Abstract Session
Osteoarthritis (OA) and related disorders
Session: Abstracts: Osteoarthritis & Joint Biology – Basic Science (1591–1596)
1594: Oral Delivery of Δ9-Tetrahydrocannabinol Provides Symptom and Disease Modification in a Mouse Model of Knee Osteoarthritis
Monday, November 13, 2023
2:45 PM - 2:55 PM PT
Location: Room 30D-E
- JR
Jason Rockel, PhD
Schroeder Arthritis Institute, University Health Network
Toronto, ON, CanadaDisclosure information not submitted.
Presenting Author(s)
Jason rockel1, Anca Maglaviceanu2, Helena Fetter Filippini3, Ewa Wasilewski4, Melissa M. Lewis-Bakker4, Sarah Gabrial1, Evgeny Rossomacha1, Johana Garcia1, Nizar Mahomed5, Timothy Leroux5, Hance Clarke6, Robert P. Bonin3, Lakshmi P. Kotra7 and Mohit Kapoor1, 1Division of Orthopaedics, Osteoarthritis Research Program, Schroeder Arthritis Institute, and Krembil Research Institute, University Health Network, Toronto, ON, Canada, 2Division of Orthopaedics, Osteoarthritis Research Program, Schroeder Arthritis Institute, and Krembil Research Institute, University Health Network; Department of Laboratory Medicine and Pathobiology, University of Toronto, Toronto, ON, Canada, 3Leslie Dan Faculty of Pharmacy, University of Toronto, Toronto, ON, Canada, 4Krembil Research Institute, and Toronto Western Hospital, University Health Network, Toronto, ON, Canada, 5Division of Orthopaedics, Osteoarthritis Research Program, Schroeder Arthritis Institute, Krembil Research Institute, and Toronto Western Hospital, University Health Network, Toronto, ON, Canada, 6Krembil Research Institute, and Department of Anesthesia and Pain Management, Toronto General Hospital, University Health Network, Toronto, ON, Canada, 7Krembil Research Institute, University Health Network; Leslie Dan Faculty of Pharmacy, University of Toronto, Toronto, ON, Canada
Background/Purpose: Osteoarthritis (OA) results in joint pain, cartilage degeneration, and synovitis. Recent studies suggest that some OA patients are self-medicating with cannabis. Δ9-tetrahydrocannabinol (THC) is a prominent psychoactive phytocannabinoid in cannabis that can signal in joint cells, including chondrocytes and fibroblast-like synoviocytes (FLS). Currently, no studies link THC to disease modification in OA. We investigated effects and mechanisms of action of THC on pain and disease modification in pre-clinical models of knee OA.
Methods: OA was induced in male C57BL/6 mice by destabilization of the medial meniscus (DMM) or monosodium iodoacetate (MIA; 0.5 mg) and mice were administered THC (0, 1, 5 or 10 mg/kg) orally 5 days/week for 9 or 3 weeks, respectively. Von Frey tests were used to evaluate mechanical allodynia (pain) and open field tests were used to evaluate locomotion and anxiety. DMM knee joints were evaluated for cartilage degeneration/synovitis (OARSI scoring) and Ki67, αSMA, and collagen degradation (C2C) expression (immunohistochemistry). Human FLS and chondrocytes were cultured with 1 mM THC for 48h. RNA was sequenced to determine differentially expressed genes (DEGs; FDR-adjusted p< 0.05; absolute FC≥1.5) and DEG-enriched pathways. Pathway and transcription factor enrichment analyses were used to determine putative cellular mechanisms and transcriptional regulators modified by THC.
