Abstract Session
Antiphospholipid Syndrome
Session: Abstracts: Antiphospholipid Syndrome (1603–1608)
1607: Autoantibodies to Transcription Factor a Mitochondria Link Mitochondrial Damage and Thrombosis in Systemic Lupus Erythematosus
Monday, November 13, 2023
3:00 PM - 3:10 PM PT
Location: Room 25A-C
- EG
Eduardo Gomez-Banuelos, MD, PhD (he/him/his)
The Johns Hopkins University
Baltimore, MD, United StatesDisclosure information not submitted.
Presenting Author(s)
Eduardo Gomez-Banuelos1, Alessandra Ida Celia2, Maria Isabel Trejo Zambrano3, Merlin Paz3, Erika Darrah3, Daniel Goldman4, Michelle Petri4 and Felipe Andrade3, 1The Johns Hopkins University, Baltimore, MD, 2John Hopkins University of Medicine, Rome, Italy, 3Johns Hopkins University, Baltimore, MD, 4Department of Medicine, Division of Rheumatology, Johns Hopkins University School of Medicine, Timonium, MD
Background/Purpose: Upon activation with interferon (IFN) and RNP-immune complexes, defective mitophagy in neutrophils results in the release of mtDNA in complex with transcription factor A mitochondria (TFAM), leading to immune activation in SLE. TFAM is a member of the high-mobility group (HMG) family of DNA-binding proteins, which is essential for mtDNA transcription and for packaging mtDNA into nucleoids. Extracellular TFAM released from necrotic cells is also a DAMP. While characterizing novel autoantigens expressed by neutrophils, we identified TFAM as a target of antibodies in SLE. The purpose of this study was to investigate the relationship between anti-TFAM antibodies and clinical and transcriptional markers of disease activity in SLE
