Abstract Session
Systemic lupus erythematosus (SLE)
Session: Abstracts: SLE – Treatment II: Nonrenal (2485–2490)
2489: Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI) Achievement and Sustained Response with Deucravacitinib, an Oral, Selective, Allosteric Tyrosine Kinase 2 Inhibitor, in a Phase 2 Trial in SLE
Tuesday, November 14, 2023
3:00 PM - 3:10 PM PT
Location: Ballroom 20D
- CA
Cristina Arriens, MD, MS
Oklahoma Medical Research Foundation
Oklahoma City, OK, United StatesDisclosure information not submitted.
Presenting Author(s)
Cristina Arriens1, Ronald van Vollenhoven2, Alice B. Gottlieb3, Coburn Hobar4, Samantha Pomponi4, Ravi Koti5, Thomas Wegman6 and Victoria P. Werth7, 1Oklahoma Medical Research Foundation and University of Oklahoma Health Sciences Center, Department of Arthritis & Clinical Immunology, Oklahoma City, OK, 2Amsterdam University Medical Centers, Amsterdam, Netherlands, 3Icahn School of Medicine at Mount Sinai, New York, NY, 4Bristol Myers Squibb, Princeton, NJ, 5Syneos Health, Morrisville, NC, 6Bristol Myers Squibb, Beaver Falls, PA, 7University of Pennsylvania, Wynnewood, PA
Background/Purpose: Deucravacitinib is a first-in-class, oral, selective, allosteric tyrosine kinase 2 inhibitor approved in multiple countries for the treatment of adults with plaque psoriasis. A 48-week, double-blind phase 2 trial (NCT03252587) in patients with SLE showed that deucravacitinib demonstrated greater efficacy compared with placebo across multiple endpoints, including the primary endpoint of Systemic Lupus Erythematosus Responder Index-4 (SRI[4]) at week 32 and the secondary endpoint of ≥50% reduction in CLASI activity (CLASI-50) for patients with moderate to severe skin involvement at baseline (CLASI score ≥10).1 Here, we explored whether patients could sustain CLASI-50 over time and achieve 100% reduction in CLASI activity. CLASI-50 was also evaluated by SLE cutaneous manifestation subtype.
Methods: Patients with active SLE and a baseline CLASI score ≥10 who received placebo (n=24), deucravacitinib 3 mg twice daily (BID) (n=23), 6 mg BID (n=25), or 12 mg once daily (n=29) were included in these analyses. Exploratory outcomes were the proportion of patients that sustained CLASI-50 for the 5 consecutive visits from weeks 32 through 48, achieved 100% reduction in CLASI activity at week 48, and achieved CLASI-50 by SLE cutaneous manifestation subtype. For the subtype analysis, patients were classified as acute, subacute, chronic, or discoid at the discretion of investigators using SLICC classification criteria at screening without biopsy confirmation. Patients could be in ≥1 subcategory based on classification criteria; all patients classified as discoid were included in the chronic subgroup. All analyses were descriptive.
