Abstract Session
Rheumatoid arthritis (RA)
Session: Abstracts: RA – Diagnosis, Manifestations, & Outcomes III: Predicting & Optimizing Outcomes (2479–2484)
2479: Tolerability and Effectiveness of Antifibrotics in Rheumatoid Arthritis-associated Interstitial Lung Disease
Tuesday, November 14, 2023
2:00 PM - 2:10 PM PT
Location: Ballroom 20B-C
- PJ
Pierre-Antoine Juge, MD, PhD (he/him/his)
Division of Rheumatology, Inflammation, and Immunity Brigham & Women’s Hospital
Boston, MA, United StatesDisclosure information not submitted.
Presenting Author(s)
Pierre-Antoine Juge1, Keigo Hayashi2, Gregory McDermott2, Kathleen Vanni2, Emily Kowalski2, Grace Qian2, Katarina Bade3, Alene Saavedra2, Philippe Dieudé4, Paul Dellaripa3, Tracy J. Doyle2 and Jeffrey Sparks5, 1Division of Rheumatology, Inflammation, and Immunity Brigham & Women's Hospital, Boston, MA, 2Brigham and Women's Hospital, Boston, MA, 3Brigham and Women's Hospital, Boston, MA, 4Assistance Publique-Hôpitaux de Paris, Bichat-Claude Bernard University Hospital, INSERM UMR1152, University de Paris Cité, Department of Rheumatology, Paris, France, 5Division of Rheumatology, Inflammation, and Immunity, Brigham and Women’s Hospital and Harvard Medical School, Boston, MA
Background/Purpose: Nintedanib and pirfenidone are antifibrotic drugs indicated for the treatment of idiopathic pulmonary fibrosis (IPF) and other forms of progressive pulmonary fibrosis. Antifibrotics have also been tested in RA-associated interstitial lung disease (ILD) within two recent randomized clinical trials (RCT). However, there are limited real-world studies regarding the use of antifibrotics, and none in RA-ILD. RA-ILD patients may be different from IPF patients since they often need concomitant immunosuppression for articular disease. Our aim was to investigate the tolerability and effectiveness of antifibrotics in a real-world cohort of patients with RA-ILD.
Methods: In this retrospective cohort study, we identified RA-ILD patients initiating antifibrotics at a large multi-hospital healthcare system. We used electronic query to find all patients with at least two RA diagnosis codes and a prescription for either nintedanib or pirfenidone (2014-2023). We then performed medical record review to confirm all patients met 2010 ACR/EULAR classification criteria for RA and had definite RA-ILD by Bongartz criteria. Data regarding adverse events (AEs), tolerability, pulmonary function test (PFT) results, and clinical data were collected. Drug retention was estimated using a Kaplan-Meier curve. A linear mixed model with random intercept was used to compare the within-patient trajectory of the percent predicted forced vital capacity (%FVC) within 18-months before to 18-months after antifibrotic initiation among those with these PFT data.
Results: We analyzed 74 patients with RA-ILD that initiated antifibrotics (mean age 67.8 years, 53% male); 40 patients initiated nintedanib and 34 initiated pirfenidone (Table 1). Median follow-up was 89 weeks (min 4, max 387). AEs were reported in 41 (55%) patients, with gastrointestinal (GI) AEs (n=30) being most common, followed by disease progression (n=6), rash (n=3), and hepatitis (n=2). The initial antifibrotic was discontinued in 34 (46%) patients due to: GI AEs (n=19), rash (n=3), transaminitis (n=2), and financial reasons (n=1). The median drug survival was 147.7 weeks (95%CI 79.1, NA; Figure 1). There was no difference in drug retention between nintedanib and pirfenidone (p=0.68). A second antifibrotic was prescribed in 14 patients, with 4 discontinuations. Change of %FVC trajectory was analyzed for 49 patients with available PFTs within 18 months pre- and post-antifibrotic (median number of PFT/patient = 4). There was a statistical difference in the estimated %FVC slope after initiation (-0.3% per year compared to -6.2% per year before initiation, p=0.031, Figure 2). Twenty-six patients (35%) died (17 due to ILD) and 4 (5%) had lung transplantation during follow-up.