Methods: Anti-neutrophil antibodies in SLE sera were detected by indirect immunofluorescence (IIF). Anti-TFAM antibodies were screened by ELISA in sera from 98 healthy controls and 158 SLE patients from the "Study of biological Pathways, Disease Activity and Response markers in patients with Systemic Lupus Erythematosus" (SPARE). SPARE is a 2-year prospective cohort of adult patients for which extensive clinical, serologic, and whole blood transcriptional data is available. Binding of anti-TFAM antibodies to activated (GM-CSF + LPS) and necrotic neutrophils was determined by IIF.
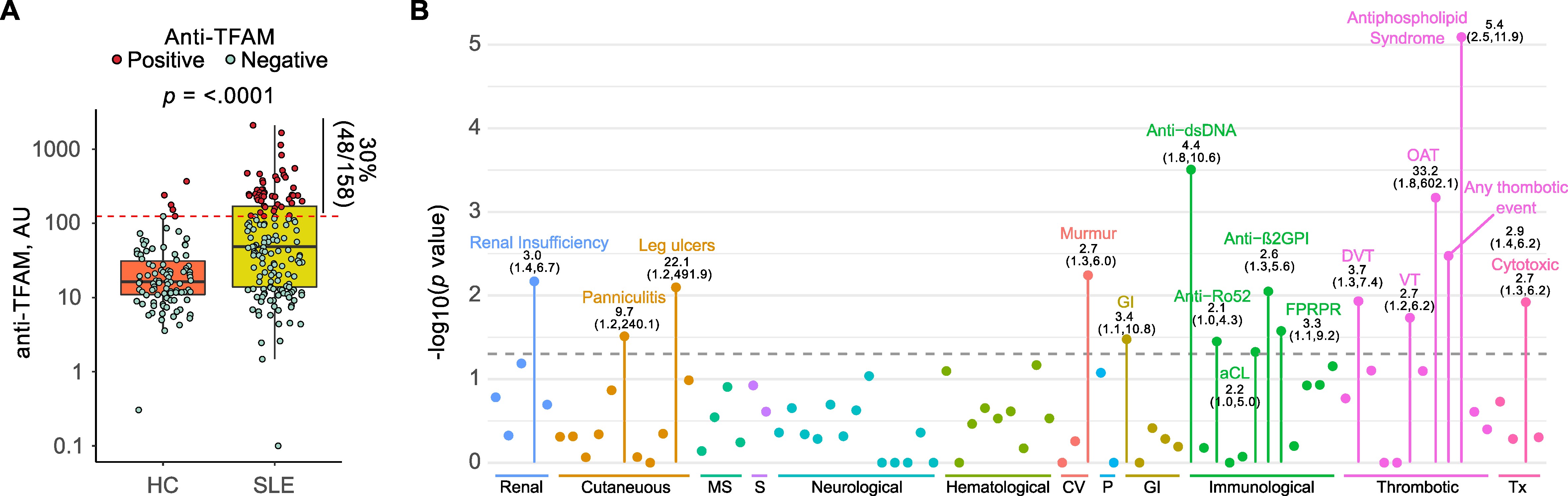
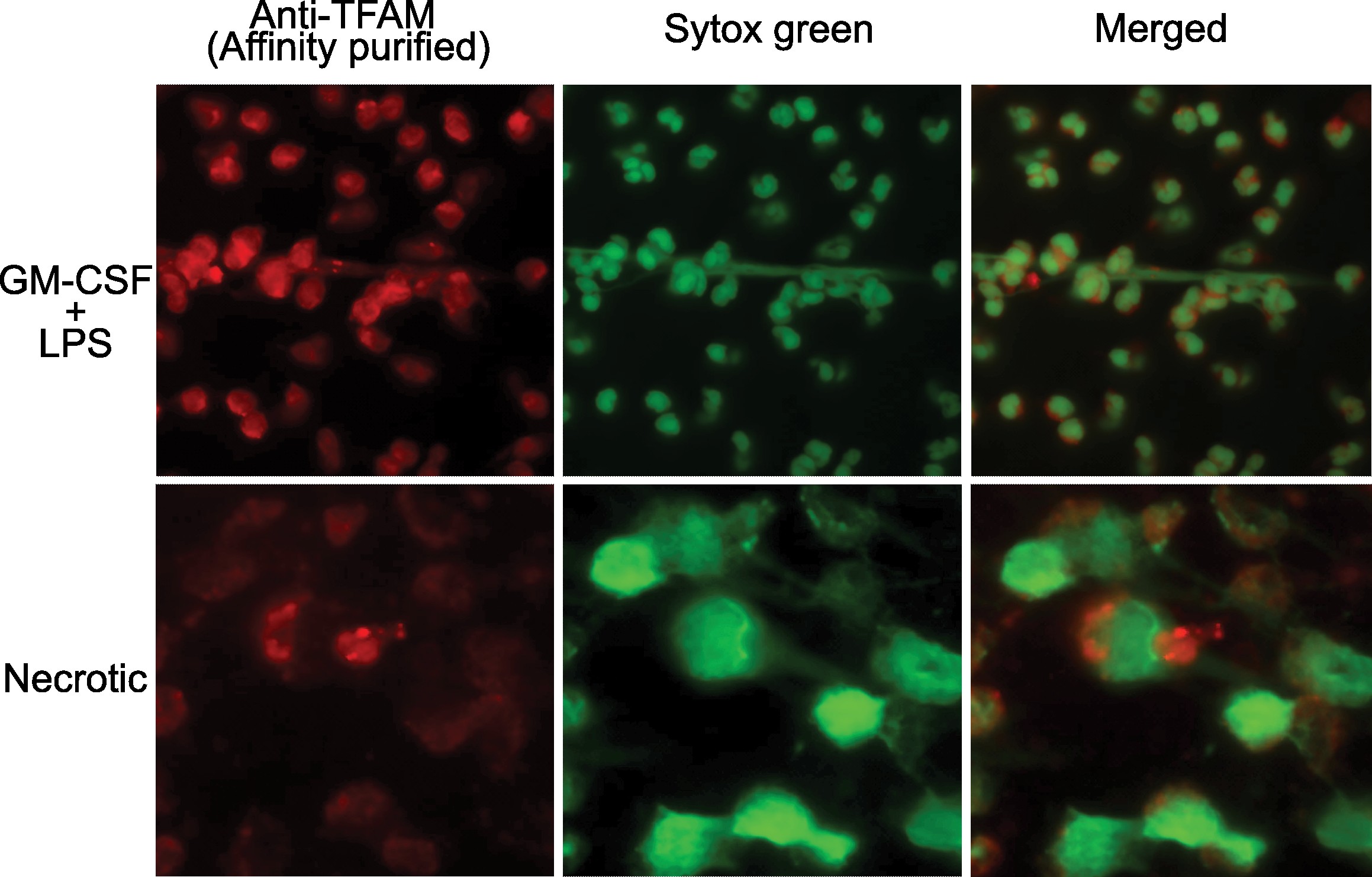
Results: A subset of antibodies in SLE serum co-localizes with TFAM in neutrophils. The existence of anti-TFAM antibodies in SLE was confirmed by ELISA and immunoblotting. Thirty percent (48/158) of SLE patients were positive for anti-TFAM antibodies (Fig. 1A). Anti-TFAM antibodies were significantly associated with antibodies to dsDNA, cardiolipin (aCL), and β2-glycoprotein I (β2GPI) (Fig 1B). Anti-TFAM antibodies were strongly associated with history of thrombotic events, OR (95% CI) 2.9 (1.4,6.2), and secondary antiphospholipid syndrome (APS), OR (95% CI) 5.4 (2.5,11.9), independently of aCL and anti- β2GPI (Fig. 1B). Unlike antibodies to dsDNA and RNPs, anti-TFAM antibodies were not associated with blood transcriptional markers of SLE disease activity, including the IFN signature, supporting a primary role of anti-TFAM antibodies in thrombosis rather than disease flaring. Anti-TFAM antibodies bind to TFAM-decorated DNA released from activated and necrotic neutrophils (Fig. 2), which is a mechanism that may contribute to thrombosis.
Conclusion: TFAM is novel autoantigen in SLE, which is associated with thrombosis and APS independently of aCL in SLE. These data support a link between mitochondrial damage and thrombosis in SLE.