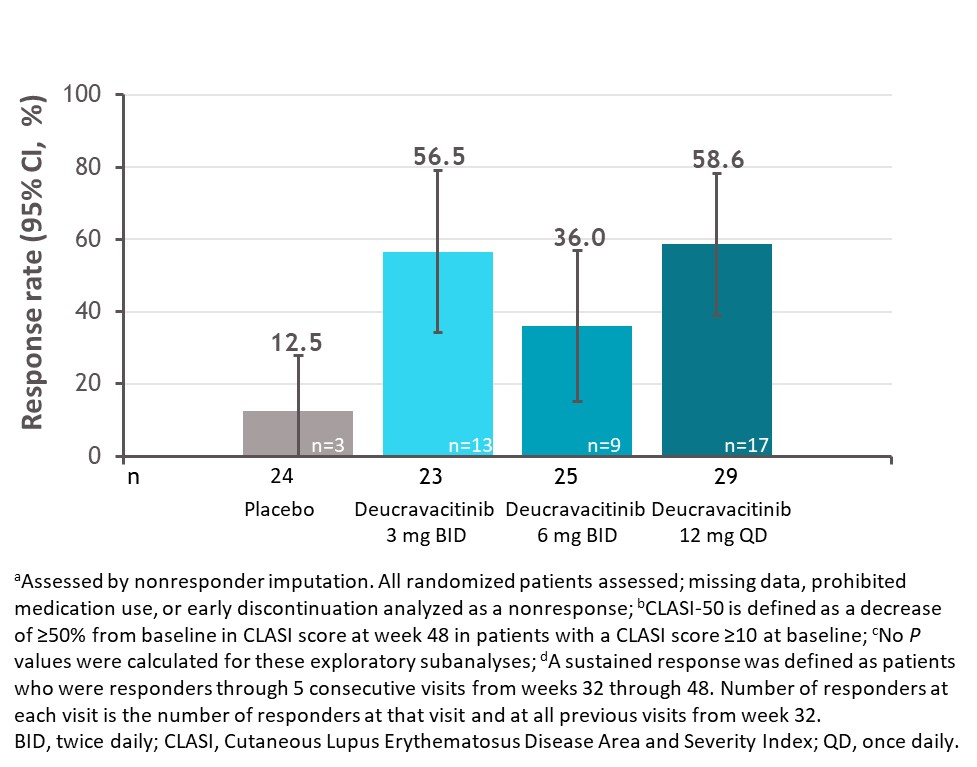
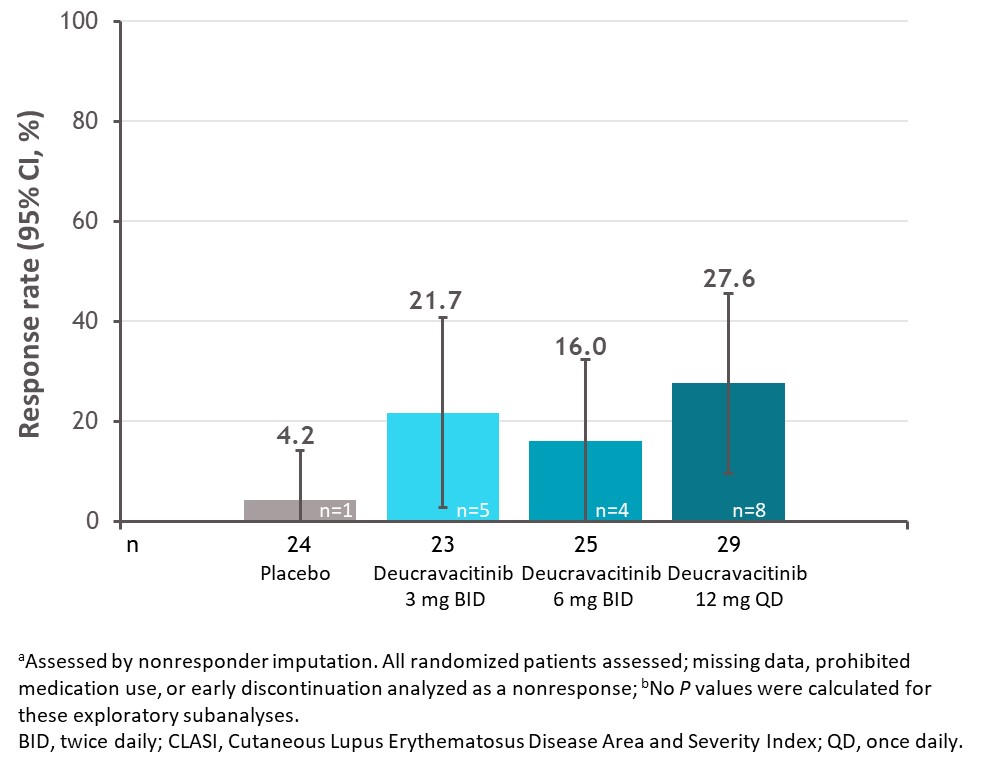
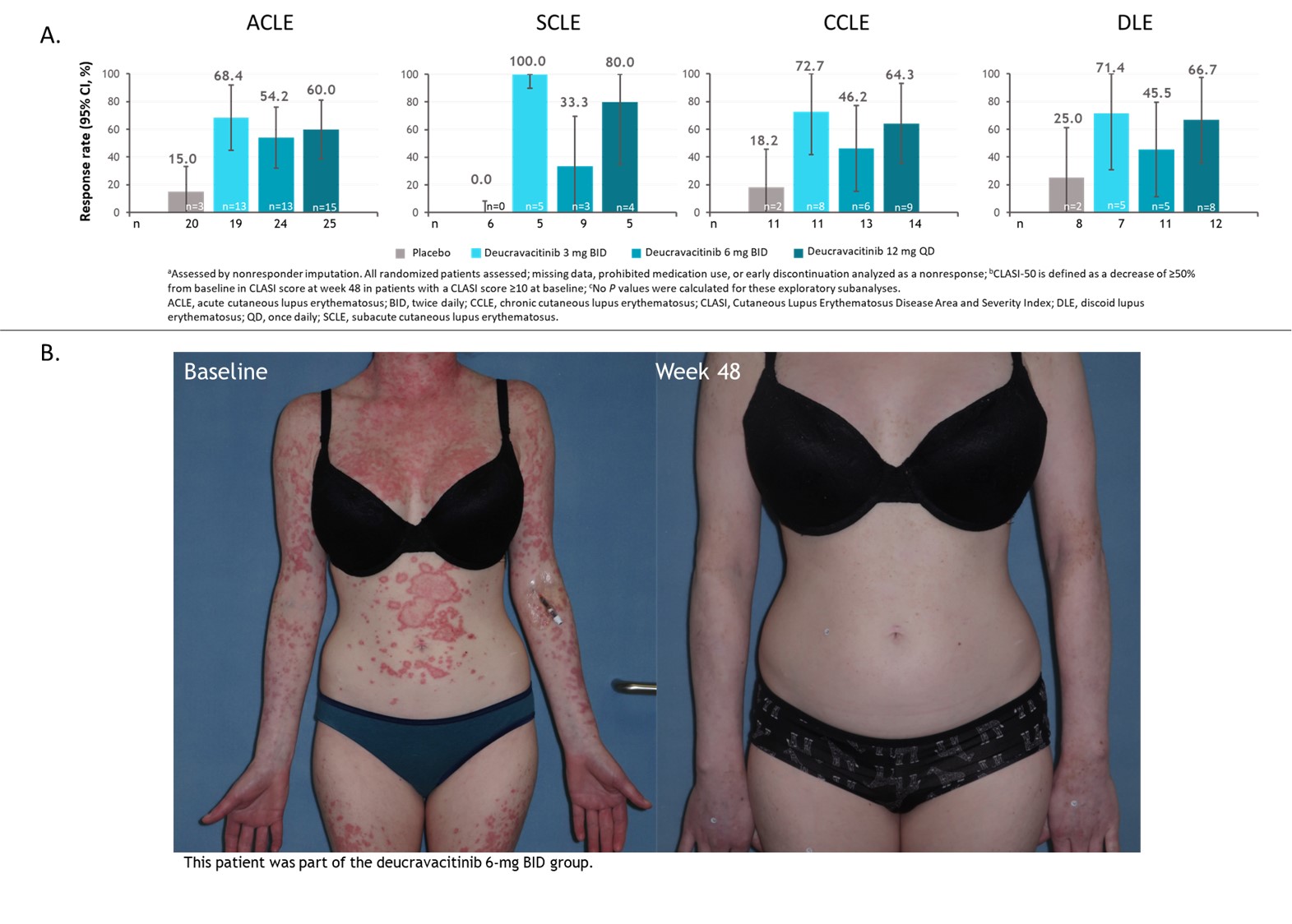
Results: Among patients with a baseline CLASI score ≥10, a numerically higher proportion of patients treated with deucravacitinib had a sustained response for the 5 consecutive visits between weeks 32 and 48 compared with placebo (Figure 1). Furthermore, a higher proportion of these patients treated with deucravacitinib achieved 100% reduction in CLASI activity at week 48 (Figure 2). Analysis by cutaneous manifestation suggested that a numerically higher proportion of patients treated with deucravacitinib achieved CLASI-50 at week 48 compared with placebo in all subtypes assessed (Figure 3).
