Abstract Session
Immunobiology
Session: Abstracts: T Cell Biology & Targets in Autoimmune & Inflammatory Disease (0799–0804)
0799: Human Chimeric Antigen Receptor (CAR)-Tregs Targeting OX40L for Treatment of Systemic Lupus Erythematosus
Sunday, November 12, 2023
4:00 PM - 4:10 PM PT
Location: Room 30D-E
- HW
Holly Wobma, MD, PhD (she/her/hers)
Boston Children's Hospital
Boston, MA, United StatesDisclosure information not submitted.
Presenting Author(s)
Holly Wobma1, Xianliang Rui2, Francesca Alvarez-Calderon2, Ulrike Gerdemann2, Connor McGuckin2, Bruce Blazar3, Victor Tkachev4 and Leslie Kean2, 1Division of Immunology, Boston Children's Hospital, Boston, MA, 2Division of Hematology-Oncology, Boston Children's Hospital, Boston, MA, 3Division of Pediatric Blood & Marrow Transplant & Cellular Therapy, University of Minnesota, Minneapolis, MN, 4Center for Transplantation Sciences, Massachusetts General Hospital, Boston, MA
Background/Purpose: Chimeric Antigen Receptor Regulatory T cells (CAR-Tregs) are an emerging strategy to restore immune tolerance during auto- or allo-immune conditions. However, most autoimmune diseases do not have a single or consistent pathogenic antigen to target. OX40L is a co-stimulatory protein expressed on activated antigen presenting cells (APCs). Polymorphisms in OX40L that lead to increased expression are associated with systemic lupus erythematosus (SLE), and the frequency of OX40L+ APCs tracks with disease activity in SLE patients. OX40L may thus serve as an ideal CAR-Treg target in SLE.
Methods: We engineered a CAR construct containing a single chain variable fragment of anti-OX40L IgG (αOX40L scFv CAR) under transcriptional control of the FOXP3 promoter to constrain CAR expression to canonical Treg. CAR-Tregs were compared with polyclonal Control-Tregs, transduced with a Neon Green reporter-encoding construct, for key features, including expression of immune regulatory proteins, suppression of T cell activation, and inhibition of APC functions in vitro.
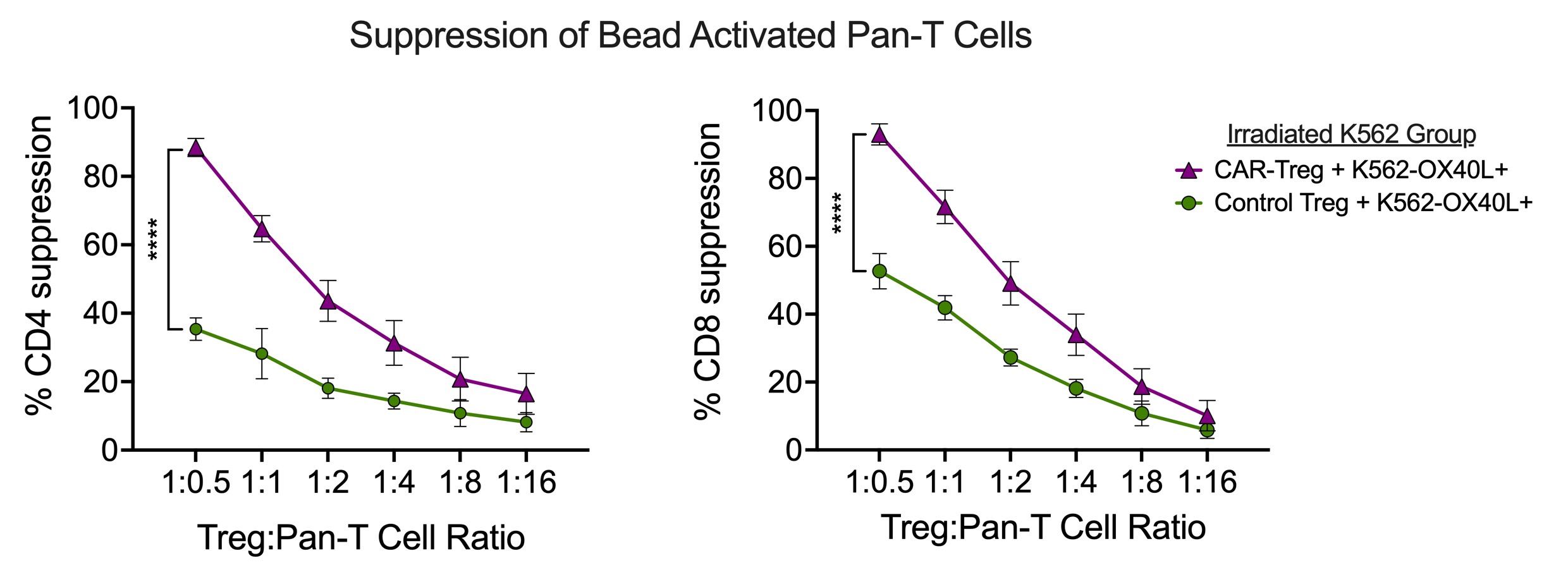