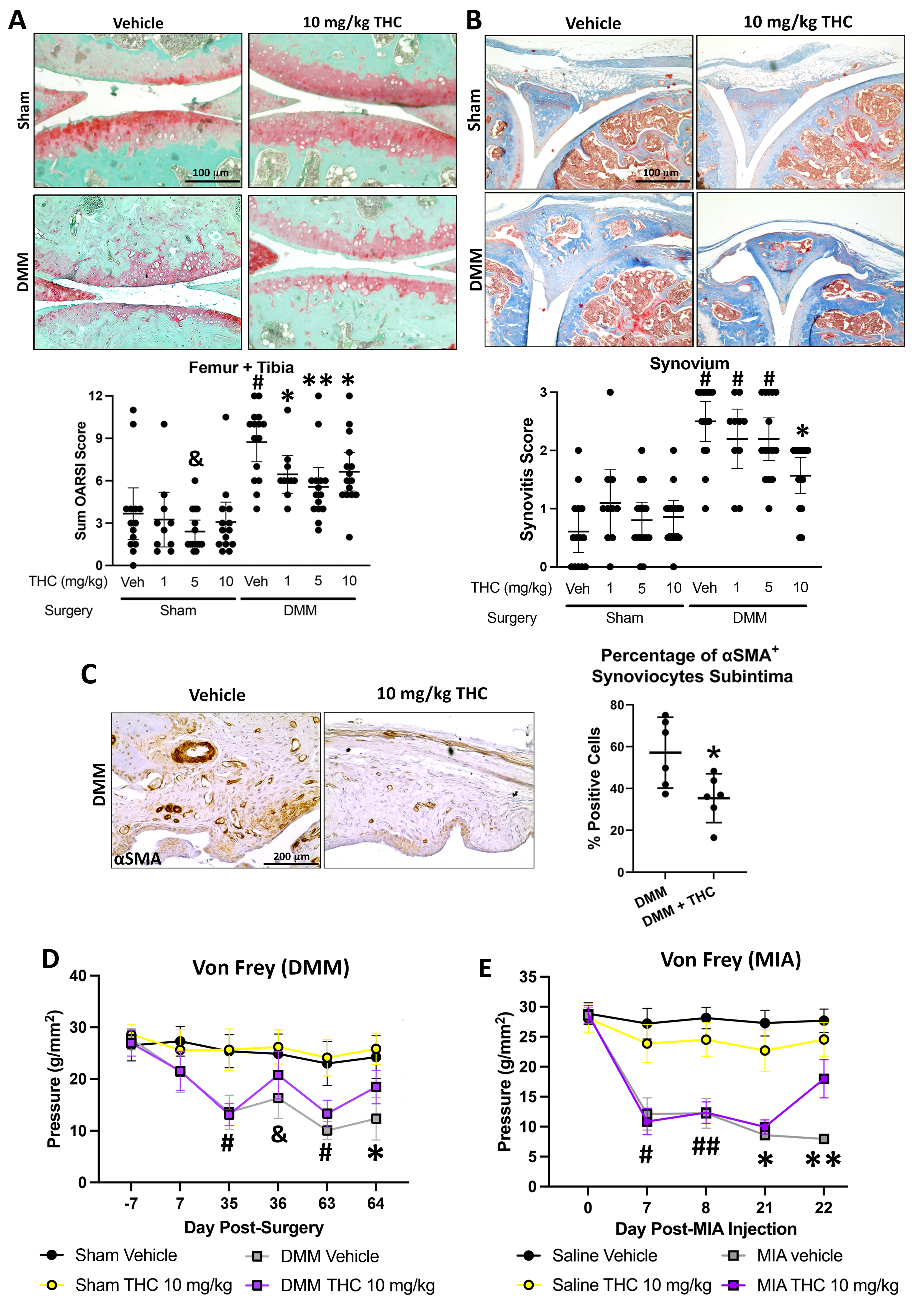
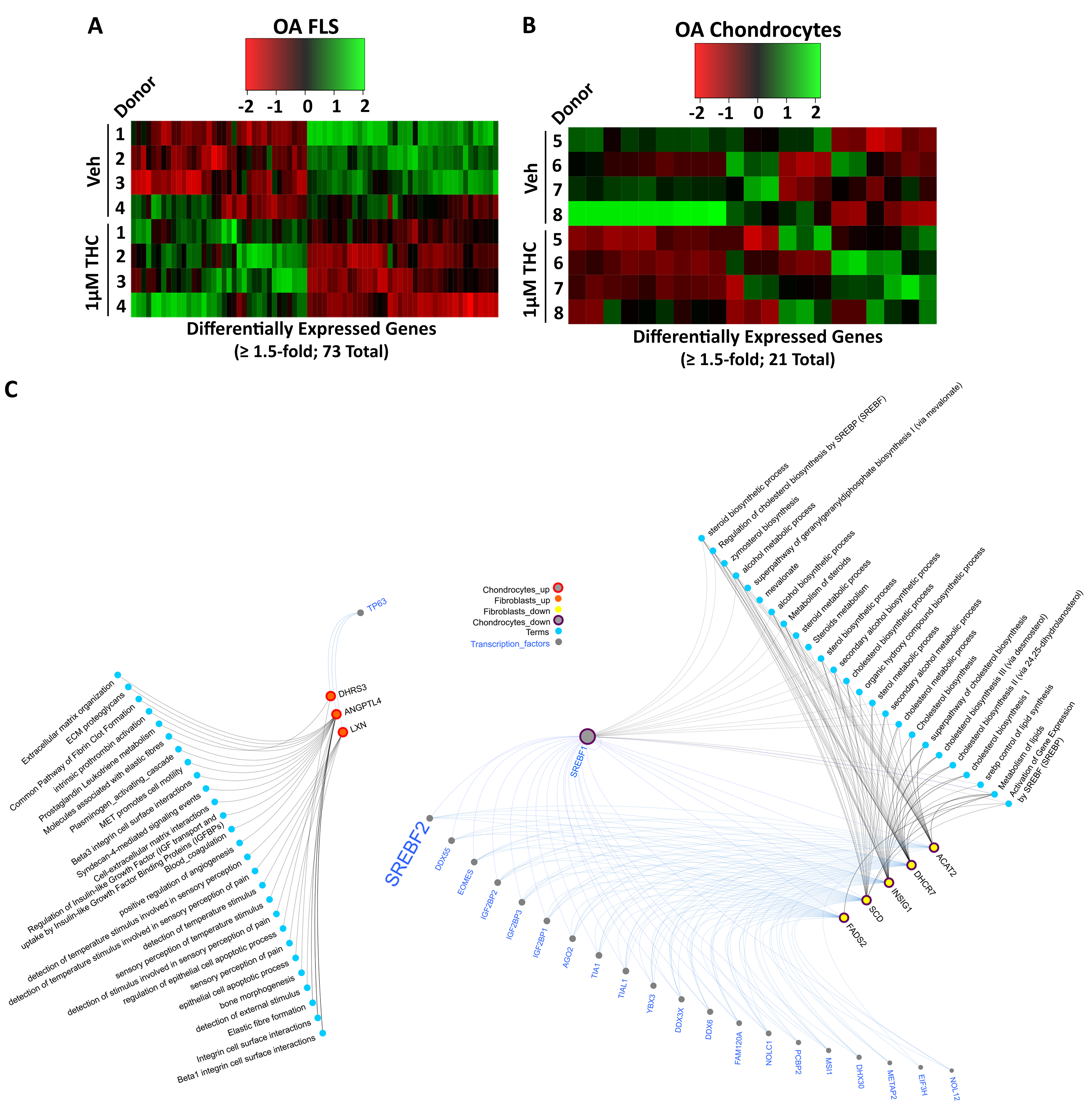
Results: In DMM mice, all THC doses reduced cartilage degeneration. 10 mg/kg THC also reduced synovitis (n=14-15/group) and synovial αSMA expression, but not Ki67 or C2C expression (n=6/group). Administration of 5 or 10 mg/kg THC in DMM (n=14-15/group) or MIA (n=10 25/group) mice reduced pain, particularly with 10 mg/kg administration (Fig. 1). Locomotion or anxiety were not modified longitudinally by THC administration. In cultured OA FLS (n=4), RNA sequencing identified 73 DEGs (35 up, 38 down) after treatment with 1 mM THC for 48h compared to vehicle. In cultured OA chondrocytes (n=4), 21 DEGs (9 up, 12 down) were identified after treatment with 1 mM THC compared to vehicle. Computational biology analyses found extracellular matrix (ECM)-related pathways were enriched in the upregulated DEGs in both fibroblasts and chondrocytes, while lipid/steroid/cholesterol-related pathways were enriched in the downregulated DEGs in both cell types. Three genes were commonly upregulated while 5 genes were commonly downregulated in FLS and chondrocytes by THC treatment. From FLS and chondrocyte downregulated DEGs, 22 common putative transcription factors were identified as potential regulators, with SREBF2 as a candidate of interest (Fig. 2).