Conclusion: Among patients with moderate to severe skin involvement at baseline, more patients treated with deucravacitinib were able to achieve improvements in skin overall, and a greater proportion of patients were able to sustain a CLASI-50 response from weeks 32 through 48 compared with placebo. CLASI-50 achievement was more frequent among patients treated with deucravacitinib regardless of cutaneous subtype.
Reference:
1. Morand E, et al. Arthritis Rheumatol 2023;75:242–252.
C. Arriens: AstraZeneca, 1, 5, 6, Aurinia, 6, Bristol-Myers Squibb, 1, 5, Cabaletta, 1, GSK, 1, Kezar, 1, UCB, 1; R. van Vollenhoven: AbbVie, 2, 6, AstraZeneca, 2, 5, 6, Biogen, 6, Bristol-Myers Squibb(BMS), 2, 5, 6, Galapagos, 2, 5, 6, GlaxoSmithKline, 6, Janssen, 2, 6, MSD/Merck Sharp and Dohme, 5, Novartis, 5, Pfizer, 2, 5, 6, RemeGen, 2, Roche, 5, Sanofi, 5, UCB, 2, 5, 6; A. Gottlieb: Amgen, 1, 2, AnaptysBio, 1, 2, 5, Avotres Therapeutics, 1, 2, Boehringer Ingelheim, 1, 2, Bristol Myers Squibb, 1, 2, 5, Dice Therapeutics, 1, 2, Eli Lilly, 1, 2, Janssen, 1, 2, MoonLake Immunotherapeutics, 5, Novartis, 1, 2, 5, Sanofi, 1, 2, UCB Pharma, 1, 2, 5, XBiotech, 1, 2; C. Hobar: Bristol-Myers Squibb(BMS), 3; S. Pomponi: Bristol-Myers Squibb(BMS), 3; R. Koti: Bristol-Myers Squibb(BMS), 12, providing statistical services, Syneos Health, 3; T. Wegman: Bristol-Myers Squibb(BMS), 3, 12, Shareholder; V. Werth: AbbVie, 2, Amgen, 2, 5, Anaptysbio, 2, Argenx, 5, AstraZeneca, 2, Biogen, 2, 5, Bristol Myers Squibb, 2, Celgene, 2, 5, Corbus, 5, CSL Behring, 2, 5, EMD Serono, 2, Galderma, 2, Genentech, 5, Gilead, 2, 5, GlaxoSmithKline, 2, Horizon Therapeutics, 5, Idera, 2, Incyte, 2, Janssen, 2, Kyowa Kirin, 2, Lilly, 2, MedImmune, 2, Medscape, 2, Merck, 2, Nektar, 2, Novartis, 2, Octapharma, 2, Pfizer, 2, 5, Principia, 2, Regeneron, 5, Resolve, 2, 2, Rome Therapeutics, 2, 5, Sanofi, 2, Ventus, 5, Viela, 2, 5, Xencor, 2.
Background/Purpose: Deucravacitinib is a first-in-class, oral, selective, allosteric tyrosine kinase 2 inhibitor approved in multiple countries for the treatment of adults with plaque psoriasis. A 48-week, double-blind phase 2 trial (NCT03252587) in patients with SLE showed that deucravacitinib demonstrated greater efficacy compared with placebo across multiple endpoints, including the primary endpoint of Systemic Lupus Erythematosus Responder Index-4 (SRI[4]) at week 32 and the secondary endpoint of ≥50% reduction in CLASI activity (CLASI-50) for patients with moderate to severe skin involvement at baseline (CLASI score ≥10).1 Here, we explored whether patients could sustain CLASI-50 over time and achieve 100% reduction in CLASI activity. CLASI-50 was also evaluated by SLE cutaneous manifestation subtype.
Methods: Patients with active SLE and a baseline CLASI score ≥10 who received placebo (n=24), deucravacitinib 3 mg twice daily (BID) (n=23), 6 mg BID (n=25), or 12 mg once daily (n=29) were included in these analyses. Exploratory outcomes were the proportion of patients that sustained CLASI-50 for the 5 consecutive visits from weeks 32 through 48, achieved 100% reduction in CLASI activity at week 48, and achieved CLASI-50 by SLE cutaneous manifestation subtype. For the subtype analysis, patients were classified as acute, subacute, chronic, or discoid at the discretion of investigators using SLICC classification criteria at screening without biopsy confirmation. Patients could be in ≥1 subcategory based on classification criteria; all patients classified as discoid were included in the chronic subgroup. All analyses were descriptive.