Results: αOX40L scFv CAR expression was stable and selectively expressed in FOXP3+ Tregs during in vitro expansion over three weeks. CAR-Tregs were strongly activated by OX40L+ K562 cells (Figure 1), which, relative to wild type K562 cells, drove expression of Treg-associated immune regulatory proteins LAG3 (5.6-fold), CTLA4 (14.8-fold), GARP (6.9-fold), and LAP (5.5-fold) without induction of pro-inflammatory cytokines (IL-2, TNFα, IL-17A, IFNγ). CAR-Tregs more potently suppressed anti-CD3/CD28 mAb mediated CD4+ and CD8+ T cell proliferation than Control-Tregs (maximum suppression at a 2:1 Treg:T-cell responder ratio: 88.5%±3.7% vs.35.3±4.6% for CD4+ responders, p < 0.0001) (Figure 2). CAR-Tregs also led to a greater reduction of OX40L on the surface of monocyte-derived dendritic cells (mo-DCs) relative to Control-Tregs as a result of enhanced OX40L trogocytosis – findings confirmed using OX40L+ K562 cells (Figure 3).
Conclusion: We designed a novel CAR-Treg stimulated by OX40L on activated APCs. The αOX40L scFv CAR was selectively expressed in Tregs due to control by a FOXP3 promoter, and CAR-Tregs preserved their anti-inflammatory expression profile. CAR-Tregs had superior in vitro suppression of both activated T cells and dendritic cells relative to Control-Tregs. This may be due to universal and potent activation of CAR-Tregs vs. polyclonal Control-Tregs but also from CAR mediated trogocytosis of OX40L on APCs making it unavailable for T-cell co-stimulation. Future work will be aimed at designing a mouse OX40L targeted CAR-Treg to assess in traditional murine SLE models. Overall, we demonstrate a unique approach to CAR-Treg design for treating autoimmune disorders by directing CAR-Treg against activated, disease-associated APCs.
.jpg)
.jpg)
H. Wobma: None; X. Rui: None; F. Alvarez-Calderon: None; U. Gerdemann: allovir, 9, 11, Pfizer, 3, Tessara Therapeutics, 5; C. McGuckin: None; B. Blazar: None; V. Tkachev: None; L. Kean: Bristol-Myers Squibb(BMS), 5, 9, Hifibio, 1, Mammoth bio, 1, Merck/MSD, 5, Novartis, 5, tessera, 5, vertex, 2.
Background/Purpose: Chimeric Antigen Receptor Regulatory T cells (CAR-Tregs) are an emerging strategy to restore immune tolerance during auto- or allo-immune conditions. However, most autoimmune diseases do not have a single or consistent pathogenic antigen to target. OX40L is a co-stimulatory protein expressed on activated antigen presenting cells (APCs). Polymorphisms in OX40L that lead to increased expression are associated with systemic lupus erythematosus (SLE), and the frequency of OX40L+ APCs tracks with disease activity in SLE patients. OX40L may thus serve as an ideal CAR-Treg target in SLE.