Conclusion: Oral administration of 10 mg/kg THC reduced cartilage degeneration synovitis and synovial αSMA expression in DMM mouse knee joints, and modified pain in both DMM and MIA OA models. THC treatment of human OA FLS or chondrocytes modified expression of genes associated with ECM and lipid-based pathways, with common genes and putative regulatory transcription factors identified, including SREBF2. We are currently investigating contributions of SREBF2 to effects of THC on joint cells in vitro and mechanisms of pain in vivo.
J. rockel: None; A. Maglaviceanu: None; H. Fetter Filippini: None; E. Wasilewski: None; M. Lewis-Bakker: None; S. Gabrial: None; E. Rossomacha: None; J. Garcia: None; N. Mahomed: AIC, 4, ARTHUR HEALTH CORP, 3; T. Leroux: Depuy Synthes, 2, Smith and Nephew, 2, Stryker, 2; H. Clarke: AbbVie/Abbott, 6, Managing Life, 2, Master Clinician Alliance, 6, Ontario Ministry of Health and Long Term Care, 5, Shoppers Drug Mart, 5; R. Bonin: None; L. Kotra: None; M. Kapoor: None.
Background/Purpose: Osteoarthritis (OA) results in joint pain, cartilage degeneration, and synovitis. Recent studies suggest that some OA patients are self-medicating with cannabis. Δ9-tetrahydrocannabinol (THC) is a prominent psychoactive phytocannabinoid in cannabis that can signal in joint cells, including chondrocytes and fibroblast-like synoviocytes (FLS). Currently, no studies link THC to disease modification in OA. We investigated effects and mechanisms of action of THC on pain and disease modification in pre-clinical models of knee OA.
Methods: OA was induced in male C57BL/6 mice by destabilization of the medial meniscus (DMM) or monosodium iodoacetate (MIA; 0.5 mg) and mice were administered THC (0, 1, 5 or 10 mg/kg) orally 5 days/week for 9 or 3 weeks, respectively. Von Frey tests were used to evaluate mechanical allodynia (pain) and open field tests were used to evaluate locomotion and anxiety. DMM knee joints were evaluated for cartilage degeneration/synovitis (OARSI scoring) and Ki67, αSMA, and collagen degradation (C2C) expression (immunohistochemistry). Human FLS and chondrocytes were cultured with 1 mM THC for 48h. RNA was sequenced to determine differentially expressed genes (DEGs; FDR-adjusted p< 0.05; absolute FC≥1.5) and DEG-enriched pathways. Pathway and transcription factor enrichment analyses were used to determine putative cellular mechanisms and transcriptional regulators modified by THC.
Results: In DMM mice, all THC doses reduced cartilage degeneration. 10 mg/kg THC also reduced synovitis (n=14-15/group) and synovial αSMA expression, but not Ki67 or C2C expression (n=6/group). Administration of 5 or 10 mg/kg THC in DMM (n=14-15/group) or MIA (n=10 25/group) mice reduced pain, particularly with 10 mg/kg administration (Fig. 1). Locomotion or anxiety were not modified longitudinally by THC administration. In cultured OA FLS (n=4), RNA sequencing identified 73 DEGs (35 up, 38 down) after treatment with 1 mM THC for 48h compared to vehicle. In cultured OA chondrocytes (n=4), 21 DEGs (9 up, 12 down) were identified after treatment with 1 mM THC compared to vehicle. Computational biology analyses found extracellular matrix (ECM)-related pathways were enriched in the upregulated DEGs in both fibroblasts and chondrocytes, while lipid/steroid/cholesterol-related pathways were enriched in the downregulated DEGs in both cell types. Three genes were commonly upregulated while 5 genes were commonly downregulated in FLS and chondrocytes by THC treatment. From FLS and chondrocyte downregulated DEGs, 22 common putative transcription factors were identified as potential regulators, with SREBF2 as a candidate of interest (Fig. 2).
Conclusion: Oral administration of 10 mg/kg THC reduced cartilage degeneration synovitis and synovial αSMA expression in DMM mouse knee joints, and modified pain in both DMM and MIA OA models. THC treatment of human OA FLS or chondrocytes modified expression of genes associated with ECM and lipid-based pathways, with common genes and putative regulatory transcription factors identified, including SREBF2. We are currently investigating contributions of SREBF2 to effects of THC on joint cells in vitro and mechanisms of pain in vivo.