Results: Among patients with a baseline CLASI score ≥10, a numerically higher proportion of patients treated with deucravacitinib had a sustained response for the 5 consecutive visits between weeks 32 and 48 compared with placebo (Figure 1). Furthermore, a higher proportion of these patients treated with deucravacitinib achieved 100% reduction in CLASI activity at week 48 (Figure 2). Analysis by cutaneous manifestation suggested that a numerically higher proportion of patients treated with deucravacitinib achieved CLASI-50 at week 48 compared with placebo in all subtypes assessed (Figure 3).
Conclusion: Among patients with moderate to severe skin involvement at baseline, more patients treated with deucravacitinib were able to achieve improvements in skin overall, and a greater proportion of patients were able to sustain a CLASI-50 response from weeks 32 through 48 compared with placebo. CLASI-50 achievement was more frequent among patients treated with deucravacitinib regardless of cutaneous subtype.
Reference:
1. Morand E, et al. Arthritis Rheumatol 2023;75:242–252.

Figure 1. Sustained CLASI-50 response among patients with CLASI ≥10 at baseline (a, b, c, d)

Figure 2. Proportion of patients with CLASI ≥10 at baseline achieving a 100% reduction in CLASI at week 48 (a, b)

Figure 3. Proportion of patients with CLASI ≥10 at baseline achieving CLASI-50 at week 48 by cutaneous manifestation subtype (a, b, c) (A) and exemplary patient photographic data (B)
C. Arriens: AstraZeneca, 1, 5, 6, Aurinia, 6, Bristol-Myers Squibb, 1, 5, Cabaletta, 1, GSK, 1, Kezar, 1, UCB, 1; R. van Vollenhoven: AbbVie, 2, 6, AstraZeneca, 2, 5, 6, Biogen, 6, Bristol-Myers Squibb(BMS), 2, 5, 6, Galapagos, 2, 5, 6, GlaxoSmithKline, 6, Janssen, 2, 6, MSD/Merck Sharp and Dohme, 5, Novartis, 5, Pfizer, 2, 5, 6, RemeGen, 2, Roche, 5, Sanofi, 5, UCB, 2, 5, 6; A. Gottlieb: Amgen, 1, 2, AnaptysBio, 1, 2, 5, Avotres Therapeutics, 1, 2, Boehringer Ingelheim, 1, 2, Bristol Myers Squibb, 1, 2, 5, Dice Therapeutics, 1, 2, Eli Lilly, 1, 2, Janssen, 1, 2, MoonLake Immunotherapeutics, 5, Novartis, 1, 2, 5, Sanofi, 1, 2, UCB Pharma, 1, 2, 5, XBiotech, 1, 2; C. Hobar: Bristol-Myers Squibb(BMS), 3; S. Pomponi: Bristol-Myers Squibb(BMS), 3; R. Koti: Bristol-Myers Squibb(BMS), 12, providing statistical services, Syneos Health, 3; T. Wegman: Bristol-Myers Squibb(BMS), 3, 12, Shareholder; V. Werth: AbbVie, 2, Amgen, 2, 5, Anaptysbio, 2, Argenx, 5, AstraZeneca, 2, Biogen, 2, 5, Bristol Myers Squibb, 2, Celgene, 2, 5, Corbus, 5, CSL Behring, 2, 5, EMD Serono, 2, Galderma, 2, Genentech, 5, Gilead, 2, 5, GlaxoSmithKline, 2, Horizon Therapeutics, 5, Idera, 2, Incyte, 2, Janssen, 2, Kyowa Kirin, 2, Lilly, 2, MedImmune, 2, Medscape, 2, Merck, 2, Nektar, 2, Novartis, 2, Octapharma, 2, Pfizer, 2, 5, Principia, 2, Regeneron, 5, Resolve, 2, 2, Rome Therapeutics, 2, 5, Sanofi, 2, Ventus, 5, Viela, 2, 5, Xencor, 2.



