5, 6, Daiichi Sankyo, 5, Eisai, 5, 6, Eli Lilly Japan, 2, 6, Gilead, 2, 6, Janssen, 6, Mitsubishi-Tanabe, 2, 5, 6, ONO, 5, Pfizer Japan, 6, Taiho, 2; S. Bae: None; F. Goldblatt: None; S. O’Neill: None; K. Ng: AbbVie/Abbott, 1; A. Law: None; B. Basnayake: None; N. Tugnet: None; S. Kumar: None; C. Tee: None; M. Tee: None; Y. Tanaka: AbbVie, 6, AstraZeneca, 6, BMS, 6, Boehringer-Ingelheim, 6, Chugai, 5, 6, Eisai, 5, 6, Eli Lilly, 6, Gilead, 6, GSK, 6, Mitsubishi-Tanabe, 5, Pfizer, 6, Taiho, 6, Taisho, 5, 6; C. Lau: AstraZeneca, 6, 12, external expert for SLE sterring committee 11.2022; V. Golder: None; A. Hoi: Abbvie, 6, AstraZeneca, 5, Australian Rheumatology Association, 4, Eli Lilly, 6, EUSA Pharma (UK) Limited, 2, Limbic, 6, Moose Republic, 6, Novartis, 6; E. Morand: AbbVie, 2, 5, Amgen, 5, AstraZeneca, 2, 5, 6, Biogen, 2, 5, Bristol Myers Squibb, 2, 5, Eli Lilly, 2, 5, EMD Serono, 2, 5, Galapagos, 2, Genentech, 2, 5, GlaxoSmithKline, 2, 5, IgM, 2, Janssen, 2, 5, Novartis, 2, Servier, 2, Takeda, 2, UCB, 5; S. Oon: None; M. Nikpour: AstraZeneca, 2, 6, Boehringer-Ingelheim, 2, 6, GSK, 2, 6, Janssen Pharmaceuticals, 2, 5, 6.
Background/Purpose: There is increasing interest in adopting the principle of treating to target (T2T) in systemic lupus erythematosus (SLE). Remission and low disease activity states have been demonstrated to have protective associations with adverse outcomes such as flare, damage accrual and mortality. However, knowledge gaps remain in relation to the timing and impact of target attainment. In this study, we aimed to identify (i) the proportion of patients achieving the lupus low disease activity state (LLDAS) and the definition of remission in SLE (DORIS-remission) subsequent to a visit with clinically active disease, (ii) time to attainment of LLDAS and DORIS-remission, (iii) determinants of attaining these targets, and (iv) frequency and time to flare and damage accrual following target attainment.
Methods: Patients in the Asia Pacific Lupus Collaboration (APLC) cohort, followed between 2013 and 2020 who had any clinical disease activity according to the clinical domains of SLEDAI-2K and did not fulfill LLDAS or DORIS-remission were followed from their first time fulfilling these criteria. SELENA-SLEDAI flare index and the SLICC/ACR Damage Index were used to assess flare and damage accrual, respectively. We used Multivariable Cox regression models to analyse factors associated with attainment of LLDAS and DORIS-remission. Generalized estimating equation (GEE) models were used to identify factors associated with flare after attainment of target.
Results: 2852 patients (92.4% female) were followed for a median of 3.3 (1.1-5.7) years. The mean (standard deviations) SLEDAI and PGA at the first visit with clinical activity were 7.0±4.3 and 0.9±0.7, respectively. 1858 (65.2%) of the patients achieved LLDAS, with median (interquartile ranges) time to first attainment of LLDAS being 0.5 (0.3-1.2) years (Figure 1). The proportion and median time to first attainment of DORIS-remission were 49.3% and 0.7 (0.4-1.6) years, respectively. Multivariable Cox model showed that nephritis, positive anti-dsDNA and low complement were associated with a longer time to attainment of LLDAS and DORIS-remission, and in addition non-Asian ethnicity was associated with a longer time taken to attain DORIS-remission (Table 1). After the first attainment of LLDAS, 887/1852 (47.9%) of patients experienced flare(s) with a median time to first flare of 0.7 (0.3-1.4) years. 679/1405 (48.3%) patients had flare(s) after the first attainment of DORIS-remission within a median time of 0.8 (0.3-1.6) years (Figure 1). In multivariable GEE model, factors associated with flare post attainment of LLDAS included tapering of corticosteroids and immunosuppressants (Table 2). 110/960 (11.5%) and 77/750 (10.2%) patients had damage accrual within 24 months of first attainment of LLDAS and DORIS-remission, respectively (Figure 1).