Conclusion: In this first real-world study of antifibrotic use in RA-ILD, AEs were frequently reported, particularly GI, and discontinuation was common (46% compared to 20% for nintedanib and 24% for pirfenidone in their respective RCTs). However, antifibrotic initiation was associated with a modestly improved trajectory in %FVC. Death and/or lung transplant were frequent, emphasizing the need for additional safe and effective RA-ILD treatment options.
.jpg)
.jpg)
P. Juge: None; K. Hayashi: None; G. McDermott: None; K. Vanni: None; E. Kowalski: None; G. Qian: None; K. Bade: None; A. Saavedra: None; P. Dieudé: AbbVie, 2, 6, Boehringer Ingelheim, 1, 2, 6, Bristol-Myers Squibb, 1, 2, 5, 6, Chugai, 5, Galapagos, 5, 6, Janssen, 2, 6, Novartis, 2, Pfizer, 1, 2, 5, 6; P. Dellaripa: None; T. Doyle: None; J. Sparks: AbbVie, 2, Amgen, 2, Boehringer Ingelheim, 2, Bristol-Myers Squibb, 2, 5, Gilead, 2, Inova Diagnostics, 2, Janssen, 2, Optum, 2, Pfizer, 2, ReCor, 2.
Background/Purpose: Nintedanib and pirfenidone are antifibrotic drugs indicated for the treatment of idiopathic pulmonary fibrosis (IPF) and other forms of progressive pulmonary fibrosis. Antifibrotics have also been tested in RA-associated interstitial lung disease (ILD) within two recent randomized clinical trials (RCT). However, there are limited real-world studies regarding the use of antifibrotics, and none in RA-ILD. RA-ILD patients may be different from IPF patients since they often need concomitant immunosuppression for articular disease. Our aim was to investigate the tolerability and effectiveness of antifibrotics in a real-world cohort of patients with RA-ILD.
Methods: In this retrospective cohort study, we identified RA-ILD patients initiating antifibrotics at a large multi-hospital healthcare system. We used electronic query to find all patients with at least two RA diagnosis codes and a prescription for either nintedanib or pirfenidone (2014-2023). We then performed medical record review to confirm all patients met 2010 ACR/EULAR classification criteria for RA and had definite RA-ILD by Bongartz criteria. Data regarding adverse events (AEs), tolerability, pulmonary function test (PFT) results, and clinical data were collected. Drug retention was estimated using a Kaplan-Meier curve. A linear mixed model with random intercept was used to compare the within-patient trajectory of the percent predicted forced vital capacity (%FVC) within 18-months before to 18-months after antifibrotic initiation among those with these PFT data.
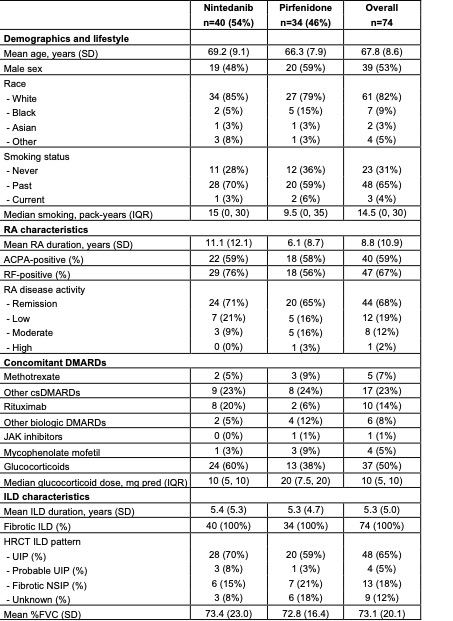
Results: We analyzed 74 patients with RA-ILD that initiated antifibrotics (mean age 67.8 years, 53% male); 40 patients initiated nintedanib and 34 initiated pirfenidone (Table 1). Median follow-up was 89 weeks (min 4, max 387). AEs were reported in 41 (55%) patients, with gastrointestinal (GI) AEs (n=30) being most common, followed by disease progression (n=6), rash (n=3), and hepatitis (n=2). The initial antifibrotic was discontinued in 34 (46%) patients due to: GI AEs (n=19), rash (n=3), transaminitis (n=2), and financial reasons (n=1). The median drug survival was 147.7 weeks (95%CI 79.1, NA; Figure 1). There was no difference in drug retention between nintedanib and pirfenidone (p=0.68). A second antifibrotic was prescribed in 14 patients, with 4 discontinuations. Change of %FVC trajectory was analyzed for 49 patients with available PFTs within 18 months pre- and post-antifibrotic (median number of PFT/patient = 4). There was a statistical difference in the estimated %FVC slope after initiation (-0.3% per year compared to -6.2% per year before initiation, p=0.031, Figure 2). Twenty-six patients (35%) died (17 due to ILD) and 4 (5%) had lung transplantation during follow-up.
