Abstract Session
Rheumatoid arthritis (RA)
Session: Abstracts: RA – Diagnosis, Manifestations, & Outcomes II: Comorbidities (1627–1632)
1631: Comparison of Peripheral Biomarker Profiles Across Unique Multimorbidity Patterns in Rheumatoid Arthritis
Monday, November 13, 2023
3:00 PM - 3:10 PM PT
Location: Ballroom 20D
- CP
Chloe Peyton, MD
University of Nebraska Medical Center
Omaha, NE, United StatesDisclosure information not submitted.
Presenting Author(s)
Chloe Peyton1, Tate Johnson1, Joshua Baker2, Yangyuna Yang1, Punyasha Roul1, Michael Duryee1, Jill Poole1, Geoffrey Thiele1, Ted R Mikuls3 and Bryant England1, 1University of Nebraska Medical Center, Omaha, NE, 2University of Pennsylvania, Philadelphia, PA, 3Division of Rheumatology and Immunology, University of Nebraska Medical Center, Omaha, NE
Background/Purpose: Rheumatoid arthritis (RA) predisposes affected individuals to develop multiple chronic conditions (i.e., multimorbidity). Mechanisms underlying multimorbidity onset in RA are incompletely understood, particularly for different multimorbidity patterns. We examined how peripheral biomarker profiles encompassing pro-inflammatory, autoantibody, tissue remodeling, and metabolic measures are associated with the clinical expression of distinct multimorbidity patterns.
Methods: From a multicenter, prospective cohort of U.S. Veterans with RA fulfilling ACR classification criteria (Veterans Affairs Rheumatoid Arthritis registry), we measured 61 peripheral biomarkers including 37 pro-inflammatory cytokines/chemokines, 4 matrix metalloproteinases, rheumatoid factor (RF), anti-CCP antibody, 12 antibodies to malondialdehyde-acetaldehyde (MAA), 3 adipokines, and 3 alarmins by the MesoScale platform, ELISA, or nephelometry on serum or plasma from registry enrollment. Principal component analysis (PCA) was performed to generate distinct biomarker profiles (PCs) from log-transformed and normalized analyte concentrations. The presence of forty-four pre-defined conditions at enrollment was determined using diagnostic codes from outpatient and inpatient encounters within linked administrative claims. Four previously developed multimorbidity patterns were then applied (mental health and substance abuse; metabolic; cardiovascular; chronic pain; England et al. Arthritis Care Res, 2023). Cross-sectional associations of PC scores with multimorbidity patterns were assessed using multivariable logistic regression models adjusting for age, sex, race, and smoking status.