Conclusion: In patients with clinically active disease, LLDAS and DORIS-remission are both achievable goals. However, flares still occur in almost 50% of patients, and 10-11.5% of patients still accrue damage over two years following LLDAS or DORIS-remission attainment, suggesting that further research is needed to inform monitoring and treatment adjustment in SLE patients to maintain states associated with optimal outcomes.



Flare-free and damage accrual-free survival after first attainment of LLDAS or DORIS-remission
Y. Hao: None; D. Hansen: None; R. Kandane-Rathnayake: None; W. Louthrenoo: None; Y. Chen: None; J. Cho: None; A. Lateef: None; L. Hamijoyo: None; S. Luo: None; Y. Wu: None; S. Navarra: Astellas, 6, AstraZeneca, 6, Biogen, 2, Boehringer-Ingelheim, 2, GSK, 6, Novartis, 6, Pfizer, 6; L. Zamora: None; Z. Li: Abbott, 2, 9, Abbvie, 2, 9, BMS, 2, 9, Celgene, 2, 9, Eli Lilly, 2, 9, GSK, 2, 9, Janssen, 2, 9, MSD, 2, 9, Novartis, 2, 9, Pfizer, 2, 9, Roche, 2, 9, UCB Pharma, 2, 9; S. Sockalingam: AstraZeneca, 2, 12, Other - for attending meetings and/or travel, Pfizer, 2, 12, Other - for attending meetings and/or travel, ZP Therapeutics, 2; Y. Katsumata: Asahi Kasei Pharma, 6, Astella, 6, AstraZeneca, 6, Chugai, 6, GlaxoSmithKlein(GSK), 6, Janssen, 6, Mitsubishi Tanabe Pharma Corporation, 6, Pfizer, 6; M. Harigai: Astellas Pharma, 6, AstraZeneca, 6, GlaxoSmithKlein(GSK), 6, 12, Post marketing surveillence, Novartis, 5; Z. Zhang: None; M. Chan: None; J. Kikuchi: None; T. Takeuchi: AbbVie, 2, 5, 6, AYUMI, 5, Bristol-Myers Squibb, 6, Chugai, 2, 5, 6, Daiichi Sankyo, 5, Eisai, 5, 6, Eli Lilly Japan, 2, 6, Gilead, 2, 6, Janssen, 6, Mitsubishi-Tanabe, 2, 5, 6, ONO, 5, Pfizer Japan, 6, Taiho, 2; S. Bae: None; F. Goldblatt: None; S. O’Neill: None; K. Ng: AbbVie/Abbott, 1; A. Law: None; B. Basnayake: None; N. Tugnet: None; S. Kumar: None; C. Tee: None; M. Tee: None; Y. Tanaka: AbbVie, 6, AstraZeneca, 6, BMS, 6, Boehringer-Ingelheim, 6, Chugai, 5, 6, Eisai, 5, 6, Eli Lilly, 6, Gilead, 6, GSK, 6, Mitsubishi-Tanabe, 5, Pfizer, 6, Taiho, 6, Taisho, 5, 6; C. Lau: AstraZeneca, 6, 12, external expert for SLE sterring committee 11.2022; V. Golder: None; A. Hoi: Abbvie, 6, AstraZeneca, 5, Australian Rheumatology Association, 4, Eli Lilly, 6, EUSA Pharma (UK) Limited, 2, Limbic, 6, Moose Republic, 6, Novartis, 6; E. Morand: AbbVie, 2, 5, Amgen, 5, AstraZeneca, 2, 5, 6, Biogen, 2, 5, Bristol Myers Squibb, 2, 5, Eli Lilly, 2, 5, EMD Serono, 2, 5, Galapagos, 2, Genentech, 2, 5, GlaxoSmithKline, 2, 5, IgM, 2, Janssen, 2, 5, Novartis, 2, Servier, 2, Takeda, 2, UCB, 5; S. Oon: None; M. Nikpour: AstraZeneca, 2, 6, Boehringer-Ingelheim, 2, 6, GSK, 2, 6, Janssen Pharmaceuticals, 2, 5, 6.