Conclusion: In this first real-world study of antifibrotic use in RA-ILD, AEs were frequently reported, particularly GI, and discontinuation was common (46% compared to 20% for nintedanib and 24% for pirfenidone in their respective RCTs). However, antifibrotic initiation was associated with a modestly improved trajectory in %FVC. Death and/or lung transplant were frequent, emphasizing the need for additional safe and effective RA-ILD treatment options.

Table 1. Characteristics of patients with RA-ILD at initiation of initial antifibrotic medication.
ACPA: anti-citrullinated peptides proteins, cs and bDMARDs: conventional and biologic disease modifying drugs, DLCO: diffusing lung capacity of the lungs for carbon monoxide, FVC: forced vital capacity, HRCT: high resolution computed tomography, ILD: Interstitial lung disease, NSIP: non-specific interstitial pneumonia, RA: rheumatoid arthritis, RF: rheumatoid factor, UIP: usual interstitial pneumonia.
ACPA: anti-citrullinated peptides proteins, cs and bDMARDs: conventional and biologic disease modifying drugs, DLCO: diffusing lung capacity of the lungs for carbon monoxide, FVC: forced vital capacity, HRCT: high resolution computed tomography, ILD: Interstitial lung disease, NSIP: non-specific interstitial pneumonia, RA: rheumatoid arthritis, RF: rheumatoid factor, UIP: usual interstitial pneumonia.
.jpg)
Figure 1. Kaplan-Meier curve for retention of initial antifibrotic used to treat RA-ILD.
.jpg)
Figure 2. Trajectories of %FVC before and after initial antifibrotic prescription for RA-ILD. The shaded grey represents the 95%CI of the %FVC trajectory. The dotted line represents the hypothetical decline in %FVC based on %FVC values prior to antifibrotic initiation. Panel A represents patients that initiated either antifibrotic (nintedanib or pirfenidone), Panel B represents patients that initiated nintedanib, and Panel C represents patients that initiated pirfenidone. Trajectories were estimated using a linear mixed model with random intercept with a knot at initial antifibrotic prescription (time 0). FVC: forced vital capacity.
P. Juge: None; K. Hayashi: None; G. McDermott: None; K. Vanni: None; E. Kowalski: None; G. Qian: None; K. Bade: None; A. Saavedra: None; P. Dieudé: AbbVie, 2, 6, Boehringer Ingelheim, 1, 2, 6, Bristol-Myers Squibb, 1, 2, 5, 6, Chugai, 5, Galapagos, 5, 6, Janssen, 2, 6, Novartis, 2, Pfizer, 1, 2, 5, 6; P. Dellaripa: None; T. Doyle: None; J. Sparks: AbbVie, 2, Amgen, 2, Boehringer Ingelheim, 2, Bristol-Myers Squibb, 2, 5, Gilead, 2, Inova Diagnostics, 2, Janssen, 2, Optum, 2, Pfizer, 2, ReCor, 2.