E. Gomez-Banuelos: None; A. Celia: None; M. Trejo Zambrano: None; M. Paz: None; E. Darrah: None; D. Goldman: None; M. Petri: Alexion, 1, Amgen, 1, AnaptysBio, 1, Annexon Bio, 1, Argenx, 1, Arhros-Focus Med/Ed, 6, AstraZeneca, 1, 5, Aurinia, 1, 5, 6, Axdev, 1, Biogen, 1, Boxer Capital, 2, Cabaletto Bio, 2, Caribou Biosciences, 2, CVS Health, 1, Eli Lilly, 1, 5, Emergent Biosolutions, 1, Exagen, 5, Exo Therapeutics, 2, Gilead Biosciences, 2, GlaxoSmithKlein(GSK), 1, 5, 6, Horizon Therapeutics, 2, Idorsia Pharmaceuticals, 2, IQVIA, 1, Janssen, 1, 5, Kira Pharmaceuticals, 2, MedShr, 6, Merck/EMD Serono, 1, Momenta Pharmaceuticals, 2, Nexstone Immunology, 2, Nimbus Lakshmi, 2, Proviant, 2, Sanofi, 2, Sinomab Biosciences, 2, Thermofisher, 5, UCB, 2; F. Andrade: None.
Background/Purpose: Upon activation with interferon (IFN) and RNP-immune complexes, defective mitophagy in neutrophils results in the release of mtDNA in complex with transcription factor A mitochondria (TFAM), leading to immune activation in SLE. TFAM is a member of the high-mobility group (HMG) family of DNA-binding proteins, which is essential for mtDNA transcription and for packaging mtDNA into nucleoids. Extracellular TFAM released from necrotic cells is also a DAMP. While characterizing novel autoantigens expressed by neutrophils, we identified TFAM as a target of antibodies in SLE. The purpose of this study was to investigate the relationship between anti-TFAM antibodies and clinical and transcriptional markers of disease activity in SLE
Methods: Anti-neutrophil antibodies in SLE sera were detected by indirect immunofluorescence (IIF). Anti-TFAM antibodies were screened by ELISA in sera from 98 healthy controls and 158 SLE patients from the "Study of biological Pathways, Disease Activity and Response markers in patients with Systemic Lupus Erythematosus" (SPARE). SPARE is a 2-year prospective cohort of adult patients for which extensive clinical, serologic, and whole blood transcriptional data is available. Binding of anti-TFAM antibodies to activated (GM-CSF + LPS) and necrotic neutrophils was determined by IIF.