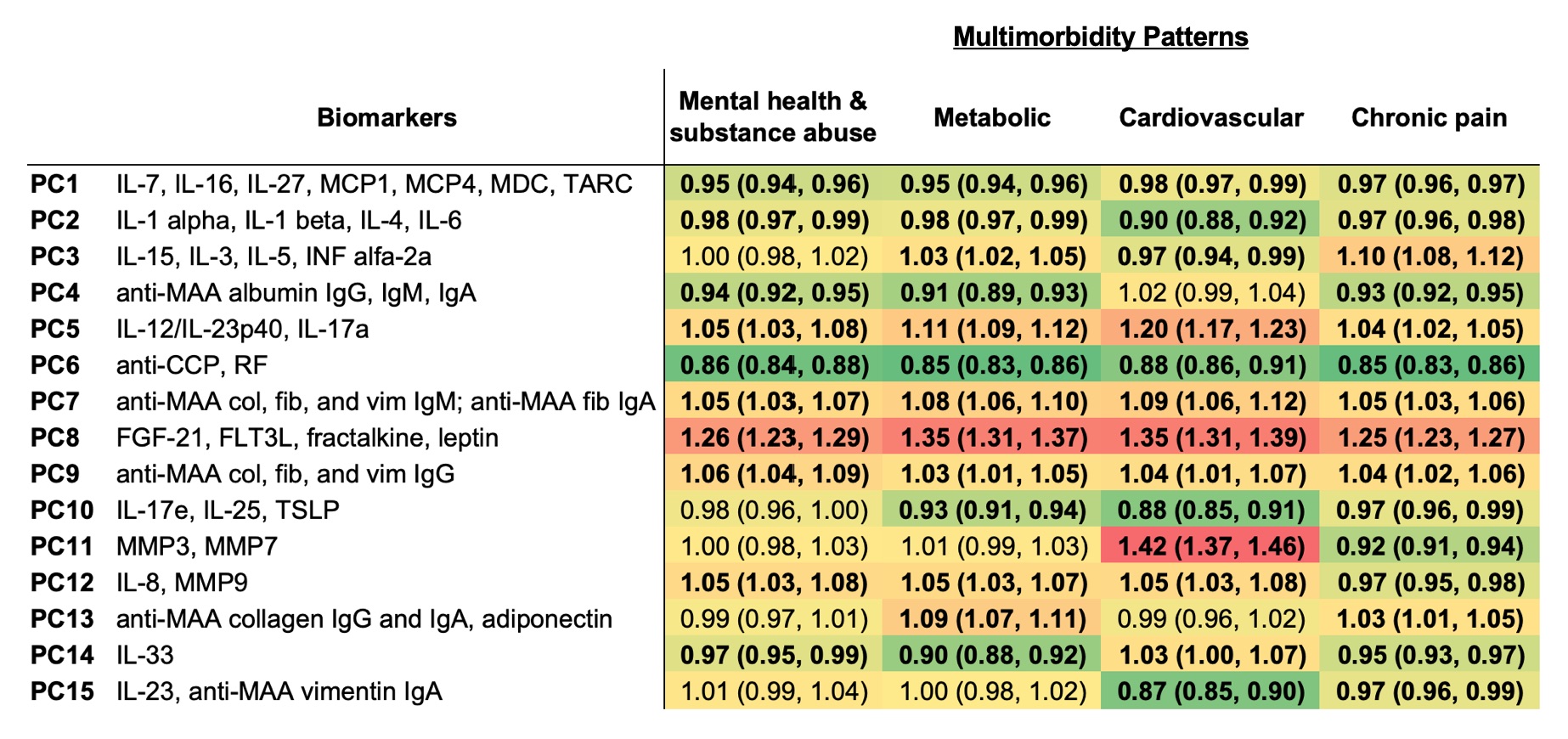
Results: Among 2,007 participants with RA (89% male, mean age 64 years), 64% had metabolic, 47% had chronic pain, 21% had mental health and substance abuse, and 13% had cardiovascular multimorbidity. PCA identified15 unique peripheral biomarker PCs with Eigenvalues >1 that together explained 66% of the variance (Figure 1). PC8 scores, primarily characterized by adipokines (FGF-21, FLT3L, fractalkine, leptin), were positively associated with all multimorbidity patterns (aORs range: 1.25-1.35). In contrast, PC6 scores (anti-CCP and RF) were negatively associated with all multimorbidity patterns (aORs range: 0.85-0.88). Cardiovascular multimorbidity was positively associated with PC5 (IL12/IL-23p40, IL-17a) and PC11 (MMP3 and MMP7), but negatively associated with PC10 (IL-17e, IL-25, TSLP) and PC15 (IL-23, anti-MAA vimentin IgA). Several additional associations of smaller magnitude were observed between other PCs and multimorbidity patterns (Table 1).
Conclusion: Biomarker profiles consisting of cytokines/chemokines, MMPs, RA autoantibodies, adipokines, and alarmins were differentially associated with multimorbidity patterns in a large, RA cohort. These findings may indicate shared pathophysiologic mechanisms underlying multimorbidity in RA, such as obesity, metabolic dysfunction, and tissue remodeling. Further, they highlight pathways that could potentially be targeted to reduce multimorbidity burden or prevent its onset.
.jpg)
C. Peyton: None; T. Johnson: None; J. Baker: CorEvitas, 2, Cumberland Pharma, 2, Horizon Pharmaceuticals, 5; Y. Yang: None; P. Roul: None; M. Duryee: None; J. Poole: AstraZeneca, 12, I have received no monies. I received anti-IL-33 monoclonal antibody from AstraZeneca for animal research sutdies.; G. Thiele: None; T. Mikuls: Elsevier, 9, Horizon Therapeutics, 2, 5, Pfizer, 2, Sanofi, 2, UCB Pharma, 2, Wolters Kluwer Health (UpToDate), 9; B. England: Boehringer-Ingelheim, 2, 5.