Fig 1. Oral administration of THC attenuates modifies disease progression and pain in OA-induced mice. Sham or DMM mice received treatment by oral gavage 5 days/week with vehicle [veh; medium chain triglyceride (MCT)] or THC (1, 5, 10 mg/kg) for 9 weeks. Knee joints were collected at 10 weeks post-surgery and stained with (A) Safranin O for OARSI scoring of cartilage degeneration and (B) Masson’s Trichrome for synovitis, respectively (n=10-15/group). Data was analyzed by Kruskal-Wallis tests followed by Mann-Whitney post-hoc tests corrected for multiple comparisons using the method of Benjamini, Kreiger and Yekutieli using R v 3.5.03 with coin package v 1.3-24 to account for ties.*, &, or #, q < 0.05 compared to all other comparison groups with different or no symbols. (C) Representative images and quantification of synovial subintima IHC staining for αSMA in knee joint synovium collected from DMM mice given MCT or 10mg/kg THC treatment by oral gavage for 9 weeks (n=6/group). Data was log transformed prior to analysis by Student’s two-tailed t-tests with Welch’s correction. *, p < 0.05. (D) Sham and DMM-surgical mice (n=10-15/group) or (E) saline and MIA-injected mice (n=10-25/group) were administered vehicle (MCT) or 10 mg/kg THC by oral gavage and were evaluated for mechanical allodynia using the von Frey test. (D & E) Data was log-transformed prior to analysis by repeated measures two-way ANOVA followed by multiple comparison’s tests using the method of Benjamini, Kreiger and Yekutieli to correct for false-discovery rate. (D) #, q < 0.0001 in DMM groups vs Sham groups. &, q < 0.01 for DMM + vehicle compared to Sham + vehicle or Sham + THC groups. *, p< 0.05 for all comparisons except between sham groups. (E)#, p < 0.05 for all comparisons except for MIA + saline vs. MIA + THC. ##, p < 0.01 for all comparisons except for MIA + saline vs. MIA + THC. *, p < 0.05 for all comparisons. **, q < 0.05 for all comparison and q < 0.0001 between MIA + vehicle and MIA + THC.

Fig 2. RNA Sequencing of THC-treated human OA fibroblast-like synoviocytes (FLS) or chondrocytes identifies differentially expressed genes (DEGs) associated with ECM and lipid metabolism pathways. Human OA FLS and chondrocytes were treated with 1 µM THC and was isolated after 48h and subjected to RNA sequencing. (A) Heat map shows the 73 DEGs from human OA FLS and (B) 21 DEGs from human OA chondrocytes stimulated with 1µM THC compared to vehicle-treated controls (veh; 0.5% DMSO) (n=4/group). (C) Network analysis shows combined transcription factors [determined by analysis through Catrin (http://ophid.utoronto.ca/Catrin/index.jsp] and protein-protein interactions (PPIs) with the overlapping DEGs and linked pathways [identified through analysis using pathdip (http://ophid.utoronto.ca/pathDIP/) and Gene Ontology Annotation analysis] in FLS and chondrocytes after THC treatment compared to vehicle-treated cells. Red nodes with red outlines represent upregulated genes and yellow nodes with purple outlines represent the downregulated genes in both FLS and chondrocytes after THC treatment. Grey lines connect genes to interacting pathways (blue nodes) and blue lines connect genes to predicted upstream TFs (grey nodes). Note SREBF2 is a putitive upstream regulator of common genes downregulated in both OA FLS and chondrocytes.
J. rockel: None; A. Maglaviceanu: None; H. Fetter Filippini: None; E. Wasilewski: None; M. Lewis-Bakker: None; S. Gabrial: None; E. Rossomacha: None; J. Garcia: None; N. Mahomed: AIC, 4, ARTHUR HEALTH CORP, 3; T. Leroux: Depuy Synthes, 2, Smith and Nephew, 2, Stryker, 2; H. Clarke: AbbVie/Abbott, 6, Managing Life, 2, Master Clinician Alliance, 6, Ontario Ministry of Health and Long Term Care, 5, Shoppers Drug Mart, 5; R. Bonin: None; L. Kotra: None; M. Kapoor: None.