Results: A subset of antibodies in SLE serum co-localizes with TFAM in neutrophils. The existence of anti-TFAM antibodies in SLE was confirmed by ELISA and immunoblotting. Thirty percent (48/158) of SLE patients were positive for anti-TFAM antibodies (Fig. 1A). Anti-TFAM antibodies were significantly associated with antibodies to dsDNA, cardiolipin (aCL), and β2-glycoprotein I (β2GPI) (Fig 1B). Anti-TFAM antibodies were strongly associated with history of thrombotic events, OR (95% CI) 2.9 (1.4,6.2), and secondary antiphospholipid syndrome (APS), OR (95% CI) 5.4 (2.5,11.9), independently of aCL and anti- β2GPI (Fig. 1B). Unlike antibodies to dsDNA and RNPs, anti-TFAM antibodies were not associated with blood transcriptional markers of SLE disease activity, including the IFN signature, supporting a primary role of anti-TFAM antibodies in thrombosis rather than disease flaring. Anti-TFAM antibodies bind to TFAM-decorated DNA released from activated and necrotic neutrophils (Fig. 2), which is a mechanism that may contribute to thrombosis.
Conclusion: TFAM is novel autoantigen in SLE, which is associated with thrombosis and APS independently of aCL in SLE. These data support a link between mitochondrial damage and thrombosis in SLE.

Anti-TFAM antibodies are associated with thrombosis and APS in SLE. (A) Serum levels of anti-TFAM antibodies in patients with SLE (n=158) and Healthy controls (HC, n-98). Dots in red represent subjects positive for anti-TFAM. (B) Clinical and serological associations of anti-TFAM antibodies in SLE. Each dot represents the p value for the indicated SLE feature. Only features with a p value < 0.05 are labeled. P values were obtained with Fisher’s exact test. Odds ratios and 95% CI, OR (95%CI), are indicated below each label.

Anti-TFAM antibodies recognize extracellular TFAM from GM-CSF + LPS activated (upper row) and necrotic neutrophils (lower row). Anti-TFAM antibodies (red) were purified by affinity chromatography, using recombinant TFAM, from a pool (n=3) of anti-TFAM positive SLE sera. DNA was stained using Sytox Green (Green channel).
E. Gomez-Banuelos: None; A. Celia: None; M. Trejo Zambrano: None; M. Paz: None; E. Darrah: None; D. Goldman: None; M. Petri: Alexion, 1, Amgen, 1, AnaptysBio, 1, Annexon Bio, 1, Argenx, 1, Arhros-Focus Med/Ed, 6, AstraZeneca, 1, 5, Aurinia, 1, 5, 6, Axdev, 1, Biogen, 1, Boxer Capital, 2, Cabaletto Bio, 2, Caribou Biosciences, 2, CVS Health, 1, Eli Lilly, 1, 5, Emergent Biosolutions, 1, Exagen, 5, Exo Therapeutics, 2, Gilead Biosciences, 2, GlaxoSmithKlein(GSK), 1, 5, 6, Horizon Therapeutics, 2, Idorsia Pharmaceuticals, 2, IQVIA, 1, Janssen, 1, 5, Kira Pharmaceuticals, 2, MedShr, 6, Merck/EMD Serono, 1, Momenta Pharmaceuticals, 2, Nexstone Immunology, 2, Nimbus Lakshmi, 2, Proviant, 2, Sanofi, 2, Sinomab Biosciences, 2, Thermofisher, 5, UCB, 2; F. Andrade: None.