Background/Purpose: Rheumatoid arthritis (RA) predisposes affected individuals to develop multiple chronic conditions (i.e., multimorbidity). Mechanisms underlying multimorbidity onset in RA are incompletely understood, particularly for different multimorbidity patterns. We examined how peripheral biomarker profiles encompassing pro-inflammatory, autoantibody, tissue remodeling, and metabolic measures are associated with the clinical expression of distinct multimorbidity patterns.
Methods: From a multicenter, prospective cohort of U.S. Veterans with RA fulfilling ACR classification criteria (Veterans Affairs Rheumatoid Arthritis registry), we measured 61 peripheral biomarkers including 37 pro-inflammatory cytokines/chemokines, 4 matrix metalloproteinases, rheumatoid factor (RF), anti-CCP antibody, 12 antibodies to malondialdehyde-acetaldehyde (MAA), 3 adipokines, and 3 alarmins by the MesoScale platform, ELISA, or nephelometry on serum or plasma from registry enrollment. Principal component analysis (PCA) was performed to generate distinct biomarker profiles (PCs) from log-transformed and normalized analyte concentrations. The presence of forty-four pre-defined conditions at enrollment was determined using diagnostic codes from outpatient and inpatient encounters within linked administrative claims. Four previously developed multimorbidity patterns were then applied (mental health and substance abuse; metabolic; cardiovascular; chronic pain; England et al. Arthritis Care Res, 2023). Cross-sectional associations of PC scores with multimorbidity patterns were assessed using multivariable logistic regression models adjusting for age, sex, race, and smoking status.
Results: Among 2,007 participants with RA (89% male, mean age 64 years), 64% had metabolic, 47% had chronic pain, 21% had mental health and substance abuse, and 13% had cardiovascular multimorbidity. PCA identified15 unique peripheral biomarker PCs with Eigenvalues >1 that together explained 66% of the variance (Figure 1). PC8 scores, primarily characterized by adipokines (FGF-21, FLT3L, fractalkine, leptin), were positively associated with all multimorbidity patterns (aORs range: 1.25-1.35). In contrast, PC6 scores (anti-CCP and RF) were negatively associated with all multimorbidity patterns (aORs range: 0.85-0.88). Cardiovascular multimorbidity was positively associated with PC5 (IL12/IL-23p40, IL-17a) and PC11 (MMP3 and MMP7), but negatively associated with PC10 (IL-17e, IL-25, TSLP) and PC15 (IL-23, anti-MAA vimentin IgA). Several additional associations of smaller magnitude were observed between other PCs and multimorbidity patterns (Table 1).
Conclusion: Biomarker profiles consisting of cytokines/chemokines, MMPs, RA autoantibodies, adipokines, and alarmins were differentially associated with multimorbidity patterns in a large, RA cohort. These findings may indicate shared pathophysiologic mechanisms underlying multimorbidity in RA, such as obesity, metabolic dysfunction, and tissue remodeling. Further, they highlight pathways that could potentially be targeted to reduce multimorbidity burden or prevent its onset.
.jpg)
Figure 1. Scree plot from principal component analysis of 61 pro-inflammatory, autoantibody, tissue remodeling, and metabolic measures.

Table 1. Associations of peripheral biomarker profiles with multimorbidity patterns in rheumatoid arthritis. Values shown are odds ratios, with 95% confidence intervals, per 1-unit change in principal component (PC) score from logistic regression models adjusting for age, sex, race, and smoking status. Biomarkers listed are those with significant loadings (>0.3) onto the PC. P < 0.05 indicated in bold. Positive associations indicated in red and negative associations indicated in green with color gradient based on effect size.
C. Peyton: None; T. Johnson: None; J. Baker: CorEvitas, 2, Cumberland Pharma, 2, Horizon Pharmaceuticals, 5; Y. Yang: None; P. Roul: None; M. Duryee: None; J. Poole: AstraZeneca, 12, I have received no monies. I received anti-IL-33 monoclonal antibody from AstraZeneca for animal research sutdies.; G. Thiele: None; T. Mikuls: Elsevier, 9, Horizon Therapeutics, 2, 5, Pfizer, 2, Sanofi, 2, UCB Pharma, 2, Wolters Kluwer Health (UpToDate), 9; B. England: Boehringer-Ingelheim, 2, 5.