Methods: We engineered a CAR construct containing a single chain variable fragment of anti-OX40L IgG (αOX40L scFv CAR) under transcriptional control of the FOXP3 promoter to constrain CAR expression to canonical Treg. CAR-Tregs were compared with polyclonal Control-Tregs, transduced with a Neon Green reporter-encoding construct, for key features, including expression of immune regulatory proteins, suppression of T cell activation, and inhibition of APC functions in vitro.
Results: αOX40L scFv CAR expression was stable and selectively expressed in FOXP3+ Tregs during in vitro expansion over three weeks. CAR-Tregs were strongly activated by OX40L+ K562 cells (Figure 1), which, relative to wild type K562 cells, drove expression of Treg-associated immune regulatory proteins LAG3 (5.6-fold), CTLA4 (14.8-fold), GARP (6.9-fold), and LAP (5.5-fold) without induction of pro-inflammatory cytokines (IL-2, TNFα, IL-17A, IFNγ). CAR-Tregs more potently suppressed anti-CD3/CD28 mAb mediated CD4+ and CD8+ T cell proliferation than Control-Tregs (maximum suppression at a 2:1 Treg:T-cell responder ratio: 88.5%±3.7% vs.35.3±4.6% for CD4+ responders, p < 0.0001) (Figure 2). CAR-Tregs also led to a greater reduction of OX40L on the surface of monocyte-derived dendritic cells (mo-DCs) relative to Control-Tregs as a result of enhanced OX40L trogocytosis – findings confirmed using OX40L+ K562 cells (Figure 3).
Conclusion: We designed a novel CAR-Treg stimulated by OX40L on activated APCs. The αOX40L scFv CAR was selectively expressed in Tregs due to control by a FOXP3 promoter, and CAR-Tregs preserved their anti-inflammatory expression profile. CAR-Tregs had superior in vitro suppression of both activated T cells and dendritic cells relative to Control-Tregs. This may be due to universal and potent activation of CAR-Tregs vs. polyclonal Control-Tregs but also from CAR mediated trogocytosis of OX40L on APCs making it unavailable for T-cell co-stimulation. Future work will be aimed at designing a mouse OX40L targeted CAR-Treg to assess in traditional murine SLE models. Overall, we demonstrate a unique approach to CAR-Treg design for treating autoimmune disorders by directing CAR-Treg against activated, disease-associated APCs.
.jpg)
Figure 1: Activation of Treg suppressive program in OX40L-exposed CAR-Tregs. OX40L+ K562 cells led to a suppressive program in CAR-Tregs that was not seen in Control Tregs or when wild type (WT) K562 cells were used as stimulators. Expression of suppressive and inflammatory markers was measured by flow cytometry with the average values shown on radar plots.

Figure 2: Superior T cell suppression by CAR-Tregs. CAR-Tregs exhibited a superior in vitro capability to suppress anti-CD3/CD28 mAb (bead) mediated CD4+ (left) and CD8+ (right) T cell proliferation. Tregs were co-cultured with irradiated OX40L+ K562 cells for one day prior to assay. Percent suppression was calculated based on cell division, comparing the ‘with Treg’ to the ‘no Treg’ conditions.
.jpg)
Figure 3. Superior trogocytosis of OX40L by CAR-Tregs. A. %OX40L+ cells gated on CD83+CD14+CD4- mo-DCs after co-culture with either Control- or CAR-Tregs for four days. B. Time-course assay showing rapid loss of OX40L from OX40L+ K562 cells (top) and gain on Control- or CAR-Tregs (bottom) upon co-culture. E:T indicates effector (Treg) to target (K562) cell ratio.
H. Wobma: None; X. Rui: None; F. Alvarez-Calderon: None; U. Gerdemann: allovir, 9, 11, Pfizer, 3, Tessara Therapeutics, 5; C. McGuckin: None; B. Blazar: None; V. Tkachev: None; L. Kean: Bristol-Myers Squibb(BMS), 5, 9, Hifibio, 1, Mammoth bio, 1, Merck/MSD, 5, Novartis, 5, tessera, 5, vertex, 2.



